Clear Sky Science · en
Honokiol blocks tumor development and metastasis through mitochondrion-targeted effects
Turning a Tree Compound into a Cancer Fighter
Many cancer drugs come from plants, and scientists are still uncovering how some of these natural molecules work inside our cells. This study focuses on honokiol, a substance found in Magnolia bark long used in traditional medicine, and shows how it can directly target tiny power plants inside cancer cells to slow tumor growth and block spread, while largely sparing normal cells.
The Power Stations Inside Our Cells
Every cell relies on mitochondria, often called the cell’s “power stations,” to generate the energy molecule ATP. In cancer, mitochondria do more than make fuel—they help decide whether a cell survives or dies. One large molecular machine, ATP synthase, sits in the inner membrane of mitochondria and makes ATP. Cancer cells often overproduce a partner protein called IF1 that latches onto ATP synthase and acts like a safety lock, helping tumors resist a form of self‑destruct known as mitochondrial permeability transition and subsequent cell death.
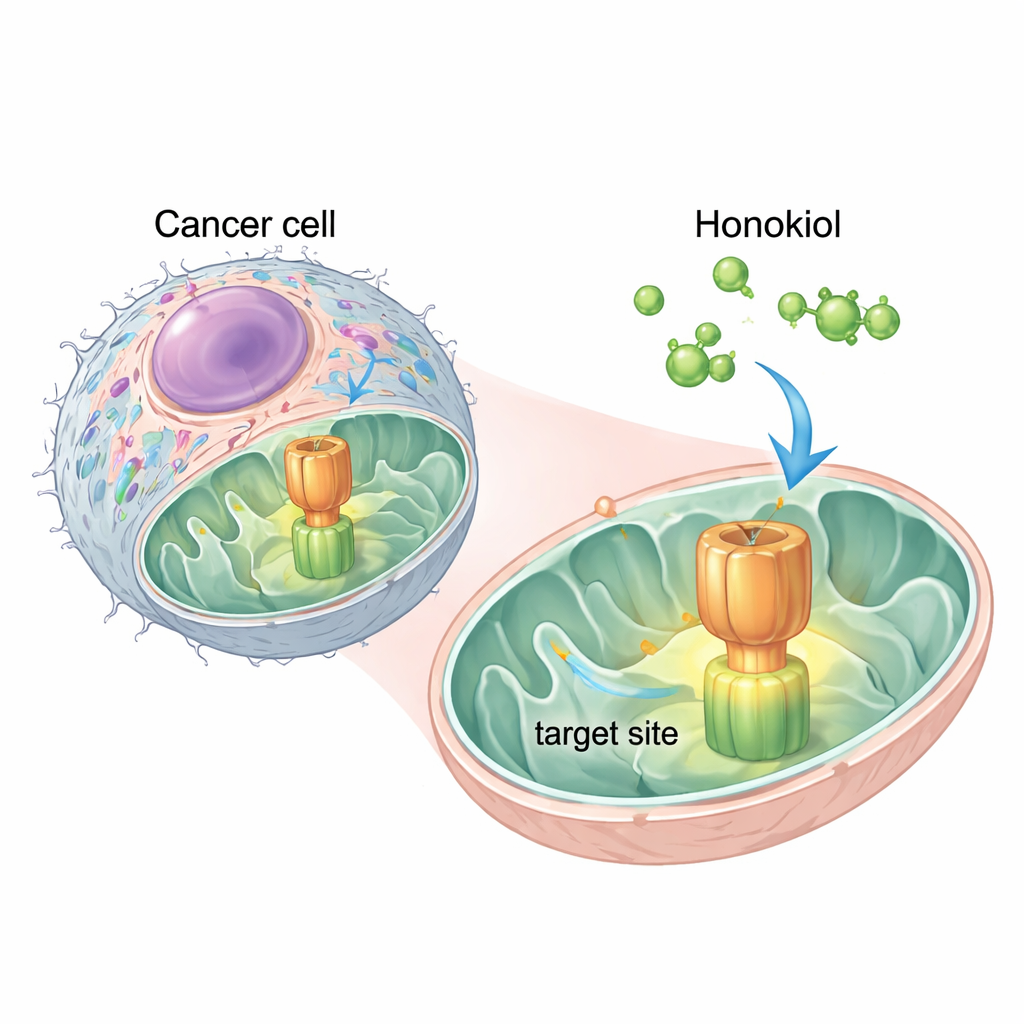
A Natural Molecule Finds a Critical Switch
The researchers used computer simulations to see where honokiol might land on ATP synthase. They found two promising “docking” sites on one of its components, a region called OSCP. The strongest site overlapped with the same patch that IF1 uses to hold onto ATP synthase. This suggested that honokiol could act like a molecular wedge, slipping into OSCP and pushing IF1 away. Experiments with human cervical cancer HeLa cells confirmed this idea: adding honokiol reduced the physical contact between IF1 and OSCP, without disrupting other parts of the enzyme, showing that the effect was specific.
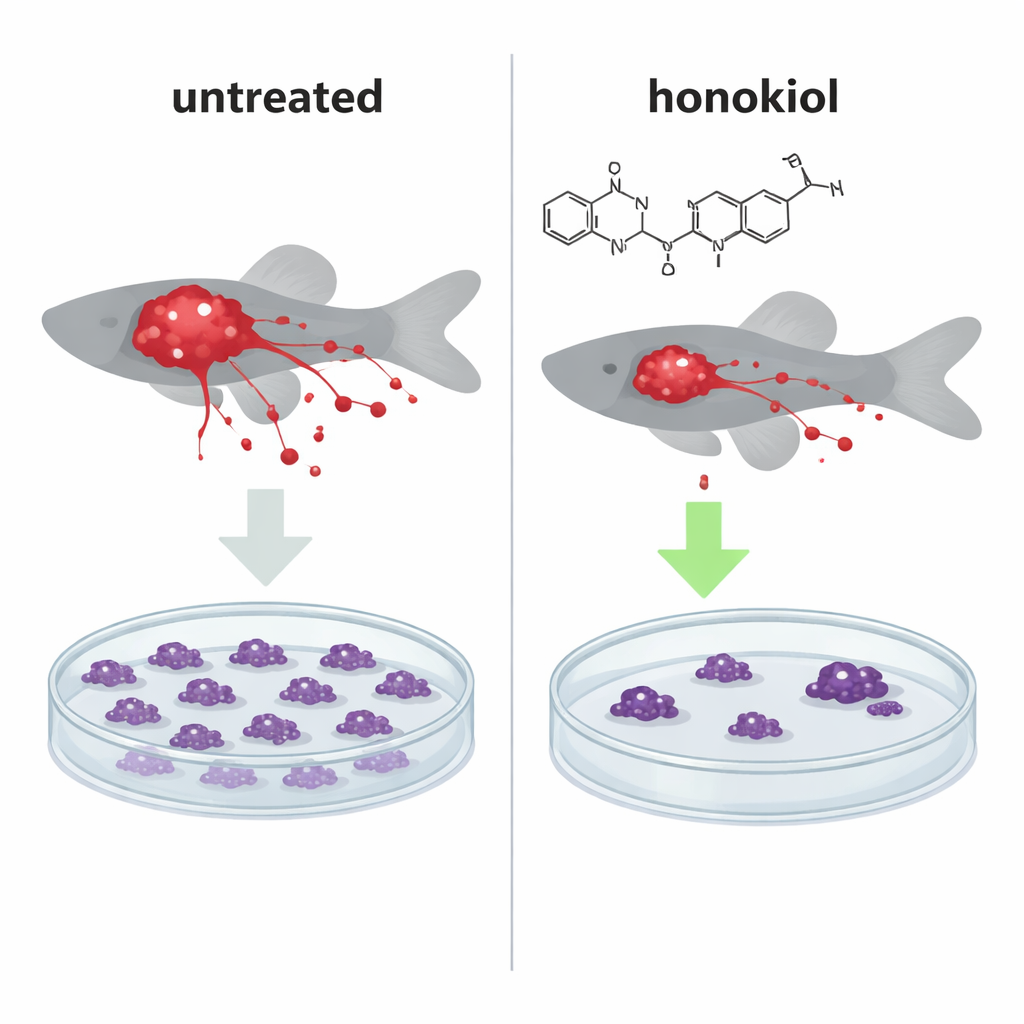
From Cells to Living Animals: Less Tumor, Less Spread
To see if this molecular push had real‑world impact, the team implanted glowing human cancer cells into tiny zebrafish embryos, an established whole‑animal model for tracking tumor growth and metastasis. In fish injected with normal, IF1‑rich HeLa cells, honokiol treatment significantly shrank tumor masses and cut the number of cancer cells that spread to distant areas, making the pattern resemble fish injected with IF1‑knockout cells that lack the protective protein. Interestingly, honokiol also reduced the number of metastatic cells even when IF1 was absent, suggesting a second, IF1‑independent way of limiting cancer spread.
Forcing Faulty Power Plants to Trigger Self‑Destruct
Back in the lab dish, honokiol reduced the ability of cancer cells to form large colonies in soft agar, a test that mimics the stressful, low‑oxygen conditions inside solid tumors. This happened without a major slowdown in ordinary cell division or in basic mitochondrial respiration, pointing instead to a change in how mitochondria decide between life and death. Measurements of calcium handling showed that honokiol made mitochondria open the permeability transition pore at lower calcium levels, a known trigger for swelling and release of death‑promoting factors. At modest doses this effect depended on IF1 displacement; at higher doses honokiol acted through its second OSCP binding site and through increased reactive oxygen species, sensitizing even IF1‑deficient cells to programmed cell death.
Stopping Cancer Cells from Going on the Move
Cancer becomes most dangerous when cells break away and migrate to new organs. In wound‑like “scratch” assays, honokiol strongly slowed the collective migration of cancer cell sheets and reduced markers of epithelial‑mesenchymal transition, a process that equips tumor cells to move and invade. Detailed electron microscope images revealed that migrating cells normally enlarge and reshape their mitochondria, adding internal folds to meet high energy demands at the leading edge. With honokiol, these front‑line mitochondria instead became swollen and structurally damaged—hallmarks of the permeability transition pore opening—while mitochondria in non‑moving regions were less affected or even increased in number, depending on IF1.
What This Means for Future Cancer Treatments
Put simply, this work shows that honokiol can home in on a small regulatory region of the mitochondrial ATP‑making machine and flip a built‑in safety switch that cancer cells often keep locked. By dislodging IF1 and directly nudging the pore toward opening, honokiol encourages tumor cells to self‑destruct and makes it harder for them to migrate and seed metastases. Because these effects arise from features that many cancers share—rather than from a single surface marker—targeting the OSCP region of ATP synthase could inspire a new class of therapies that exploit the vulnerabilities of cancer cell power stations while limiting harm to healthy tissues.
Citation: Grandi, M., Boldrin, F., Risato, G. et al. Honokiol blocks tumor development and metastasis through mitochondrion-targeted effects. Cell Death Dis 17, 186 (2026). https://doi.org/10.1038/s41419-026-08441-6
Keywords: honokiol, mitochondria, ATP synthase, apoptosis, metastasis