Clear Sky Science · en
Oocyte-specific knockout of eIF2 subunits causes apoptosis of mouse oocytes within the early growing follicles via mitochondrial dysfunctions and DNA damage
Why the Life of Egg Cells Matters
Women are born with all the egg cells they will ever have, stored inside tiny structures in the ovary called follicles. When these follicles are lost too quickly, women can develop premature ovarian insufficiency (POI), a condition that leads to early infertility and often menopause-like symptoms at a young age. This study in mice asks a basic but crucial question: what happens inside egg cells when a key step in making new proteins goes wrong, and how can that lead to the early loss of fertility?
The Cell’s Protein-Starting Switch
To stay healthy, egg cells must constantly make the right proteins at the right time. A central part of this process is a three-part molecular machine called eIF2, which helps launch protein production from genetic messages. The researchers selectively turned off two of eIF2’s parts, called eIF2α and eIF2β, only in mouse egg cells at early stages of follicle growth. When either subunit was missing, female mice became completely infertile. Their ovaries were smaller, and the normal progression from early follicles to mature, egg-releasing follicles largely stalled, with far fewer follicles at every later stage. Over time, essentially all follicles were depleted, mimicking a severe, early-onset POI-like condition.
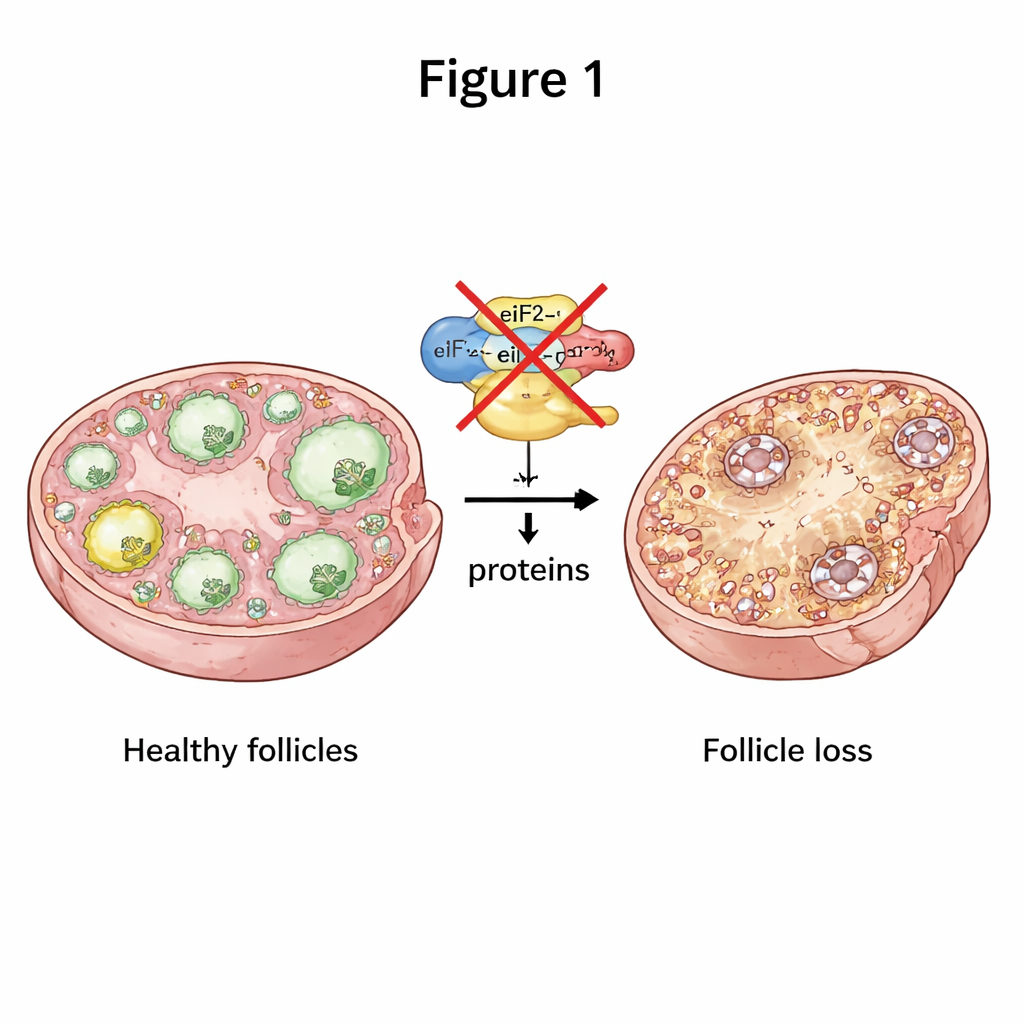
When Communication in the Ovary Breaks Down
Follicles are not just egg cells floating alone; they are tight partnerships between an egg and surrounding “helper” cells called granulosa cells. The team found that without eIF2β, egg cells made far less of several crucial signaling proteins that normally support granulosa cell growth and nutrient delivery. The physical bridges between egg and granulosa cells were disorganized and shortened, and the egg’s surface microvilli looked stunted and damaged under the electron microscope. Granulosa cells in these follicles divided less and died more often. This breakdown in two-way communication meant that the follicles could not grow properly, pushing the ovary toward follicle loss.
Power Failure in the Egg Cell’s Batteries
Mitochondria, often called the cell’s power plants, are especially important in egg cells because they supply the energy needed for growth and later embryo development. In eIF2β-deficient oocytes, the overall rate of new protein production dropped, and many mitochondrial-related proteins were reduced. Mitochondria became abnormally long, clumped near the cell surface, and showed weaker membrane potential, lower energy (ATP) levels, and fewer copies of mitochondrial DNA. At the same time, the cells accumulated high levels of reactive oxygen species (ROS), aggressive oxygen-based molecules that can damage cellular components. Together, these changes revealed a profound collapse of mitochondrial dynamics and function.
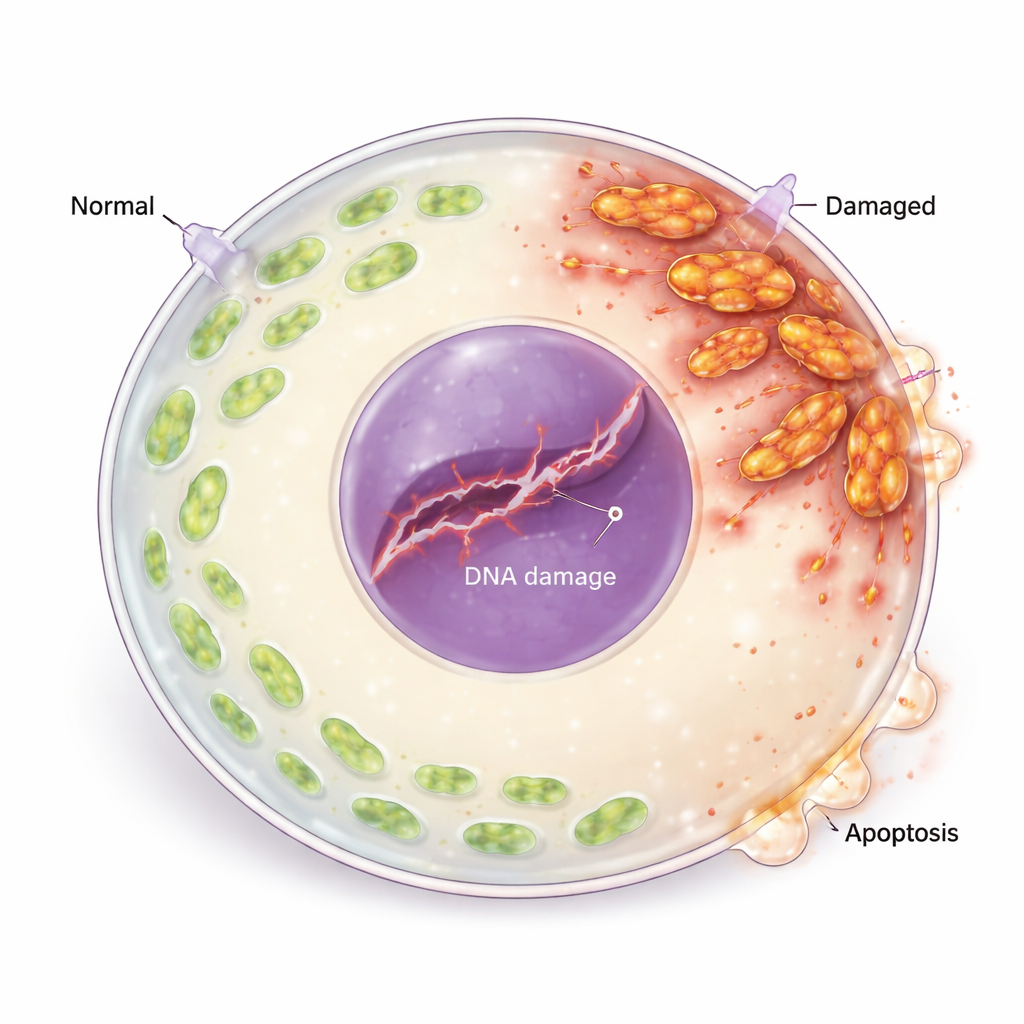
From Oxidative Stress to DNA Damage and Cell Death
Excess ROS did not just harm mitochondria; it also injured the egg cell’s genetic material. The researchers saw increased markers of broken DNA strands and activation of the cell’s DNA damage response machinery, including proteins that sense and signal genetic injury. A key repair protein was reduced, suggesting the damage was not being efficiently fixed. Downstream, the balance of life-and-death regulators shifted: pro-death proteins rose while protective anti-death proteins fell, and egg cells showed clear signs of programmed cell death (apoptosis). When the team treated the oocytes with an antioxidant (N-acetylcysteine), ROS levels dropped, DNA damage and apoptosis markers decreased, and egg maturation improved, directly linking oxidative stress to egg cell loss.
What This Means for Early Ovarian Failure
By following events from a single molecular complex (eIF2) to faulty protein production, mitochondrial breakdown, oxidative stress, DNA damage, and finally egg-cell death, this work outlines a detailed chain of events that can destroy the ovarian reserve. The study strengthens the idea that mutations in translation-starting factors, already seen in some women with POI, can directly harm egg cells through this pathway. It also points to potential ways to help: antioxidants, stress-response modulators, or treatments that protect early follicles (such as anti-Müllerian hormone, which partly preserved dormant follicles in these mice) may one day support fertility in patients whose ovaries are vulnerable to similar stresses.
Citation: Liu, H., Wang, W., Li, B. et al. Oocyte-specific knockout of eIF2 subunits causes apoptosis of mouse oocytes within the early growing follicles via mitochondrial dysfunctions and DNA damage. Cell Death Dis 17, 196 (2026). https://doi.org/10.1038/s41419-026-08449-y
Keywords: premature ovarian insufficiency, oocyte apoptosis, mitochondrial dysfunction, protein translation, reactive oxygen species