Clear Sky Science · en
Proximity proteomics reveals OTUD6B regulation of stress granule dynamics through coalescence with VCP/p97
How Cells Cope When Life Gets Rough
Every cell in your body has to weather storms—heat, toxins, lack of nutrients, viral attacks. This paper explores how cells temporarily "pause and protect" their genetic messages during such stress and reveals a previously unknown player, an enzyme called OTUD6B, that helps control this emergency response. Because problems in this system are tied to aging and brain disorders such as neurodegeneration, understanding how it works could point to new ways to keep cells healthier for longer.
Little Droplets That Guard Cell Messages
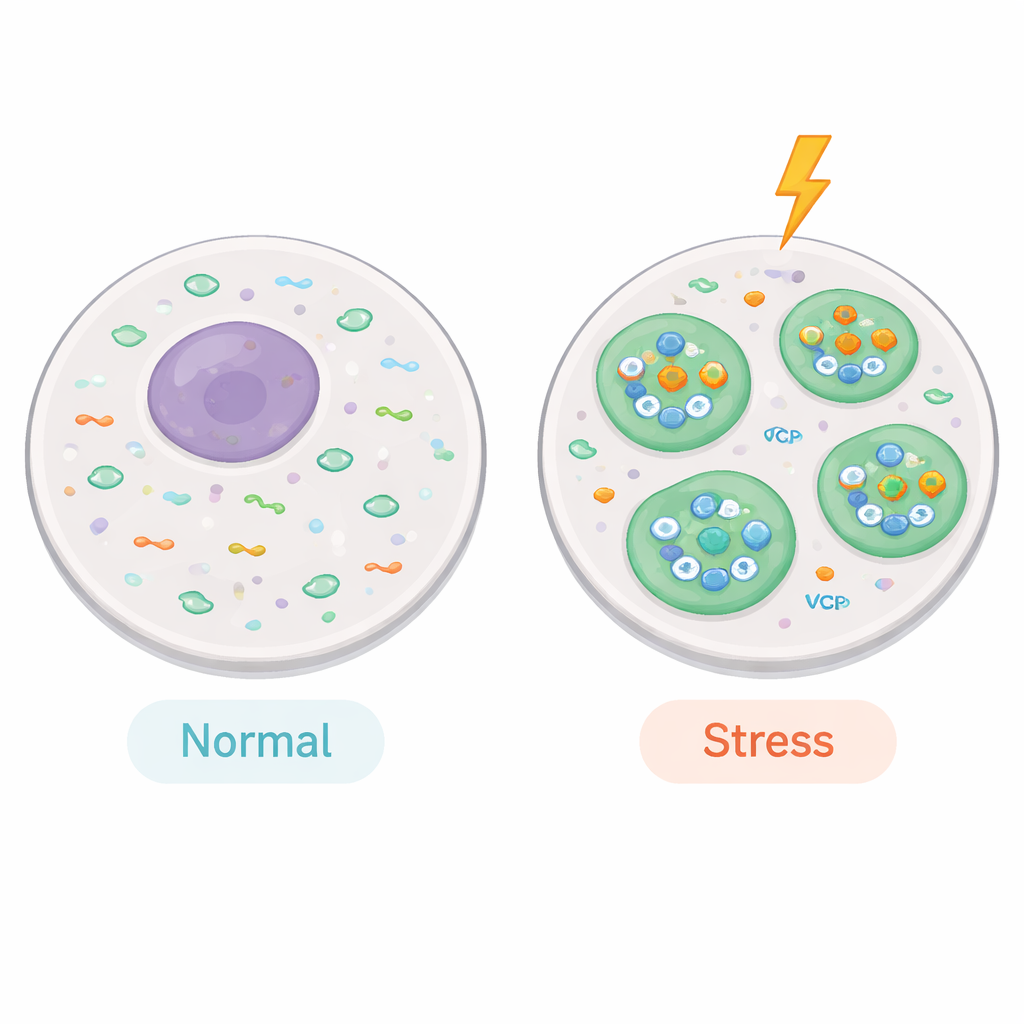
When cells are stressed, they rapidly gather certain molecules into tiny droplets called stress granules. These are not membrane-bound bubbles like classic organelles, but liquid-like clumps where RNA and proteins condense together. Inside, key genetic messages (mRNAs) and the machinery that reads them are parked for safekeeping until the danger passes. Stress granules have a dense "core" and a more changeable "shell" of surrounding proteins. While scientists have studied many core components, they know much less about the shell, which is thought to fine-tune when granules form and how quickly they disappear.
Spotlighting a New Regulator, OTUD6B
The researchers focused on OTUD6B, an enzyme that removes small molecular tags called ubiquitin from proteins and has been linked to cell growth, immunity, and brain development. Because OTUD6B’s relatives interact with the protein-making machinery, the team wondered if it might also influence stress granules, which are rich in pieces of ribosomes (the cell’s protein factories). Using two large-scale protein mapping methods—classic pull-down experiments and a modern proximity-labeling approach—they cataloged hundreds of molecules that associate with OTUD6B inside human cells. Both maps strongly pointed to stress granules and RNA-processing factors, suggesting OTUD6B is deeply embedded in this protective droplet network.
Helping Granules Form—and Then Let Go
Microscope imaging confirmed that OTUD6B moves into stress granules when cells are exposed to oxidative damage (arsenite) or heat shock. When the team artificially boosted OTUD6B levels, stress granules appeared faster in the first minutes after stress. When they dialed OTUD6B down, granule formation lagged behind. The same pattern held true for both types of stress, and in different cell lines, pointing to a general role rather than a niche effect. Importantly, a mutant version of OTUD6B that cannot perform its normal enzyme chemistry only partly rescued these defects, showing that its ability to trim ubiquitin tags contributes to its control over early granule assembly.
Guiding a Cellular Unfolding Machine
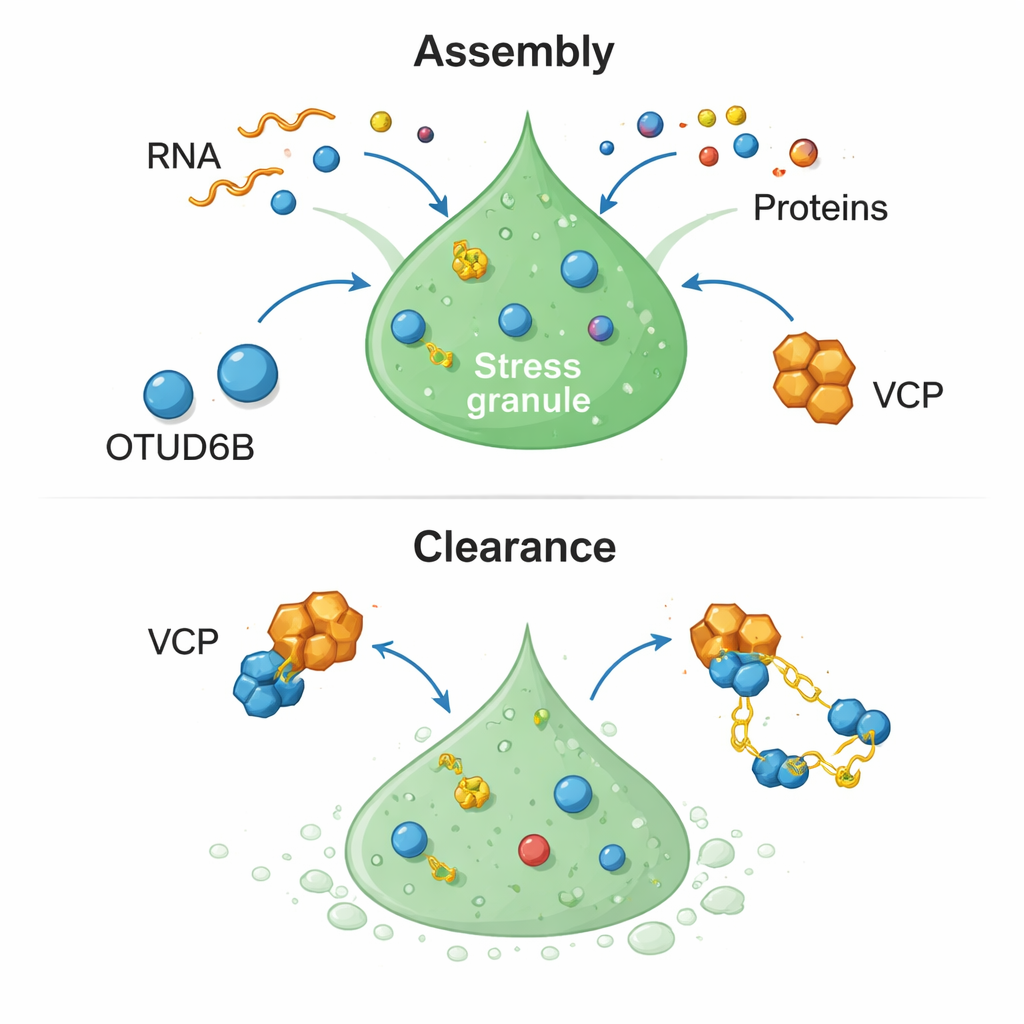
The story does not stop at granule formation. Normally, once stress is gone, stress granules must melt away so that mRNAs can re-enter everyday use. Cells lacking OTUD6B showed a sluggish clean-up: granules persisted long after the stressor had been removed. To uncover why, the scientists examined which partners dropped out of granules when OTUD6B was missing. A key hit was VCP (also called p97), a powerful molecular machine that uses chemical energy to extract and unfold proteins and is already known to help dismantle protein clumps. OTUD6B and VCP turned out to be physically linked through flexible, disordered tail regions of each protein. Under stress, VCP was normally drawn into stress granules, but this enrichment was sharply reduced when OTUD6B was silenced, indicating that OTUD6B acts as a recruiter that brings VCP to the right place at the right time.
One Pathway, Two Phases of Control
When VCP itself was blocked—either by genetic knockdown or by a small-molecule drug—cells showed nearly the same early delay in stress granule formation as they did when OTUD6B was lost, and the combined blockade was no worse than either treatment alone. That pattern suggests OTUD6B and VCP function in the same pathway. OTUD6B does not simply change how much VCP is made; instead, it helps usher VCP into stress granules, where VCP’s energy-driven activity contributes both to building properly composed droplets early on and to dismantling them once the threat is gone. A catalytically dead OTUD6B mutant bound VCP more weakly and less effectively supported this process, underscoring a fine interplay between physical recruitment and enzyme activity.
Why This Matters for the Aging and Diseased Brain
In everyday terms, OTUD6B acts like a dispatcher that calls in a clean-up crew (VCP) to manage protective protein-RNA droplets during crisis. It speeds up their formation when needed and ensures they are efficiently cleared when conditions improve. Faulty stress granule clearance and toxic protein clumps are hallmarks of many neurodegenerative diseases, and inherited OTUD6B defects already cause a form of intellectual disability. By linking OTUD6B directly to the life cycle of stress granules, this work highlights a new molecular lever that could, in principle, be targeted to tune how cells respond to stress and to reduce long-term damage from lingering aggregates.
Citation: Yang, D., Liu, Y., Hong, Y. et al. Proximity proteomics reveals OTUD6B regulation of stress granule dynamics through coalescence with VCP/p97. Cell Death Dis 17, 206 (2026). https://doi.org/10.1038/s41419-026-08451-4
Keywords: stress granules, OTUD6B, VCP p97, cell stress response, neurodegeneration