Clear Sky Science · en
SOX21 suppresses glioblastoma growth by repressing AP-1 activity
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers, largely because a small population of stubborn “starter” cells can keep the tumor growing and make it come back after treatment. This study uncovers how a naturally occurring protein in brain cells, called SOX21, can shut down these dangerous cells by turning off a growth-driving program inside their DNA. Understanding this internal off‑switch could inspire new strategies to slow glioblastoma and help current treatments work better.
The hidden drivers inside brain tumors
Within a glioblastoma, not all cells are equal. A minority known as glioblastoma precursor cells (GPCs) behave like stem cells: they self‑renew, resist therapy and can re‑seed the tumor after surgery, radiation or chemotherapy. These cells are controlled by networks of transcription factors, proteins that decide which genes are active or silent. Some well‑known factors, such as SOX2 and members of the AP‑1 family, typically boost growth and survival. In contrast, SOX21 has been linked to reduced cell division and increased maturation of normal brain stem cells, and earlier work hinted that it might protect against brain tumors. However, it was unclear whether raising SOX21 levels in an already established tumor could actually slow its growth, and how exactly SOX21 acted inside cancer cells.
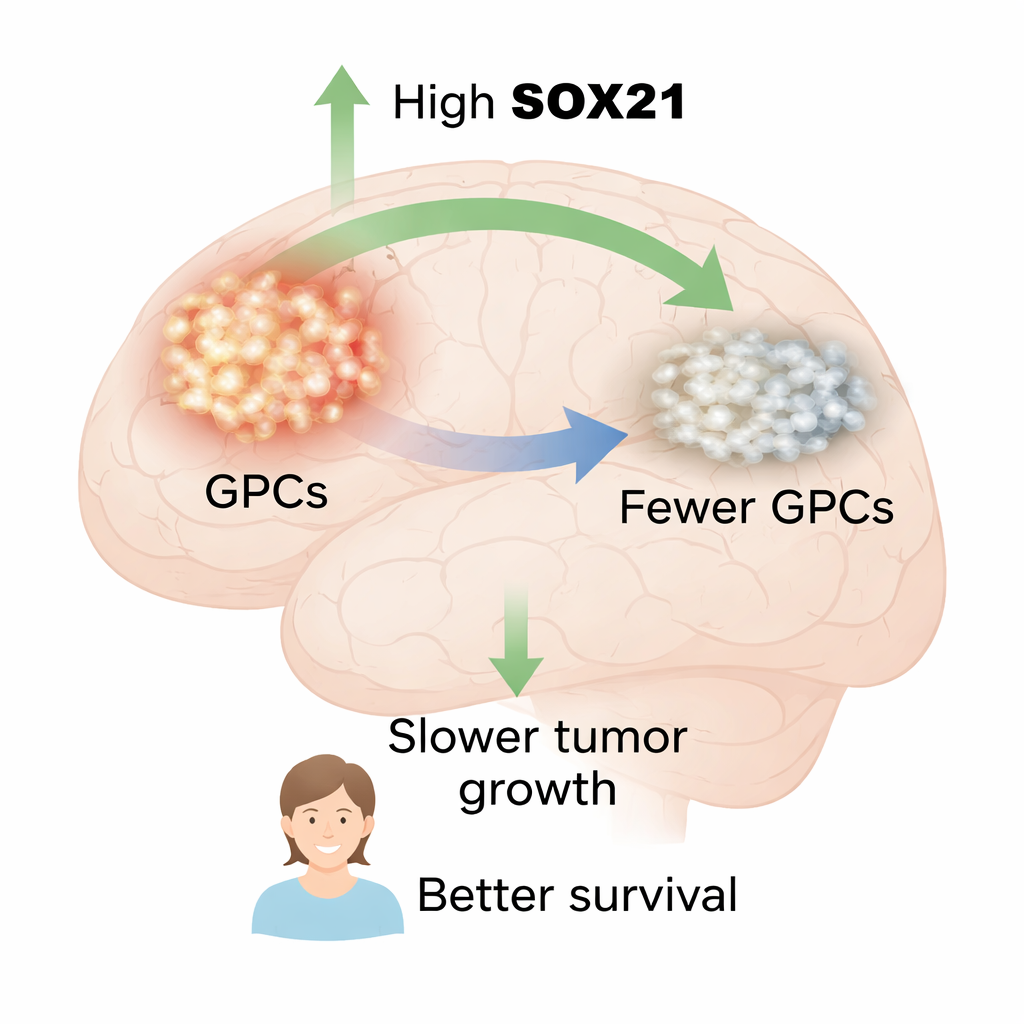
SOX21 marks a better prognosis and weakens tumor cells
The researchers began by examining human glioblastoma samples. They found that SOX21 protein was mainly present in cells that also carried stem‑like markers such as SOX2 and OLIG2, and in cells actively dividing, identified by the marker KI67. This showed that SOX21 lives precisely in the tumor’s GPC compartment. Looking at large public datasets of patient tumors, they then compared survival in people whose cancers had high versus low SOX21 gene activity. In two independent cohorts, patients with higher SOX21 expression lived significantly longer, linking SOX21 to a more favorable disease course.
Turning SOX21 on slows growth and shrinks tumor‑forming potential
To test cause and effect, the team engineered GPCs taken from patient tumors to carry a switchable SOX21 gene that could be turned on with the drug doxycycline. When SOX21 was induced in lab dishes, these cells divided less, incorporated less DNA building block (a sign of slower proliferation), and showed more hallmarks of programmed cell death. Their ability to form free‑floating spheres, a measure of stem‑like self‑renewal, dropped sharply, and they became more sensitive to the standard glioblastoma drug temozolomide. In mouse experiments, human GPCs were first allowed to establish brain tumors, and only then was SOX21 turned on. Under these conditions, SOX21—but not SOX2—slowed tumor growth, reduced the fraction of dividing and stem‑like cells inside the mass, and significantly prolonged the animals’ survival. This demonstrates that boosting SOX21 can act on existing tumors, not just prevent their formation.
How SOX21 disarms a cancer‑promoting program
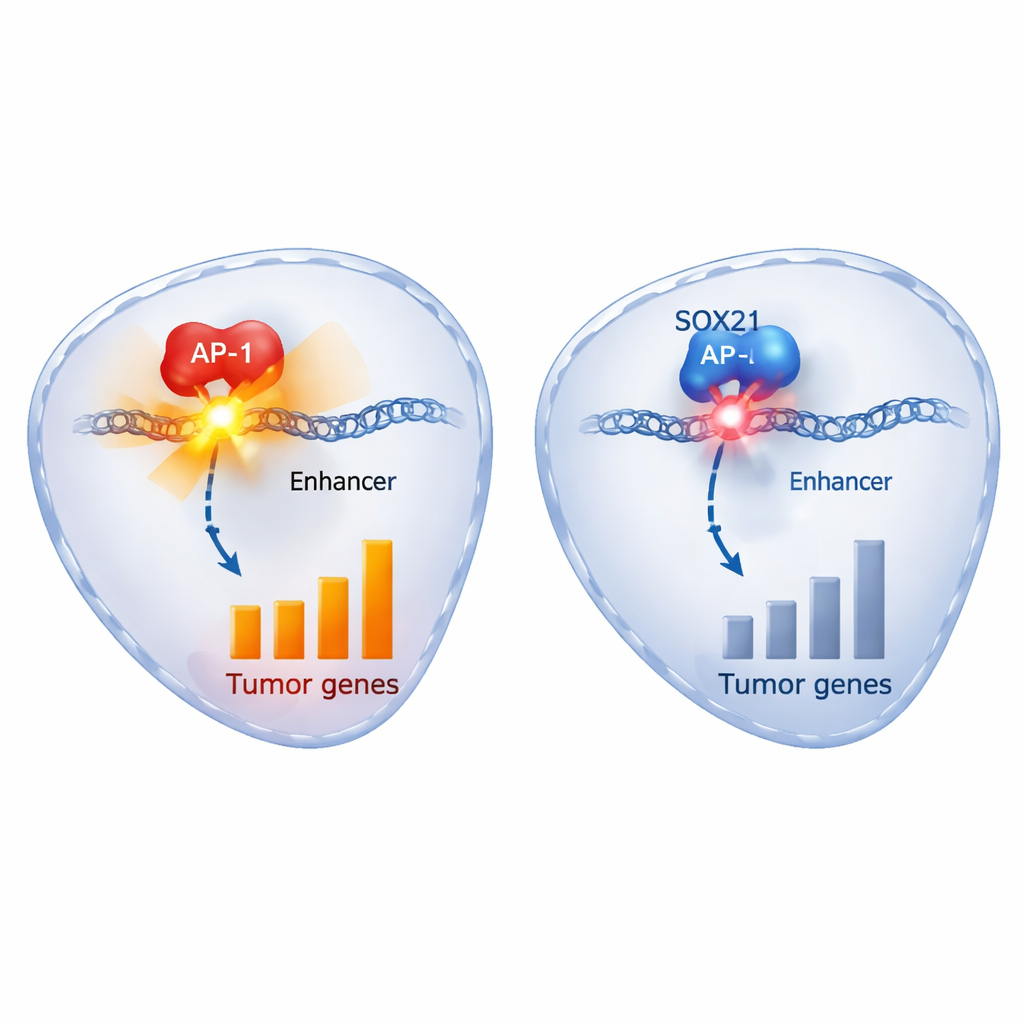
Diving into the molecular machinery, the authors mapped which genes changed when SOX21 was switched on and where along the DNA SOX21 physically bound. SOX21 increased genes involved in cell‑cycle arrest and cell death, including the tumor‑suppressor p21, and reduced genes previously linked to glioma progression, such as CDK6, EFNB2, HDAC9 and SOX2 itself. Strikingly, SOX21 bound strongly to DNA regions that were also occupied by c‑JUN, a key member of the AP‑1 family that usually activates growth‑promoting genes. These shared regions carried classic AP‑1 sequence motifs and chemical marks of active enhancers—stretches of DNA that boost gene expression. When SOX21 levels rose, these enhancers became less accessible and lost activation marks, and the associated genes were turned down. Drugs that block AP‑1 reproduced many of the same gene changes and biological effects as SOX21, while forcing cells to overproduce c‑JUN could rescue them from SOX21’s suppressive influence.
What this means for future glioblastoma treatment
Put simply, SOX21 acts as a brake inside glioblastoma’s most dangerous cells by latching onto the same DNA switches used by AP‑1 to drive tumor growth, and then dimming those switches. Patients whose tumors naturally make more SOX21 tend to fare better, and in animal models, turning SOX21 on in established tumors slows them down and helps them respond to chemotherapy. Although directly delivering or activating SOX21 in patients is not yet practical, the pathways it controls—and its tug‑of‑war with AP‑1—offer promising entry points for new drugs designed to weaken the tumor’s stem‑like core and reduce the chance of relapse.
Citation: Rrapaj, E., Yuan, J., Kurtsdotter, I. et al. SOX21 suppresses glioblastoma growth by repressing AP-1 activity. Cell Death Dis 17, 191 (2026). https://doi.org/10.1038/s41419-026-08442-5
Keywords: glioblastoma, cancer stem cells, SOX21, AP-1, brain tumor therapy