Clear Sky Science · en
A MIF-p38-GSDMD inflammatory loop in keratinocytes underlies UVB-induced cutaneous lupus
Why sunlight can be a serious problem in lupus
For most people, sunshine simply causes a tan or, at worst, a sunburn. But for many patients with lupus, a few minutes of ultraviolet B (UVB) light can trigger painful, long‑lasting rashes and even worsen their whole disease. This study uncovers a hidden “feedback loop” inside skin cells that helps explain why their skin is so sensitive to light—and points to new, very local treatments that might calm the reaction without suppressing the entire immune system.
Skin cells as unexpected troublemakers
Doctors have long known that cutaneous lupus erythematosus (CLE) involves chronic inflammation and scarring of the skin, but attention often focused on invading immune cells. Using single‑cell RNA sequencing, the authors instead zoomed in on the skin’s own building‑block cells—keratinocytes in the outer layer and fibroblasts beneath. They found that in lupus patients, specific subgroups of keratinocytes are expanded and display a strong “interferon signature,” a pattern of antiviral alarm genes switched permanently on. Within these abnormal keratinocytes, one messenger protein stood out: macrophage migration inhibitory factor, or MIF, which was produced at much higher levels than more familiar inflammatory molecules like TNF or IL‑6.
A hidden messenger released by sunlight
Finding high MIF inside keratinocytes did not yet explain why sunlight is so dangerous. When the team exposed cultured keratinocytes to UVB light, they discovered that the total amount of MIF in the cells barely changed—but the protein was released into the surrounding fluid in a dose‑dependent way. The more UVB delivered, the more MIF leaked out, closely tracking a marker of membrane damage. When this MIF‑rich fluid was applied to fresh keratinocytes and fibroblasts, it drove classic CLE‑like changes: more inflammatory cytokines and enzymes that break down and rebuild the skin’s supporting matrix. Silencing MIF with genetic tools or blocking it with a small‑molecule drug sharply reduced these harmful reactions, showing that keratinocyte‑derived MIF is not just a bystander but a key amplifier of UVB‑driven damage. 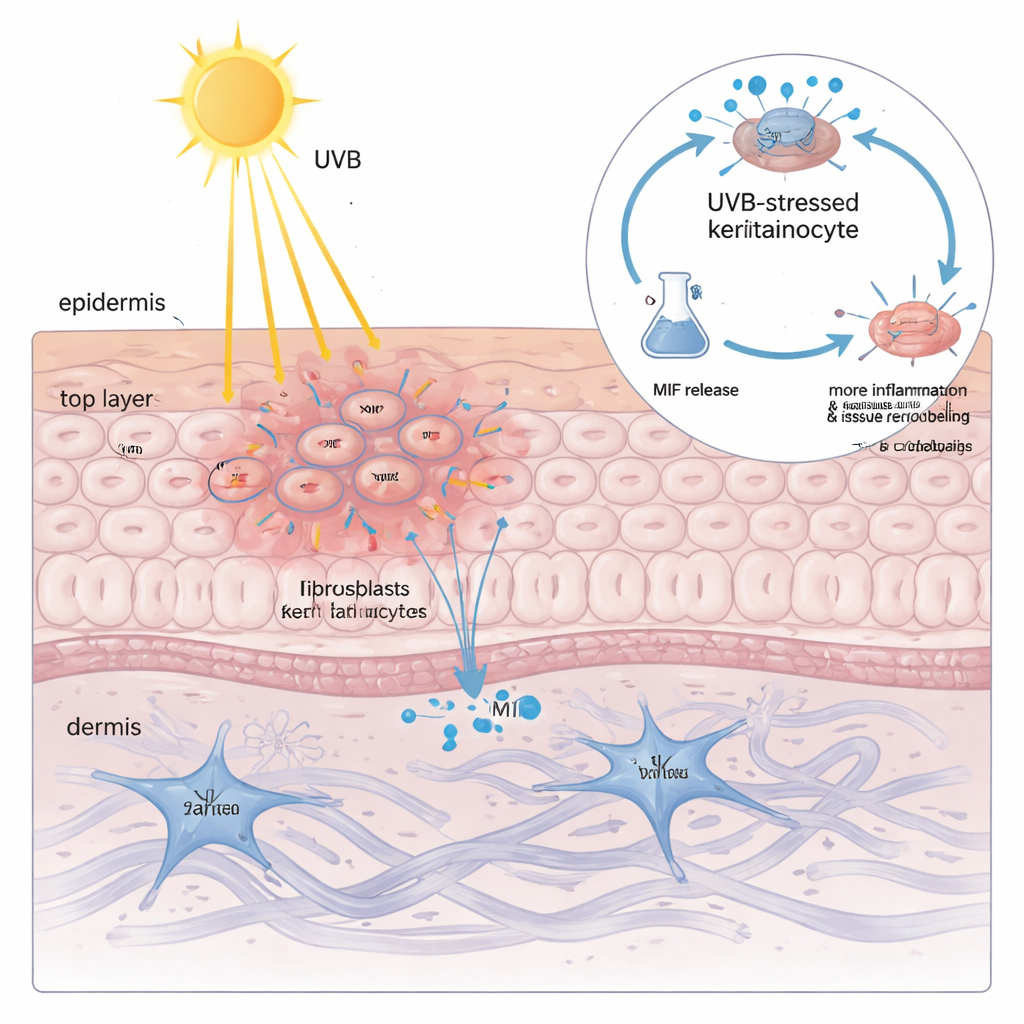
A self‑sustaining inflammatory loop inside keratinocytes
To understand how UVB forces keratinocytes to release MIF, the researchers built a “lupus‑like” keratinocyte model by loading cells with their own DNA and RNA, mimicking the mismanaged genetic debris seen in lupus. In this primed state, UVB activated a so‑called ribotoxic stress response: a damage signal from ribosomes that switched on a kinase called ZAKα and, in turn, the stress enzyme p38. This pathway boosted another protein, the transcription factor C/EBPβ, which bound to the control region of the NLRP3 gene and turned it on. NLRP3 then drove cleavage of GSDMD, a pore‑forming protein that punches holes in the cell membrane and causes an inflammatory form of cell death known as pyroptosis. Crucially, MIF escaped mainly through these GSDMD pores—not via normal secretory vesicles. Once outside, MIF bound to its surface receptor CD74 on keratinocytes, re‑activating p38, NLRP3 and GSDMD and thereby closing a vicious circle: UVB → p38 → NLRP3 → GSDMD pores → MIF release → more p38 activation.
From molecular insight to new local treatments
This loop was not just a lab curiosity. In a lupus‑prone mouse strain, UVB exposure produced severe skin lesions with high levels of MIF, p38 activation, NLRP3 and cleaved GSDMD in the epidermis. When the researchers used a gene‑therapy virus to silence the Mif gene specifically in skin, UVB‑induced rashes became much milder, and markers of inflammation and tissue remodeling dropped in both keratinocytes and fibroblasts. To avoid whole‑body drug side effects, they also engineered dissolvable microneedle patches loaded with the MIF inhibitor ISO‑1. Pressed briefly into the skin, these tiny needles painlessly delivered the drug into the upper layers. Treated lupus‑prone mice developed far fewer and less severe lesions after UVB, and the molecular signature of the MIF‑p38‑GSDMD loop was markedly dampened. 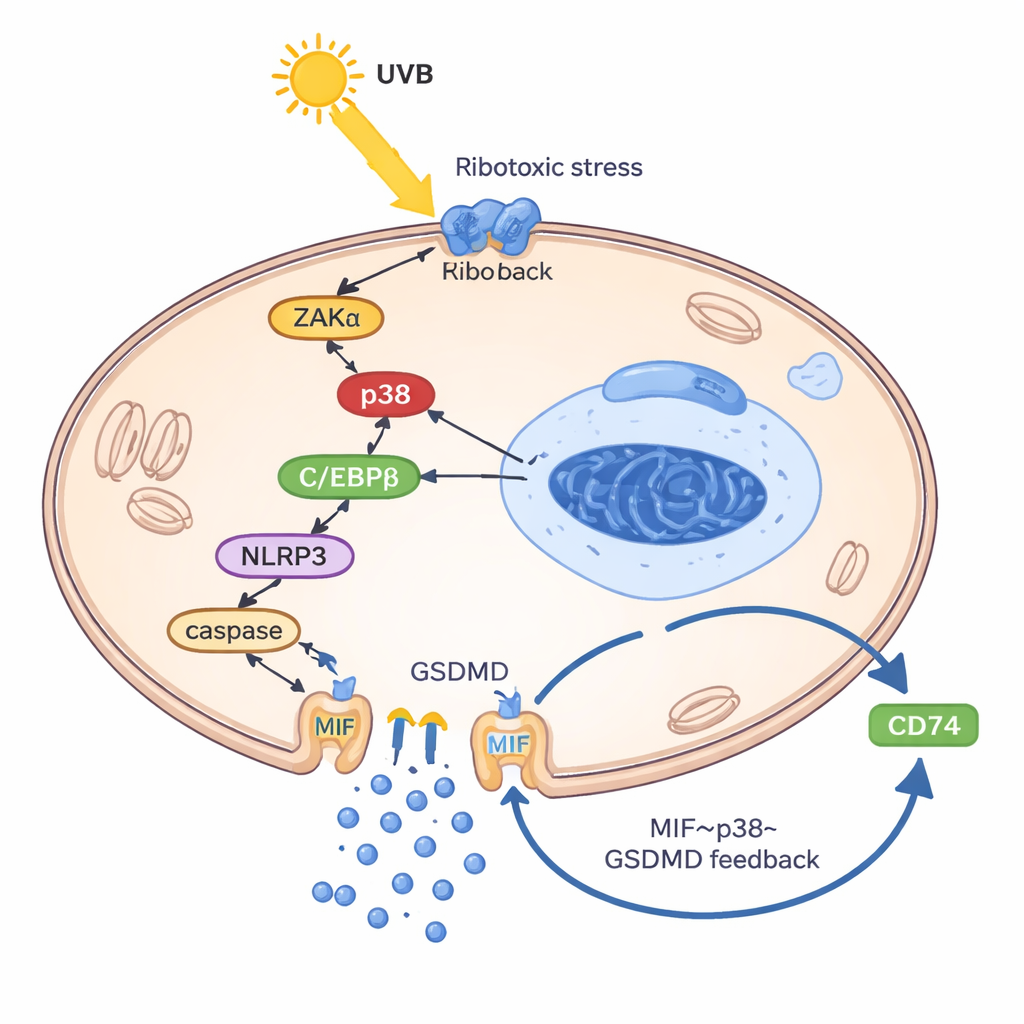
What this means for people with lupus
In plain terms, this work shows that in lupus, certain skin cells are wired to overreact to UVB light by releasing a powerful “booster” molecule, MIF, through self‑inflicted membrane pores. That boost then feeds back to keep the inflammatory machinery running, even after the initial light exposure ends. By mapping this loop step by step, the study suggests that blocking MIF right in the skin—using gene silencing or smart delivery systems like microneedle patches—could break the cycle of photosensitive flares without broadly shutting down the immune system. If similar strategies prove safe and effective in humans, they could offer people with lupus new ways to live more comfortably in the light.
Citation: Guo, C., Luo, S., Luo, J. et al. A MIF-p38-GSDMD inflammatory loop in keratinocytes underlies UVB-induced cutaneous lupus. Cell Death Dis 17, 198 (2026). https://doi.org/10.1038/s41419-026-08443-4
Keywords: cutaneous lupus, photosensitivity, keratinocytes, inflammatory loop, microneedle therapy