Clear Sky Science · en
Transglutaminase 2 exacerbates ovarian cancer survival by directly inactivating GSK3β
Why this research matters for women’s health
Ovarian cancer is one of the deadliest cancers in women because it is usually found late and often returns even after strong chemotherapy. This study uncovers how a little-known protein, transglutaminase 2 (TGase 2), helps ovarian cancer cells survive treatment and spread through the body. By exposing this hidden helper of cancer, the work points to new ways to make existing drugs more effective and to slow or stop deadly metastasis.
A hidden driver inside aggressive tumors
Doctors have long observed that many ovarian cancers become resistant to chemotherapy and spread widely in the abdomen. One major culprit is a process called epithelial–mesenchymal transition (EMT), in which cancer cells loosen their connections, become more mobile, and behave more like invasive “wanderers” than organized tissue cells. The authors show that TGase 2, which is found at very low levels in normal ovary but at much higher levels in ovarian tumors, is strongly tied to this shift. In a large panel of human tissue samples, TGase 2 levels rose steadily from early-stage tumors to advanced and metastatic disease, and its abundance closely matched the activity of many EMT-related genes that control cell movement, growth, and blood vessel formation. 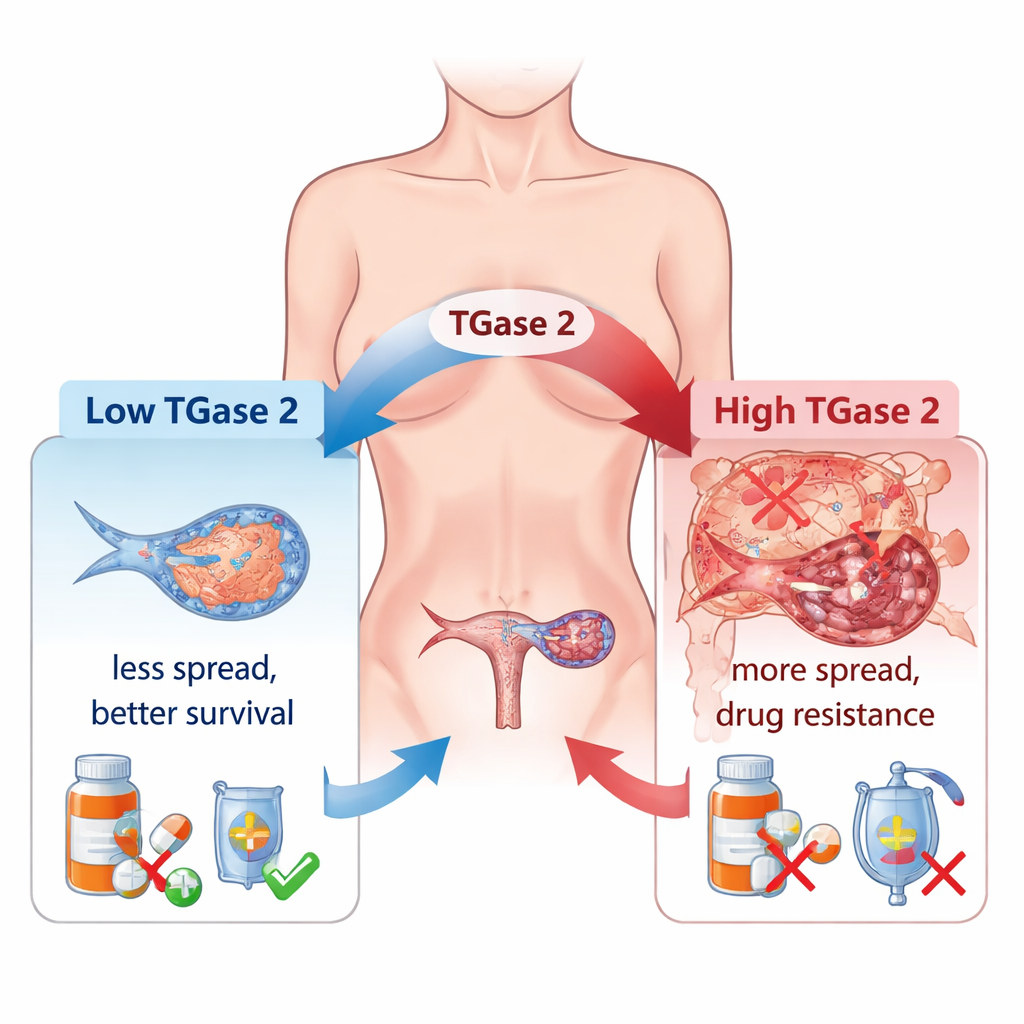
How TGase 2 flips a key safety switch
To understand what TGase 2 actually does inside cancer cells, the researchers focused on its relationship with another protein, GSK3β. Under healthy conditions, GSK3β acts like a brake on invasion: it keeps levels of a signaling molecule called β-catenin low, which in turn restrains genes that drive EMT. The team discovered that TGase 2 physically binds to GSK3β and both weakens its activity and helps send it for destruction through the cell’s recycling system, a pathway called autophagy. When TGase 2 was abundant, ovarian cancer cells migrated more readily and showed higher levels of classic EMT markers such as fibronectin, vimentin, and β-catenin. When the researchers reduced TGase 2 with genetic tools, these invasive features dropped, and β-catenin signaling was dampened, showing that TGase 2 disables a crucial safety switch against metastasis.
From cancer cells to living animals
The team next asked whether blocking TGase 2 could actually slow cancer in animals. They engineered ovarian cancer cells lacking the TGase 2 gene and implanted them into mice. Compared with mice receiving normal cancer cells, animals given TGase 2–deficient cells developed smaller tumor burdens and lived longer. In a separate model that mimics cancer spread through the bloodstream, mice injected with TGase 2–deficient cells developed far fewer and smaller lung metastases. Importantly, these metastatic deposits showed higher levels of GSK3β, fitting the idea that removing TGase 2 restores the natural brake on invasion and helps contain the disease.
Turning a laboratory clue into a drug strategy
Because deleting a gene is not practical in patients, the authors tested a small molecule called streptonigrin, previously known to bind the same end of TGase 2 used to grab other proteins. They found that streptonigrin could disrupt the TGase 2–GSK3β interaction in cells, reduce their co-localization, and limit cancer cell migration. In mouse models of ovarian cancer, oral streptonigrin treatment slowed tumor growth, shrank metastatic lesions, and extended survival. Most strikingly, when streptonigrin was combined with standard chemotherapy drugs such as cisplatin or paclitaxel, the mice lived weeks longer than with chemotherapy alone. Chemotherapy by itself tended to raise TGase 2 levels and lower GSK3β, indirectly pushing cells toward EMT, but adding the TGase 2 blocker reversed this trend and curbed the cells’ newfound mobility. 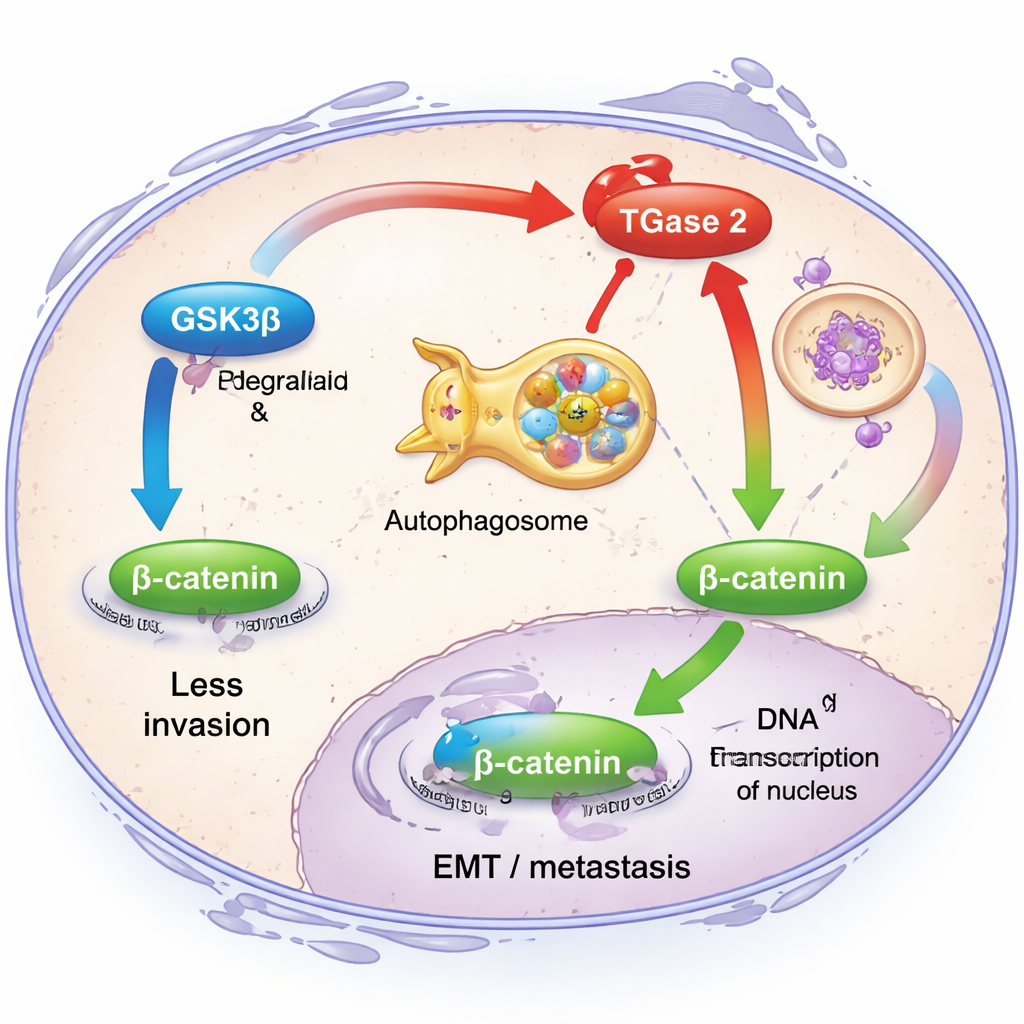
What this could mean for future treatment
For a non-specialist, the take-home message is that ovarian cancer cells exploit TGase 2 to switch off a built-in anti-metastasis brake and to weather the storm of chemotherapy. By directly inactivating GSK3β and stabilizing β-catenin, TGase 2 helps tumors become more invasive and drug-resistant. This work suggests that drugs aimed at the N-terminal region of TGase 2—like streptonigrin or future, safer compounds modeled on it—could make existing chemotherapies work better, reduce the chance of relapse, and improve survival. In simple terms, targeting TGase 2 may turn off a cancer “escape route,” keeping ovarian tumors more sensitive to treatment and less able to spread.
Citation: Lee, H., Kang, J.H., Kim, H.J. et al. Transglutaminase 2 exacerbates ovarian cancer survival by directly inactivating GSK3β. Cell Death Dis 17, 199 (2026). https://doi.org/10.1038/s41419-026-08447-0
Keywords: ovarian cancer, drug resistance, metastasis, epithelial mesenchymal transition, targeted therapy