Clear Sky Science · en
Circular RNAs in metabolic health: bridging the gap between molecular biology and therapy
Why tiny RNA loops may matter for weight and health
Obesity is often framed as a simple problem of willpower and calories, yet many people struggle despite diet and exercise. This article explains how a newly appreciated class of genetic molecules—circular RNAs, or circRNAs—helps control whether our fat cells store energy or burn it. By shifting white “storage” fat toward brown, heat‑producing fat, these RNA loops could one day inspire precision treatments for obesity and related diseases, from type 2 diabetes to fatty liver disease.
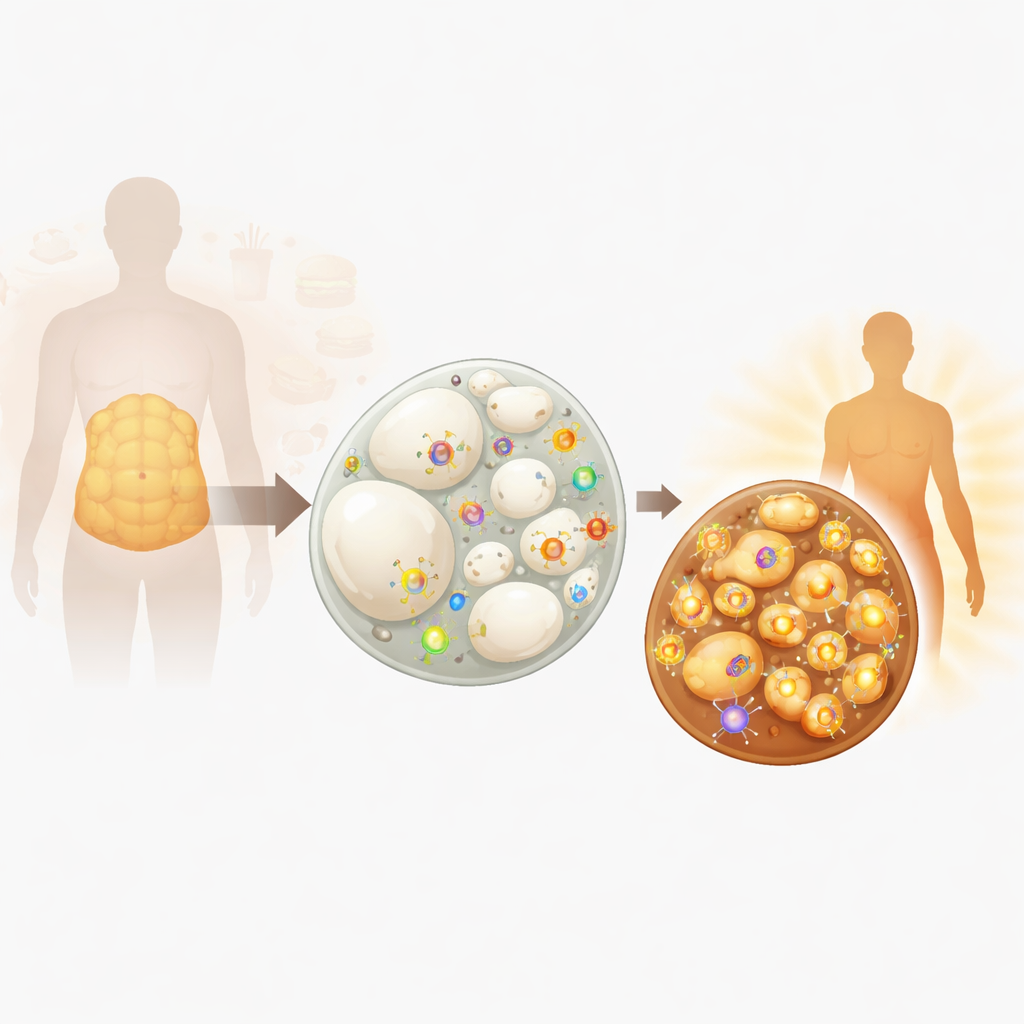
The two faces of body fat
Not all body fat behaves the same. White fat, which cushions organs and piles up around our waist, mainly serves as an energy warehouse: its cells hold one giant droplet of fat and contain relatively few mitochondria, the cell’s power plants. Brown fat, concentrated in the upper back and neck, is packed with mitochondria that burn fuel to produce heat, helping keep us warm and limiting weight gain. Under certain conditions—such as cold exposure, overeating, or exercise signals—white fat can remodel into “beige” fat, an in‑between type that learns brown fat’s trick of burning calories. This remodeling, called fat browning, is now seen as a promising way to counter metabolic disease.
What makes circular RNAs special
RNA is usually thought of as a straight strand that carries genetic instructions from DNA to protein. CircRNAs defy that textbook picture: their ends are joined to form loops. This closed ring structure makes them unusually stable and able to survive inside cells and even in blood and other body fluids. Far from being genetic junk, circRNAs can soak up microRNAs (tiny regulators that silence genes), dock specific proteins, influence how genes are switched on, and in some cases even direct the production of short peptides. Because particular circRNAs are enriched in fat tissue and respond to diet and hormones, they are emerging as key coordinators of how fat cells use and store energy.
How circular RNAs push white fat toward brown
In brown and beige fat, many circRNAs change their activity compared with white fat. Some act like molecular sponges, binding microRNAs that would otherwise block genes needed for breaking down fat or building mitochondria. For example, certain circRNAs free up genes that drive lipolysis (fat breakdown) or activate pathways such as AMPK and mTOR that rewire how cells handle sugars and fats. Others bind directly to proteins that control fat synthesis, oxidation, or heat production, stabilizing helpful enzymes or steering them to new tasks. A growing number of circRNAs even encode small peptides that tweak mitochondrial function or shift how cells process nutrients, nudging white fat cells toward a browner, more energy‑hungry state.
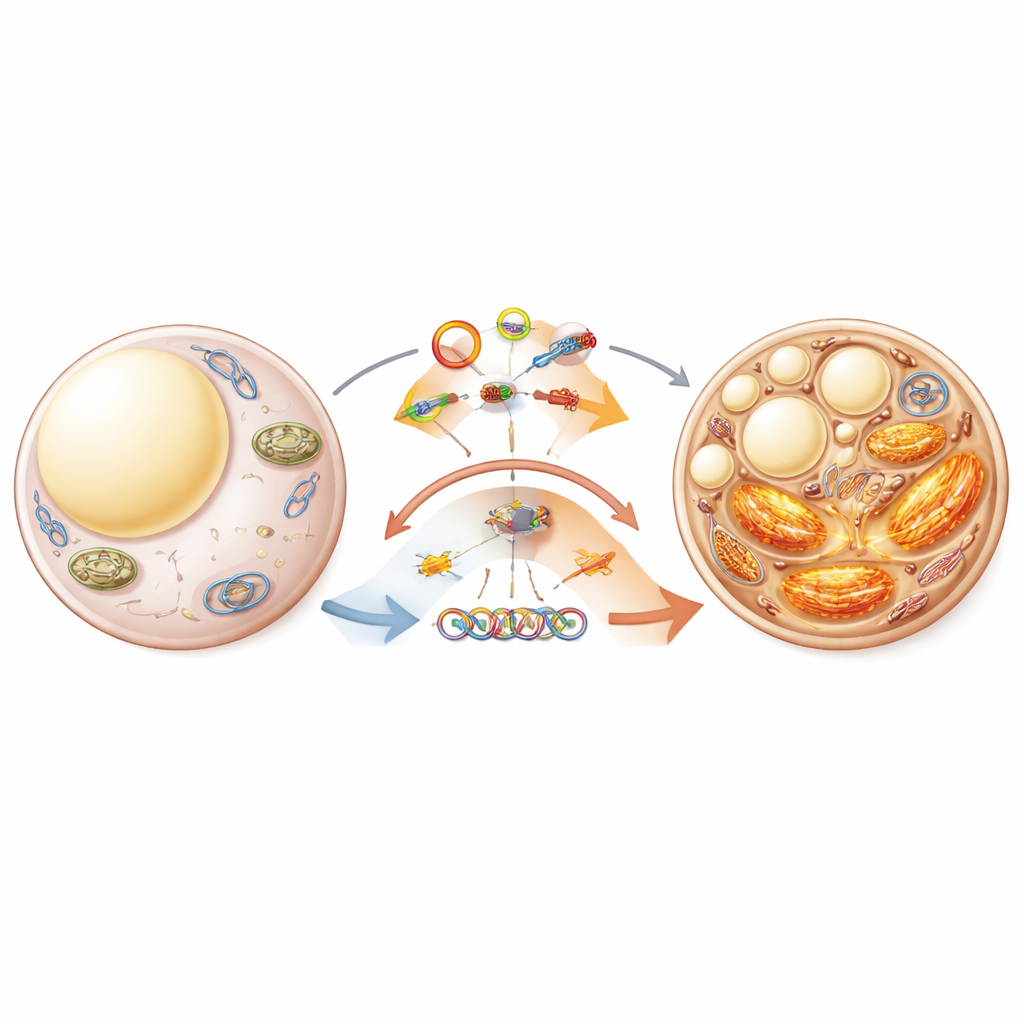
Tuning the cell’s power plants and stress systems
Fat browning depends heavily on mitochondria and on the endoplasmic reticulum, a membrane network that helps manage stress inside cells. The review highlights how circRNAs influence these inner structures. Some circRNAs protect mitochondria by promoting the orderly removal of damaged ones, while others dial this cleanup up or down by interacting with proteins on the mitochondrial surface. CircRNAs also shape how the endoplasmic reticulum responds to overload, which can either block or support the browning program. Because mitochondria and the endoplasmic reticulum physically and functionally cooperate, circRNAs that sit at this crossroads may act as master switches for whether fat cells hoard energy or burn it off as heat.
From lab discovery to future therapies
Thanks to their durability and precision, circRNAs are attracting attention as diagnostic markers, drug targets, and even as therapeutic molecules themselves. Engineered circRNAs can be built to produce beneficial peptides, adjust gene activity, or serve as long‑lasting vaccines, and nanoparticle or exosome carriers are being developed to deliver them into tissues such as fat and liver. The article cautions, however, that many circRNAs differ between species, delivery tools are still crude, and long‑term safety is unknown. Overall, the authors argue that understanding and harnessing circRNAs offers a way to move beyond calorie‑only views of obesity, opening paths to treatments that retrain our fat cells to act more like brown fat and restore metabolic balance.
Citation: Huang, Y., He, T., Zheng, J. et al. Circular RNAs in metabolic health: bridging the gap between molecular biology and therapy. Cell Death Dis 17, 258 (2026). https://doi.org/10.1038/s41419-026-08450-5
Keywords: circular RNA, fat browning, obesity, metabolic disease, brown adipose tissue