Clear Sky Science · en
Reversible arginine methylation regulates mitochondrial IDH2 activity: coordinated control by CARM1 and KDM3A/4A
Fine-Tuning the Cell’s Power Plants
Mitochondria, often called the power plants of our cells, do far more than just make energy. They constantly adjust how they burn fuel to meet our body’s needs, and when this goes wrong it can contribute to diseases like diabetes, heart failure, and cancer. This study uncovers a hidden “dimmer switch” inside mitochondria: a tiny chemical tag on a single building block of a key enzyme, IDH2, that helps decide whether mitochondria idle low or rev up their energy output.
A Tiny Tag with Big Consequences
Proteins in our cells are not static; they are frequently modified with small chemical groups that change how they behave. While many such modifications in the cell’s nucleus and fluid interior are well known, those inside mitochondria are less explored. The authors focused on one particular type of tag called arginine methylation and asked whether it might control how mitochondrial enzymes work. They homed in on an enzyme called IDH2, a workhorse of the tricarboxylic acid (TCA) cycle, which helps turn nutrients into both energy and important metabolic building blocks. Using biochemical tools and mass spectrometry, they found that IDH2 carries a specific methyl mark on one amino acid, arginine 188, and that this mark is placed by an enzyme called CARM1 that surprisingly operates inside mitochondria.
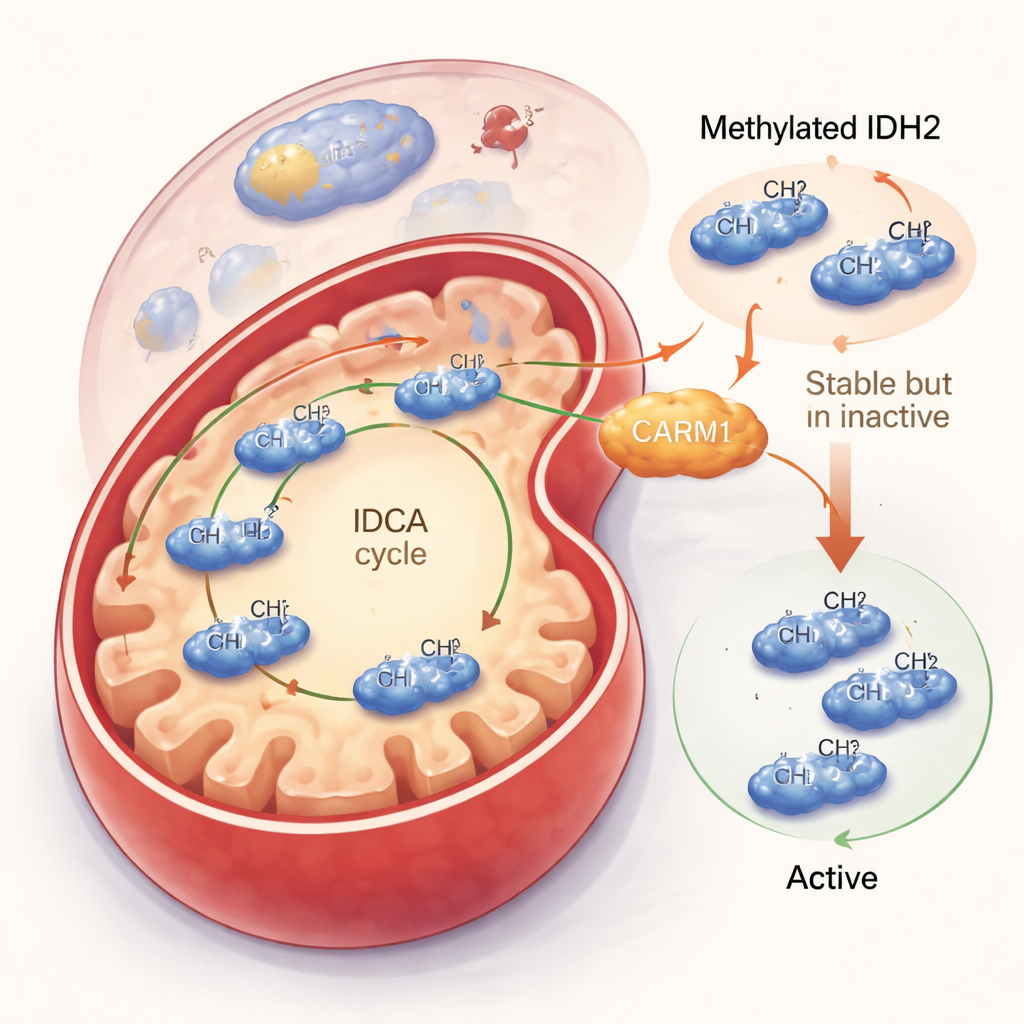
Turning IDH2 Down but Making It Last Longer
The team then asked what this methyl tag actually does. When CARM1 was active and able to methylate IDH2, the IDH2 protein became more stable and stuck around longer in the cell. But this apparent bonus came with a trade-off: methylated IDH2 was less active. In cell experiments and purified protein tests, methylated IDH2 produced less of its main product, alpha-ketoglutarate, and generated less of the protective molecule NADPH. In contrast, a version of IDH2 that could not be methylated at arginine 188 formed more of the active dimer shape, worked faster, and boosted both alpha-ketoglutarate and NADPH levels. Even though there was less of this unmethylated IDH2 overall, each molecule did more work, leading to a net increase in mitochondrial output.
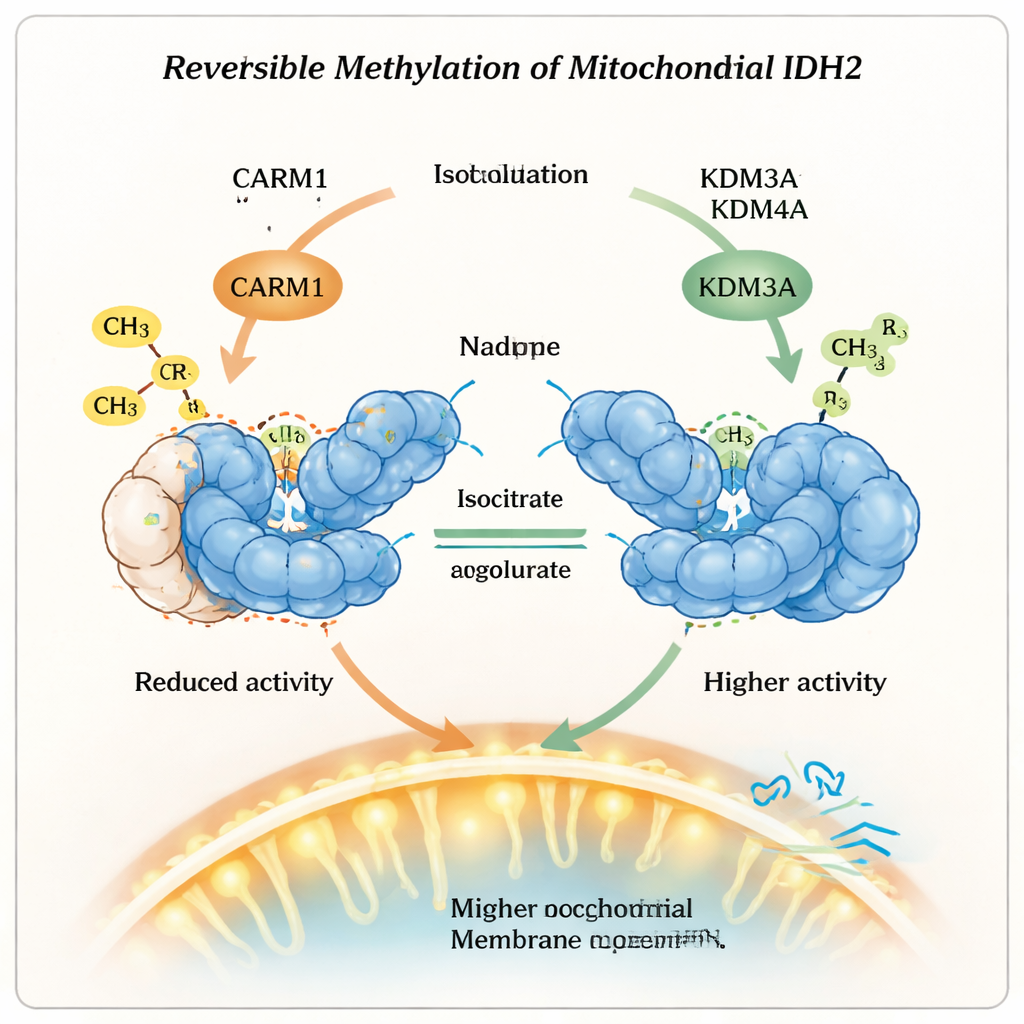
A Reversible Switch with Two Erasers
Crucially, this methyl mark is not permanent. The researchers showed that two enzymes normally known for removing a different kind of tag, lysine demethylases KDM3A and KDM4A, can also erase the arginine methyl mark from IDH2. When these demethylases were present, the methylated arginine at position 188 lost its extra chemical groups, IDH2 became less stable but more active, and its dimer form increased. Cells with more active KDM3A or KDM4A showed higher mitochondrial membrane potential and consumed more oxygen, signs that their mitochondria were running hotter on oxidative metabolism. When these enzymes were reduced, the opposite happened: IDH2 activity dropped and mitochondrial performance declined, underscoring that this is a reversible control system rather than a one-way change.
Linking Metabolism and Disease
Because IDH2 has been tied to cancer and other disorders, the authors examined how this methylation switch might matter in disease-like settings. In various normal and cancer cell lines, they observed that higher IDH2 protein levels often went hand-in-hand with more methylation and surprisingly lower enzyme activity, suggesting that “more protein” does not always mean “more function.” In breast cancer datasets, patients with higher IDH2 protein levels tended to have poorer survival, even though IDH2 gene activity did not track with outcome. The results support a model in which CARM1-driven methylation slows the TCA cycle and nudges cells toward alternative, cancer-favored ways of using nutrients, while demethylases like KDM3A and KDM4A can push cells back toward more efficient energy production.
What This Means for Health and Therapy
In everyday terms, this work shows that mitochondria do not simply turn enzymes on or off, but use reversible chemical tags as fine-tuned dials. By adding a methyl group, CARM1 turns IDH2 into a longer-lived but less active form; by removing that tag, KDM3A and KDM4A sacrifice some stability to gain more power output. This balance influences how cells choose between fast but inefficient sugar burning and slower, more efficient energy production. Understanding this methylation “dimmer switch” on IDH2 opens new possibilities for therapies that aim to correct faulty mitochondrial metabolism in conditions like cancer and metabolic disease, by targeting the enzymes that write and erase these tiny but powerful chemical marks.
Citation: Cho, Y., Winarto, J., Song, DG. et al. Reversible arginine methylation regulates mitochondrial IDH2 activity: coordinated control by CARM1 and KDM3A/4A. Cell Death Dis 17, 195 (2026). https://doi.org/10.1038/s41419-026-08444-3
Keywords: mitochondria, IDH2, arginine methylation, cell metabolism, CARM1