Clear Sky Science · en
Immune checkpoint inhibitor-related pneumonitis: current advances and the putative role of mesenchymal stem cell therapy
When Cancer Treatment Backfires on the Lungs
Drugs that unleash the immune system against cancer have transformed care for diseases like lung cancer and melanoma. Yet this new power comes with a catch: in some patients, the same immune attack that targets tumors also turns on the lungs, causing a condition called immune checkpoint inhibitor-related pneumonitis (ICIP). This review explains what ICIP is, why it happens, how doctors currently treat it, and why a special type of stem cell—mesenchymal stem cells (MSCs)—might one day offer a safer, more precise way to calm the storm without sacrificing cancer control. 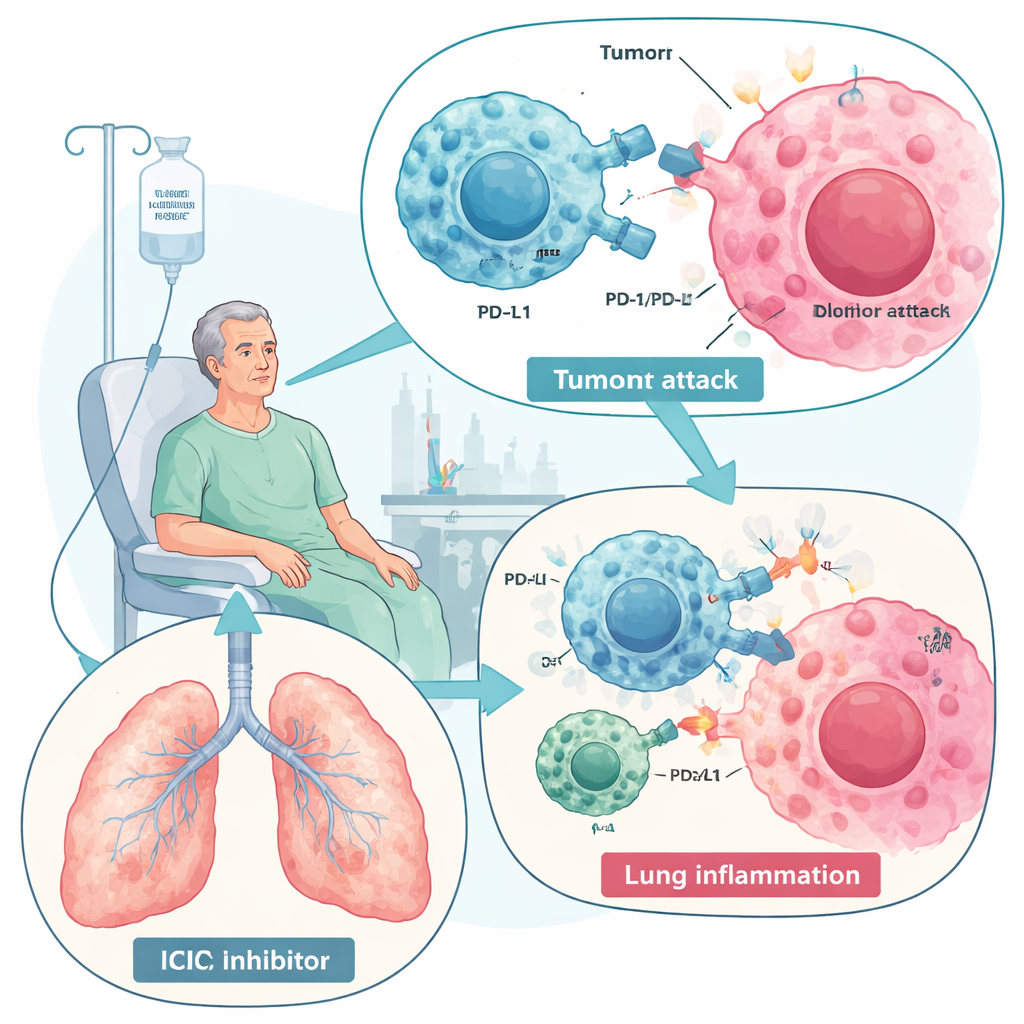
Powerful Cancer Drugs with a Hidden Lung Risk
Immune checkpoint inhibitors, especially those blocking PD-1 and PD-L1, work by removing molecular “brakes” that normally keep immune responses in check. With the brakes off, T cells can better recognize and destroy cancer cells, improving survival in many tumor types. But this same boost can break normal tolerance and trigger immune-related side effects in multiple organs. ICIP is the most serious lung complication of these drugs and one of the leading immune-related causes of treatment-related death. It affects anywhere from a few percent to nearly a third of patients in some real-world studies, particularly those with lung cancer or pre‑existing lung disease such as COPD or interstitial lung disease. Symptoms range from mild cough and breathlessness to life‑threatening respiratory failure.
How the Immune System Damages the Lung
The review describes ICIP as the result of a disturbed immune balance inside the lungs. Overactive “fighter” cells—CD8 T cells, certain helper T cells, inflammatory macrophages, neutrophils, and natural killer cells—flood lung tissue and release aggressive signaling proteins, including interferon‑gamma, TNF‑alpha, and interleukins like IL‑6 and IL‑17. At the same time, normally protective cells that dampen inflammation, such as regulatory T cells and repair‑oriented macrophages, are reduced. Some patients also show rising levels of self‑reactive antibodies before or during treatment, suggesting that ICIP may partly resemble an autoimmune disease. In fluid washed from the lungs, doctors find a T‑cell–rich inflammatory pattern and a cytokine mix distinct from infection or tumor progression, which may help with diagnosis.
Seeing, Grading, and Treating the Damage
On CT scans, ICIP most often shows up as hazy “ground‑glass” areas or patchy consolidations scattered through both lungs, patterns that differ from radiation damage, which stays confined to the irradiated field. Doctors diagnose ICIP by combining scan findings, symptoms, and a careful exclusion of infection and other causes. International guidelines then grade severity from 1 (mild, often radiologic only) to 4 (life‑threatening respiratory failure). Today’s mainstay of treatment is glucocorticoids—strong anti‑inflammatory steroids—sometimes combined with other immune‑dampening drugs like tocilizumab (which blocks IL‑6) or infliximab (which blocks TNF‑alpha) in severe or steroid‑resistant cases. These approaches can be life‑saving, but they are blunt tools: steroids carry many side effects, some patients do not respond, and broad immune suppression may weaken the very anti‑tumor response these drugs were meant to boost.
Why Stem Cells Are Entering the Conversation
Mesenchymal stem cells, which can be harvested from bone marrow, fat, or umbilical cord tissue, have emerged as promising “immune modulators” rather than simple tissue builders. When infused into the bloodstream, many of these cells become temporarily trapped in the lung’s fine blood vessels—a so‑called first‑pass effect—which is a disadvantage for treating distant organs but a potential advantage for lung disease. In animal models and early human trials of conditions like severe pneumonia, acute respiratory distress syndrome, inflammatory bowel disease, and graft‑versus‑host disease, MSCs tone down overactive immune responses, shift immune cells toward a more balanced state, and secrete growth factors that encourage tissue repair while limiting scarring. Importantly, their benefits often arise not from turning into lung cells themselves but from the mix of signaling molecules and tiny vesicles they release. 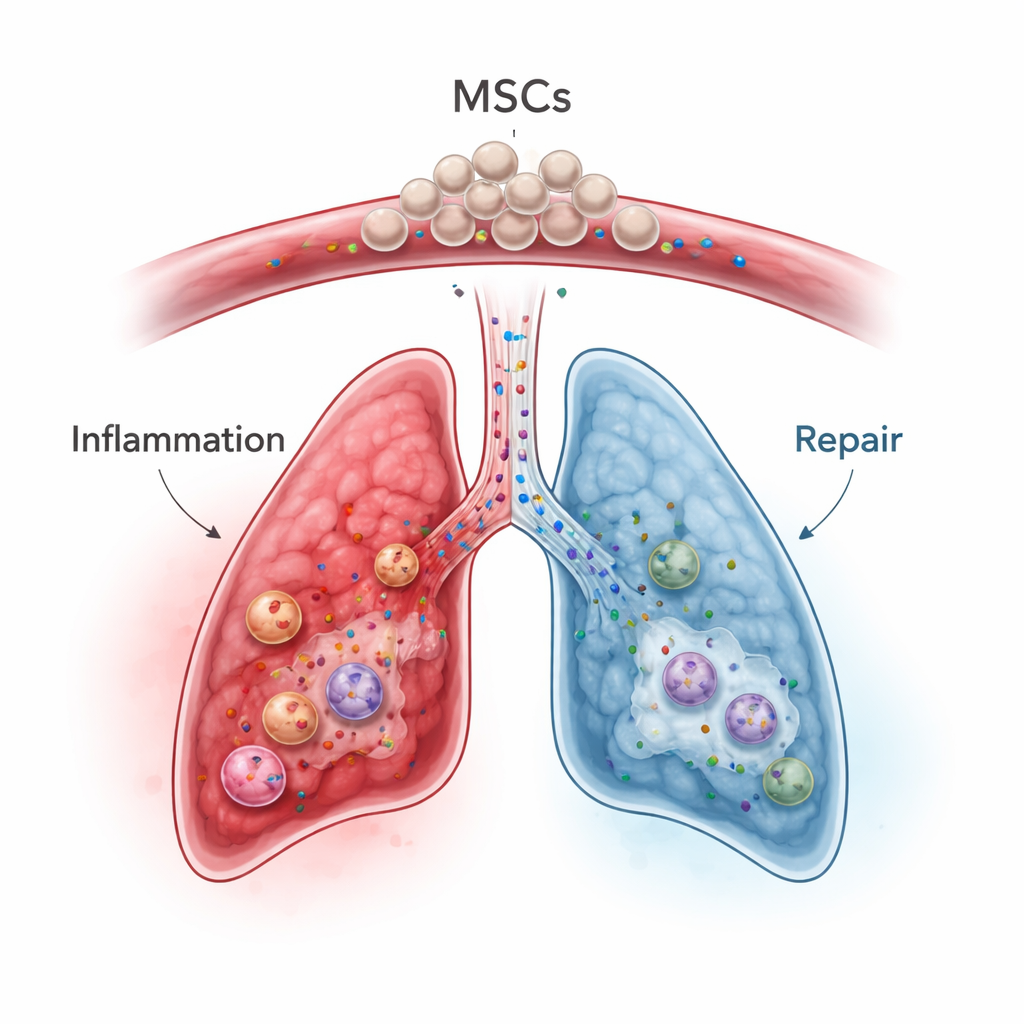
How MSCs Might Calm ICIP Without Killing the Cure
In the context of ICIP, MSCs could, in theory, tackle several problems at once. They can directly restrain activated T cells, promote the growth and function of regulatory T cells, and nudge macrophages away from an aggressive, tissue‑damaging mode toward a healing one. They also lower key inflammatory messengers such as IL‑6, IL‑1β, and TNF‑alpha, and boost anti‑inflammatory factors and protective proteins like TSG‑6 that help limit lung injury and fibrosis. Preclinical work suggests that MSCs or engineered MSC‑derived exosomes can reduce cytokine storms and organ damage induced by checkpoint inhibitors or radiation, while in some settings preserving anti‑tumor activity better than standard immunosuppressants. Still, the authors caution that MSCs may also interact with tumors and blood clotting in complex ways, and that their effects depend heavily on the surrounding inflammatory environment and even on the MSC source (bone marrow, adipose, or umbilical cord).
Looking Ahead: Promise with Careful Testing
For patients whose cancer treatment is halted or threatened by severe lung toxicity, MSC‑based therapies could eventually provide a more targeted way to cool inflammation, repair lung tissue, and prevent long‑term scarring—ideally without undoing the life‑saving benefits of immune checkpoint inhibition. However, no ICIP‑specific MSC trials have yet been completed. The authors argue that carefully designed animal studies and clinical trials are urgently needed to define which type of MSC or exosome product works best, how much to give, when to give it relative to steroids and immunotherapy, and how to monitor for risks like tumor growth or clotting. Until then, MSCs remain a hopeful but still experimental option on the horizon for managing this serious side effect of modern cancer care.
Citation: Li, Z., Zheng, X., Xia, H. et al. Immune checkpoint inhibitor-related pneumonitis: current advances and the putative role of mesenchymal stem cell therapy. Cell Death Dis 17, 200 (2026). https://doi.org/10.1038/s41419-026-08440-7
Keywords: immunotherapy side effects, pneumonitis, checkpoint inhibitors, mesenchymal stem cells, lung inflammation