Clear Sky Science · en
MLKL in liver parenchymal cells promotes liver cancer in murine metabolic dysfunction-associated steatotic liver disease
Why a fatty liver can turn into cancer
As more people develop obesity and type 2 diabetes, a growing number also develop a fatty, inflamed liver condition now called metabolic dysfunction-associated steatohepatitis (MASH). Some of these patients eventually go on to liver cancer, but we still do not fully understand why. This study looks at a protein called MLKL inside liver cells and asks a simple question with big implications: does MLKL help push a sick, fatty liver toward cancer, and could turning it off make liver cancer less likely?
A closer look at liver damage in a modern epidemic
Liver cancer is now one of the top killers among cancers worldwide, and much of this rise is driven by fatty liver disease linked to poor diet and metabolic problems. In MASH, liver cells are overloaded with fat, stressed, and surrounded by chronic inflammation. Over years, this environment can damage DNA and allow mutant cells to grow into tumors. The protein MLKL is best known for driving a violent form of cell death that bursts cells open and fuels inflammation. Because of this, scientists suspected that MLKL might be one of the switches that turn long‑lasting liver injury into full-blown liver cancer.
Testing MLKL’s role in a mouse model of fatty liver cancer
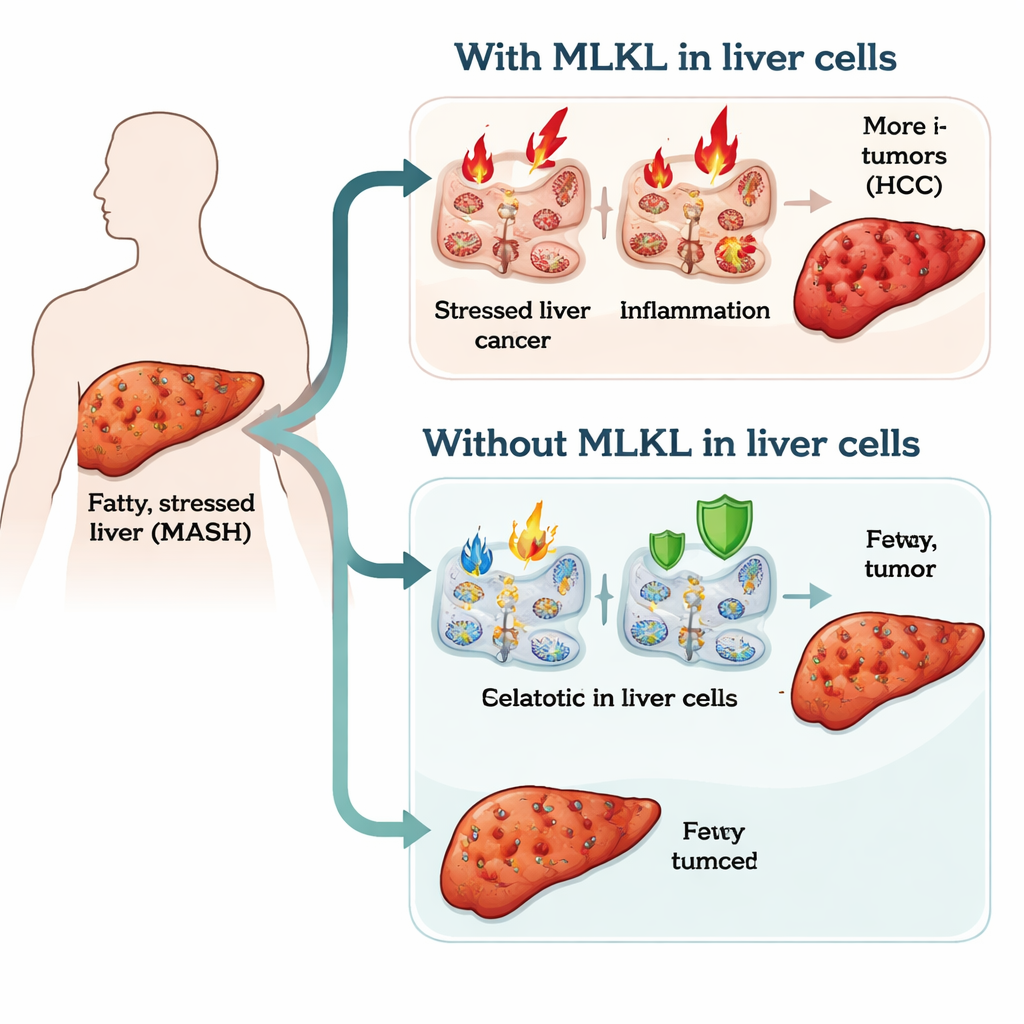
To probe MLKL’s role, the researchers engineered mice in which MLKL was specifically removed from liver parenchymal cells – mainly the working liver cells called hepatocytes – while leaving other tissues intact. All mice were made diabetic shortly after birth and then fed either a regular diet or a high‑fat, high‑sugar diet that mimics the food environment driving human MASH. Over 12 weeks, this diet reliably caused fatty liver, mild scarring, and, importantly, liver tumors resembling human hepatocellular carcinoma. This allowed the team to directly compare normal mice with MLKL‑deficient liver cells to see how the disease unfolded in each case.
Same fatty liver, but delayed inflammation and fewer tumors
Surprisingly, taking MLKL out of liver cells did not prevent the early stages of disease: both groups of mice gained weight similarly, accumulated fat in the liver, and developed comparable mild fibrosis. The striking differences appeared in how quickly inflammation and cancer emerged. Mice with normal MLKL showed an earlier rise in liver inflammatory signals, more activated immune cells in the liver and spleen, and enlarged spleens – signs of a strong, body‑wide inflammatory response. In contrast, mice lacking MLKL in liver cells had a delayed inflammatory phase and fewer highly activated immune cells at early time points. Over time, this translated into fewer and smaller liver tumors, a lower rate of advanced precancerous lesions, and a markedly reduced incidence of full hepatocellular carcinoma.
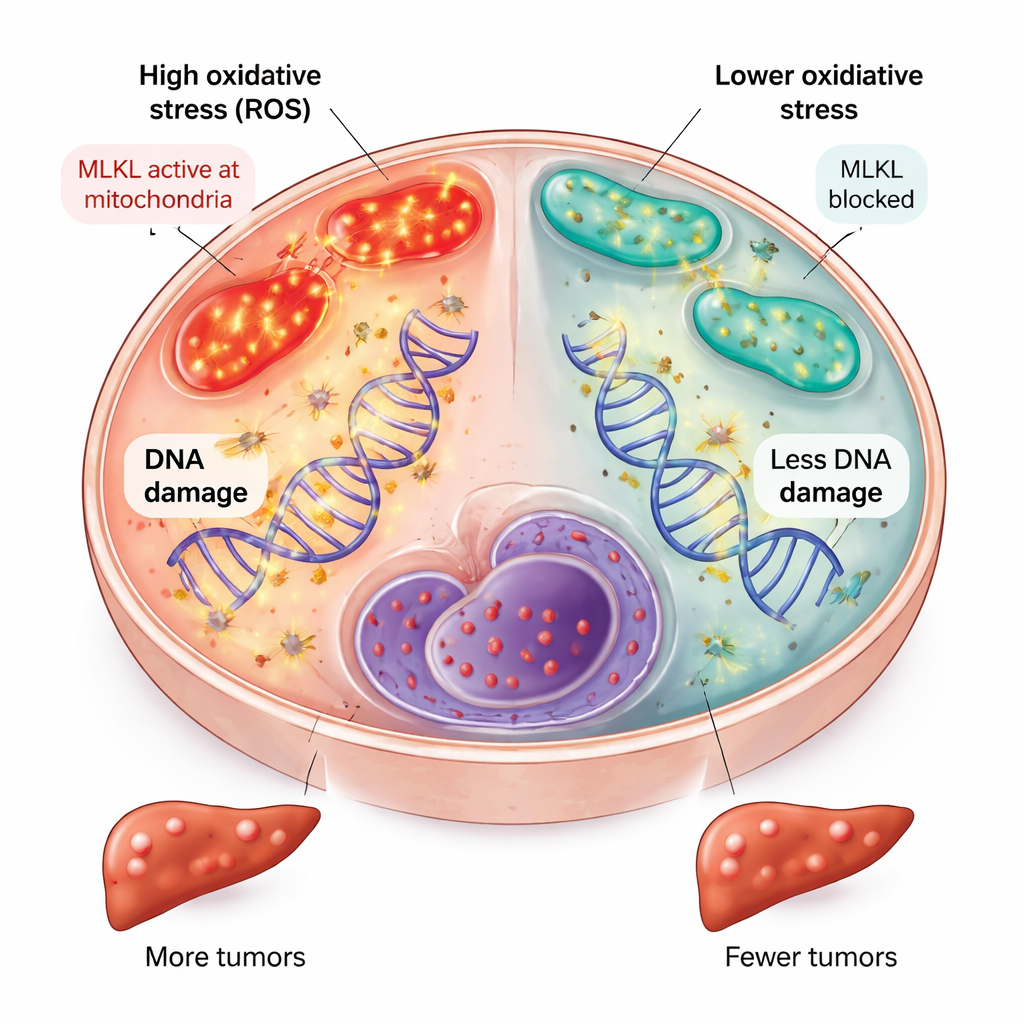
Less oxidative stress and DNA damage without MLKL
The team then asked why the absence of MLKL in liver cells slowed cancer development. They found that, early in the disease course, livers lacking MLKL showed lower expression of genes turned on by oxidative stress, reduced biochemical signs of metabolic stress in certain fats called sphingolipids, and a more balanced state of coenzyme Q in mitochondria, the cell’s energy factories. These changes all point to fewer damaging reactive oxygen species. Consistent with this, liver cells without MLKL accumulated less oxidative DNA damage, as shown by reduced staining of a damage marker in cell nuclei. Because long‑term oxidative DNA damage is a key driver of cancer‑causing mutations, dialing down this stress likely contributed to the lower tumor burden.
What this could mean for future treatments
Together, these findings suggest that MLKL inside liver parenchymal cells acts as a quiet promoter of liver cancer in the setting of fatty, inflamed liver disease. It appears to do so not simply by killing cells, but also by disturbing mitochondrial balance, boosting oxidative stress, and helping sustain damaging inflammation and DNA injury. For patients, the message is that MLKL may be an attractive drug target: blocking its action specifically in liver cells could help prevent or delay MASH‑related liver cancer. At the same time, because MLKL plays other helpful roles in immune cells, any future therapy will need to be precisely aimed so that turning off this protein protects the liver without weakening the body’s broader defenses.
Citation: Imerzoukene, G., Kara-Ali, G.H., Heitz-Marchaland, C. et al. MLKL in liver parenchymal cells promotes liver cancer in murine metabolic dysfunction-associated steatotic liver disease. Cell Death Dis 17, 229 (2026). https://doi.org/10.1038/s41419-026-08458-x
Keywords: fatty liver disease, liver cancer, inflammation, oxidative stress, MLKL protein