Clear Sky Science · en
Time‑resolved multi-omic analysis of paclitaxel exposure in human iPSC‑derived sensory neurons unveils mechanisms of chemotherapy‑induced peripheral neuropathy
Why some cancer drugs hurt nerves
Chemotherapy has helped millions of people survive cancer, but many patients pay a hidden price: months or years of burning pain, tingling, and numbness in their hands and feet. This study asks a simple but important question: what exactly does the widely used cancer drug paclitaxel do to human sensory nerve cells, and can understanding those changes point the way to better prevention and treatment of this nerve damage?
From patients’ cells to lab-grown pain-sensing nerves
Instead of relying on animal experiments, the researchers started with cells from five human donors, including breast‑cancer patients who had received paclitaxel. They reprogrammed these cells into induced pluripotent stem cells and then guided them to become sensory neurons—the same type of nerve cell that carries touch and pain signals from the skin to the spinal cord. These lab‑grown neurons formed long, delicate fibers and showed electrical activity much like real pain‑sensing nerves in the body. The team then exposed them to increasing doses of paclitaxel, mimicking drug levels seen in patients, and tracked cell health over several days. At low concentrations the neurons coped well, but at a clinically relevant dose (100 nM) their survival began to drop after about two days, marking the onset of toxic nerve damage.
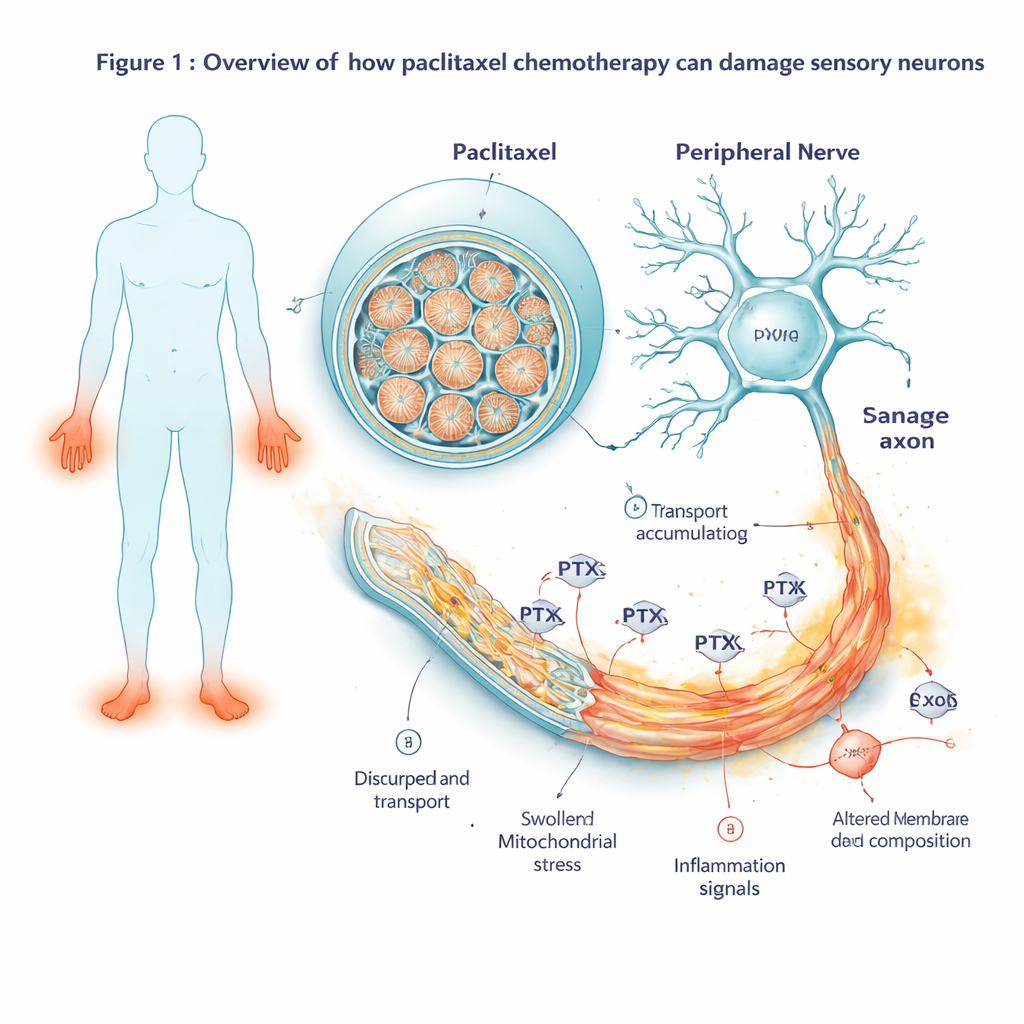
Watching gene activity change over time
To see how the cells responded before they visibly deteriorated, the scientists measured which genes turned on or off at multiple time points from two hours after drug exposure to several days after the drug was washed away. Early on, they saw activation of a classic cellular stress program centered on a gene called JUN. As exposure continued, this stress signal broadened into a full‑blown self‑destruct cascade: genes that push cells toward programmed death were strongly switched on, while protective partners were overwhelmed. At the same time, genes linked to inflammation and pain signaling became more active. The neurons began to produce more inflammatory messengers, pain‑related peptides, and receptors that make cells more sensitive to noxious stimuli, changes that mirror what is seen in painful nerve disorders.
Deep look at proteins and fats inside neurons
Because genes are only blueprints, the team also examined the proteins actually present in the neurons, as well as their lipid (fat) composition. After 48 hours of paclitaxel exposure, many of the same stress and inflammatory signals detected at the RNA level also appeared as increased proteins, confirming that the neurons were executing these harmful programs. Strikingly, proteins needed for axonal transport—the molecular motors and scaffolds that move cargo along the long nerve fibers—were sharply reduced. This included kinesins and regulatory proteins that keep microtubule tracks stable. At the lipid level, the neurons showed a drop in key building blocks for cholesterol and membrane lipids, alongside a rise in stored fats called triacylglycerols. Together, these shifts suggest that paclitaxel not only triggers cell‑death pathways but also weakens the physical structure and energy balance of the nerve fibers.
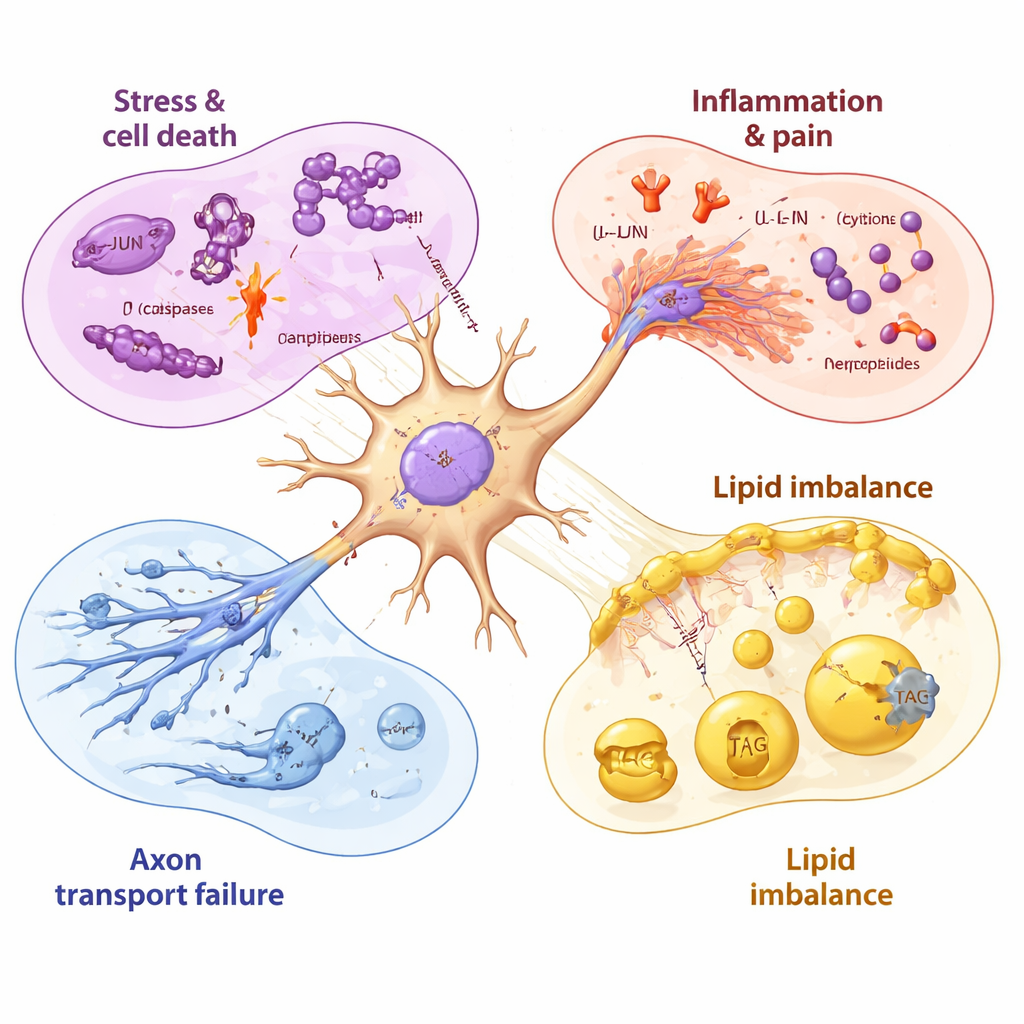
A timeline of nerve injury and incomplete recovery
By sampling the neurons before, during, and after drug exposure, the researchers could order these events in time. Mitochondrial (energy‑related) genes were disrupted within hours, followed by the stress gene JUN, then by stronger activation of cell‑death and inflammatory programs as viability began to fall. Even after paclitaxel was removed, many harmful signals stayed elevated for days, while the genes needed to make healthy membrane lipids continued to decline. Some late‑rising molecules, such as a drug‑efflux pump that can expel paclitaxel and certain growth factors, hinted that neurons were trying to protect and repair themselves—but these responses appeared relatively slow and incomplete compared with the rapid, early push toward damage.
What this means for patients living with nerve pain
For a layperson, the message is that paclitaxel harms sensory nerves through a coordinated series of blows: it stresses their energy systems, flips genetic switches toward cell death, stirs up inflammation and pain signaling, disrupts the internal “transport highways” inside nerve fibers, and alters the fats that keep nerve membranes stable. By mapping these changes in human‑derived neurons with exquisite detail, the study highlights concrete, druggable targets—such as specific stress regulators, inflammatory receptors, and lipid‑making enzymes—that might be blocked or supported to protect nerves without weakening cancer treatment. While new therapies will require further testing, this work offers a clearer roadmap to preventing or easing chemotherapy‑induced nerve damage, improving quality of life for cancer survivors.
Citation: Schinke, C., Maierhof, S.K., Hew, L. et al. Time‑resolved multi-omic analysis of paclitaxel exposure in human iPSC‑derived sensory neurons unveils mechanisms of chemotherapy‑induced peripheral neuropathy. Cell Death Dis 17, 211 (2026). https://doi.org/10.1038/s41419-026-08445-2
Keywords: chemotherapy-induced peripheral neuropathy, paclitaxel, sensory neurons, neuroinflammation, axon degeneration