Clear Sky Science · en
Inhibition of autoantigen-induced B-cell receptor (BCR) internalization as a therapeutic strategy in diffuse large B cell lymphoma (DLBCL)
Why this matters for patients
Diffuse large B cell lymphoma (DLBCL) is the most common fast-growing blood cancer in adults, and many patients still relapse after standard chemo‑immunotherapy. This study explores a surprising weak spot in a major subset of these tumors: their dependence on a cell‑surface antenna called the B‑cell receptor (BCR). By showing that blocking how this antenna is pulled into the cell can cripple cancer cell survival, the work opens the door to repurposing long‑used anti‑nausea and antipsychotic drugs as targeted helpers against lymphoma.
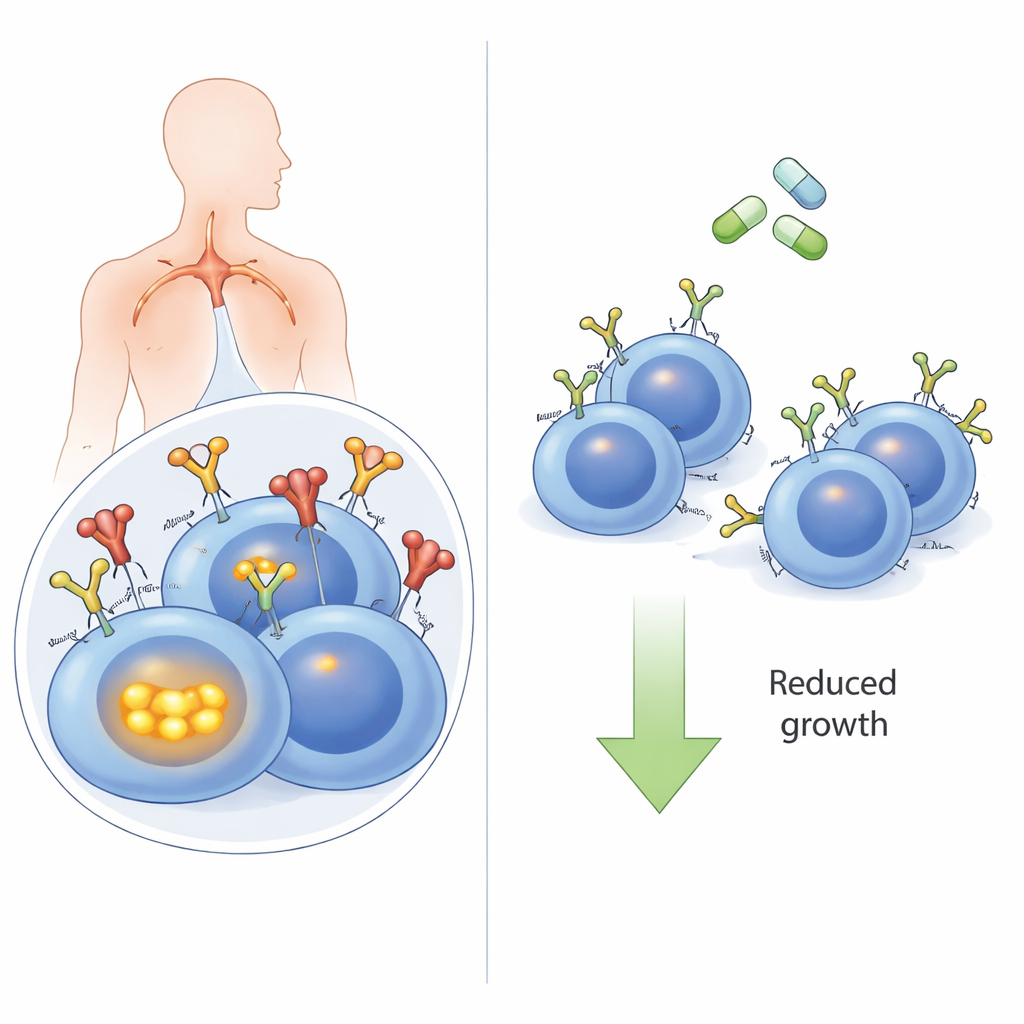
The cancer cell’s antenna
B cells, a type of white blood cell, use the B‑cell receptor on their surface to recognize threats. In many DLBCLs, especially the high‑risk “activated B‑cell” (ABC) subtype, this same receptor is hijacked to send constant “stay alive and grow” messages. Often, these BCRs recognize the body’s own molecules (autoantigens), which act like a stuck doorbell. When autoantigens bind, the BCR does more than just signal at the surface: it is pulled into the cell and joins an internal protein cluster with sensors called TLR9 and MYD88. This super‑complex then switches on NFκB, a powerful growth‑promoting signaling pathway. Until now, it was unclear whether this inward journey of the receptor was truly required for the cancer‑driving signals.
Rewiring the antenna to test its limits
To answer that question, the researchers used CRISPR gene‑editing to precisely change the “tips” of the BCR in lymphoma cell lines. They replaced the natural, self‑reactive recognition regions with versions that recognize ovalbumin, a harmless egg protein used as a laboratory tool. These modified receptors no longer grabbed their usual autoantigens, but could still be triggered in a controlled way with added ovalbumin. In ABC‑type lymphoma cells that normally depend on self‑antigen binding, this switch sharply reduced the activity of many key signaling enzymes and dampened NFκB‑responsive genes. The cells grew more slowly, despite actually having more BCR sitting on their surface, showing that ongoing engagement by autoantigens and subsequent internal signaling are critical for their survival.
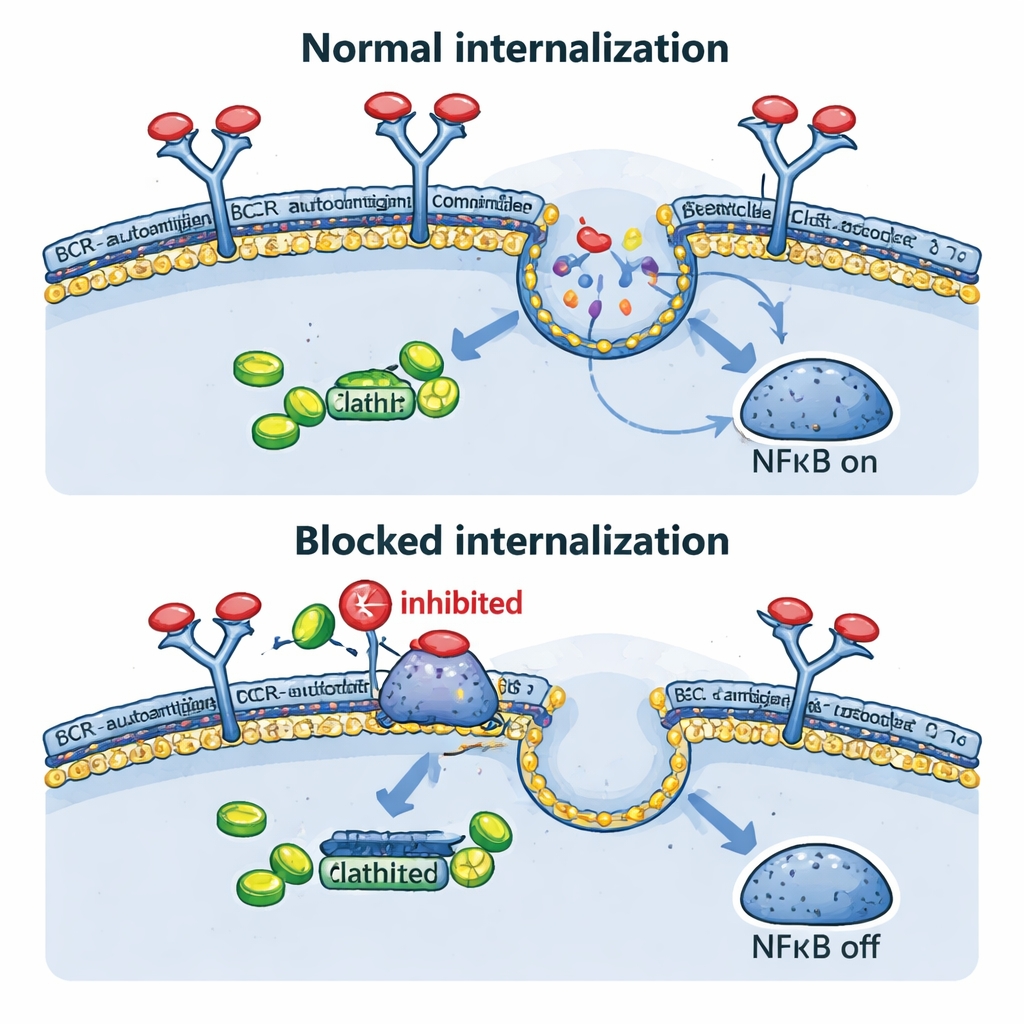
Pulling receptors inside: a crucial step
The team then looked directly at what happens to the BCR after it meets its antigen. In their engineered models, both antibody‑based triggers and ovalbumin caused the receptor to disappear from the cell surface within minutes, confirming rapid internalization. This occurred not only when the antigen floated outside the cell, but also when it was artificially displayed on the same cell’s membrane, mimicking certain real tumor situations. Blocking this inward traffic genetically, using a dominant‑negative form of a protein called dynamin‑2 that is essential for clathrin‑mediated endocytosis, kept BCR on the surface, shrank the internal BCR–TLR9–NFκB complexes, reduced NFκB target genes, and slowed cell growth. Interestingly, when endocytosis was blocked, some “background” BCR signals at the surface increased, suggesting that cancer cells may try to compensate by turning up a weaker, tonic signaling mode.
Old drugs, new tricks
Because building new drugs is slow and expensive, the authors next asked whether existing medicines that block endocytosis could mimic the genetic experiments. Phenothiazines, a family of antipsychotic and anti‑nausea drugs, are known to inhibit dynamin‑2 and clathrin‑dependent uptake of receptors. In lymphoma cells, compounds like prochlorperazine and chlorpromazine raised BCR levels on the surface and strongly reduced antigen‑driven internalization. This led to lower NFκB‑driven gene activity and reduced viability of ABC‑type DLBCL cells, particularly those with intact BCR components. In mice carrying human lymphoma grafts, prochlorperazine at clinically achievable doses significantly slowed tumor growth. Moreover, combining phenothiazines with drugs that block other BCR‑linked enzymes, such as SYK and PI3Kδ, produced stronger cancer‑killing effects than either treatment alone.
What this could mean for treatment
Overall, the study shows that for a substantial subgroup of DLBCLs, the cancer‑driving signals do not simply come from receptors at the cell surface; they critically depend on pulling the BCR–antigen complex inside through a specific endocytosis pathway. Interrupting this step—either by genetic tricks or by phenothiazine drugs—weakens NFκB signaling and impairs tumor cell survival, while potentially sensitizing cells to existing BCR‑pathway inhibitors. Because phenothiazines already have well‑understood dosing and safety as anti‑nausea medicines, this work provides a realistic blueprint for clinical trials that test them, alone or in combination, in patients whose lymphomas show autoantigen‑dependent BCR activity.
Citation: Górniak, P., Polak, A., Rams, A. et al. Inhibition of autoantigen-induced B-cell receptor (BCR) internalization as a therapeutic strategy in diffuse large B cell lymphoma (DLBCL). Cell Death Dis 17, 216 (2026). https://doi.org/10.1038/s41419-026-08446-1
Keywords: diffuse large B cell lymphoma, B cell receptor, endocytosis, phenothiazines, NFkB signaling