Clear Sky Science · en
M5C-driven stabilization of SERPINB5 promotes cervical cancer progression and chemotherapy resistance
Why this research matters for patients
Cervical cancer is still a deadly disease for many women worldwide, especially when it no longer responds to standard chemotherapy drugs like paclitaxel and vincristine. This study uncovers a hidden molecular pathway inside cervical cancer cells that helps them grow, spread, and shrug off these commonly used treatments. By revealing this pathway, the work points to new ways doctors might predict which tumors will resist therapy and how to design drugs that restore sensitivity.
A chemical tag on RNA that fuels cancer
Most people are familiar with DNA as the carrier of genetic information, but cells also rely on RNA as a working copy of genetic instructions. The authors focused on a small chemical tag on RNA called 5-methylcytosine (m5C). Using high-resolution mapping in patient samples, they found that cervical tumors carry many more of these m5C marks on their RNA than normal cervical tissue. These marks were especially common in segments of RNA that code for proteins, and they tended to appear on genes linked to cell growth, invasion, and the ability of cancer cells to escape the immune system. This pattern suggests that the cancer cells are rewiring their RNA chemistry to boost programs that drive malignancy.
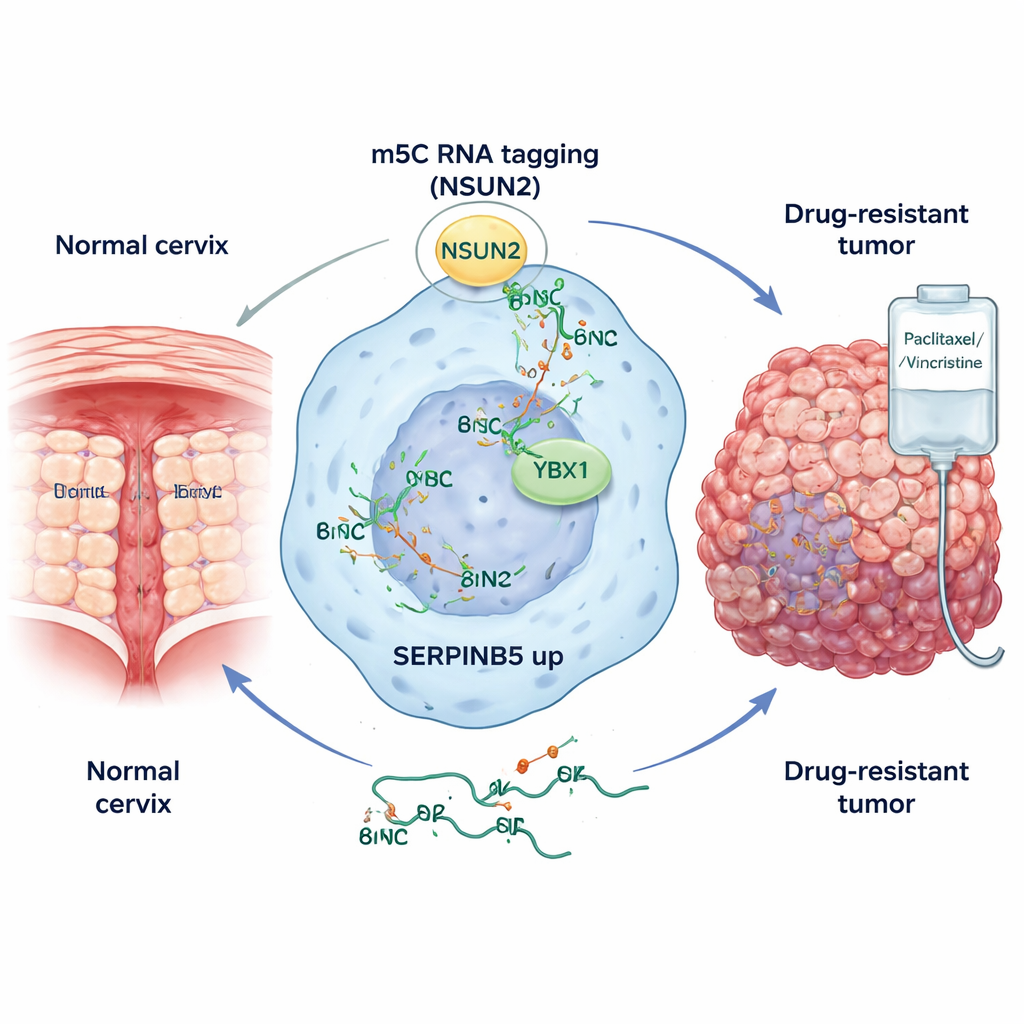
Spotlighting a key troublemaker: SERPINB5
To work out which m5C-marked RNAs actually matter for cancer behavior, the team combined several advanced methods: bulk RNA sequencing, spatial transcriptomics that shows where genes are active within a tumor slice, and single-cell sequencing that profiles individual cells. They then built a custom computer model, MORGAN, to rank candidate genes whose activity rises together with m5C marks. Among several cancer-linked genes, one stood out: SERPINB5, also called maspin. In cervical tumors, SERPINB5 was highly expressed in a distinct subset of cancer cells, rarely seen in normal tissue. These SERPINB5-positive cells were packed in tumor regions rich in fast-dividing cells and showed gene signatures tied to DNA replication, changes in cell shape, and the ability to remodel surrounding tissue.
How a methylation circuit stabilizes SERPINB5
The researchers then asked how SERPINB5 becomes so abundant in cervical cancer. They discovered that an enzyme called NSUN2 is largely responsible for writing m5C marks onto the SERPINB5 RNA. In tumor samples and cell lines, NSUN2 levels were elevated, and its preferred sequence motif matched the m5C sites found on SERPINB5 transcripts. When NSUN2 was reduced or genetically knocked out, SERPINB5 RNA became less stable and its protein levels dropped, but only when NSUN2 retained its catalytic activity. A second protein, YBX1, acted as a molecular reader: it selectively bound to the m5C-marked SERPINB5 RNA and further protected it from decay. Together, NSUN2 and YBX1 form an RNA methylation circuit that keeps SERPINB5 levels high in cancer cells.
A driver of aggressive growth and drug resistance
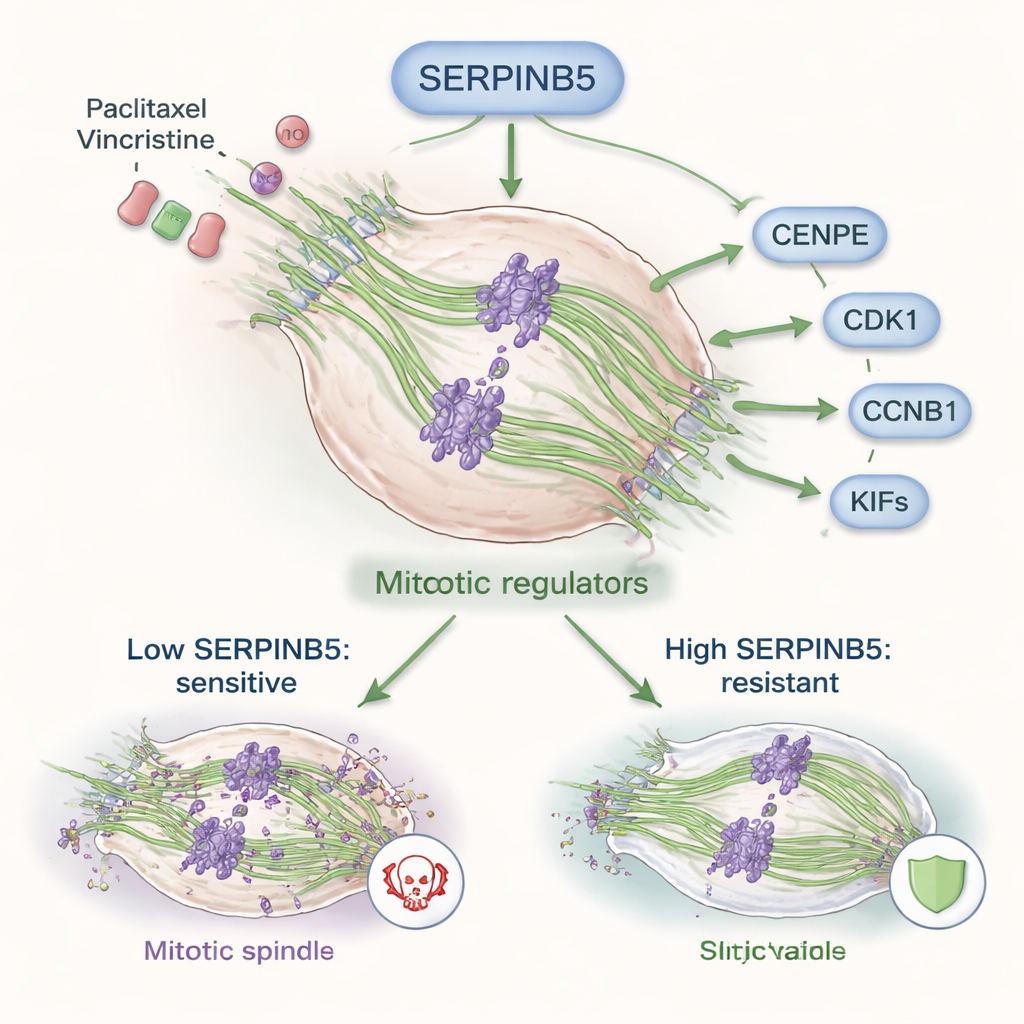
Functional tests revealed just how much SERPINB5 matters for tumor behavior. When the team reduced SERPINB5 in cervical cancer cell lines, the cells divided more slowly, migrated and invaded less, and formed much smaller tumors in mice. Gene analyses showed that SERPINB5 helps maintain stem cell–like traits, supports blood vessel formation, and encourages cells to transition into more mobile, invasive states. Crucially, SERPINB5 also rewired how cells handle division: it boosted the production of mitotic regulators and microtubule motor proteins such as CENPE, CDK1, and CCNB1, which oversee the spindle machinery that pulls chromosomes apart during cell division.
Making chemotherapy less effective
Paclitaxel and vincristine kill cancer cells mainly by disrupting microtubules, the structural fibers that form the mitotic spindle. By jamming this machinery, the drugs force cells into a prolonged, deadly pause in division. The study shows that high SERPINB5 levels help cancer cells push through this blockade. When SERPINB5 was knocked down, cells became more sensitive to both drugs, with lower doses needed to reduce viability. The same effect was seen when NSUN2 was eliminated, and reintroducing SERPINB5 into NSUN2-deficient cells restored much of the drug resistance. Patient tumor samples that had failed paclitaxel-based therapy also showed higher SERPINB5 protein levels than drug-sensitive tumors, underscoring its clinical relevance.
What this means for future treatment
To a lay reader, the key message is that some cervical cancers survive and grow by protecting a single, influential RNA and its protein product, SERPINB5, using a chemical tag system on RNA. This NSUN2–YBX1–SERPINB5 axis acts like a molecular shield, helping tumors divide rapidly and resist frontline microtubule-targeting chemotherapy. Targeting this axis—by inhibiting NSUN2, blocking YBX1’s binding, directly lowering SERPINB5, or monitoring SERPINB5 as a biomarker—could make standard drugs work better and help identify patients most likely to benefit from specific treatments.
Citation: Liu, J., Zhou, L., Yao, P. et al. M5C-driven stabilization of SERPINB5 promotes cervical cancer progression and chemotherapy resistance. Cell Death Dis 17, 215 (2026). https://doi.org/10.1038/s41419-026-08453-2
Keywords: cervical cancer, chemotherapy resistance, RNA methylation, SERPINB5, NSUN2