Clear Sky Science · en
KDM5B cooperates with CRL4B complex to promote the tumorigenesis of ER+ breast cancer via regulating cholesterol metabolism
Why this research matters to everyday health
Breast cancer is the most common cancer in women, and many tumors grow in response to the hormone estrogen. These cancers do more than divide quickly—they also rewire how they use fats like cholesterol. This study uncovers how a gene-regulating protein called KDM5B teams up with another protein complex, CRL4B, to boost cholesterol inside estrogen‑receptor–positive (ER+) breast cancer cells. By understanding this hidden partnership, scientists hope to find new ways to slow tumor growth and improve treatments that already exist, such as hormone therapy and cholesterol‑lowering drugs.
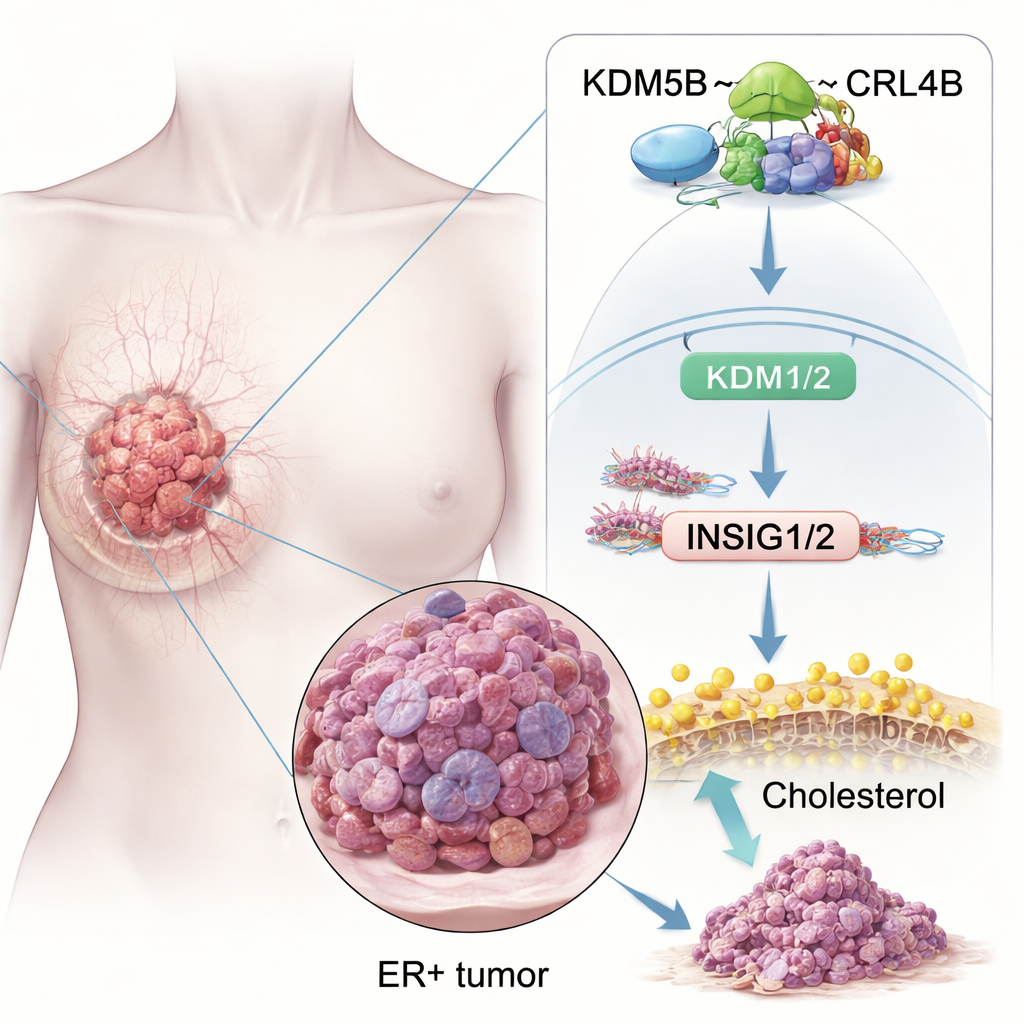
A protein that tips the balance toward cancer
The researchers first asked whether KDM5B is simply present in ER+ breast cancers or actually helps drive them. By analyzing large cancer databases and tissue samples, they found that KDM5B levels are higher in breast tumors than in normal breast tissue, and especially high in ER+ tumors. Patients whose cancers produce more KDM5B tend to have worse survival, even when they receive standard chemotherapy or hormone therapy. In cell culture experiments, turning KDM5B up made ER+ breast cancer cells grow faster, invade surrounding tissue more easily, and form more stem‑like clusters, which are thought to seed new tumors. Turning KDM5B down had the opposite effect, shrinking colonies in dishes and tumors in mice.
A powerful partnership inside cancer cells
To see how KDM5B exerts these effects, the team looked for its protein partners. They discovered that KDM5B physically binds to parts of a molecular machine called the CRL4B complex, an enzyme system that tags proteins and also changes how DNA is packaged. Detailed biochemical tests showed KDM5B directly interacts with two CRL4B components, CUL4B and DDB1, through specific regions of each protein. In ER+ breast cancer cells, this KDM5B–CRL4B complex acts on many genes at their on/off switches. Using genome‑wide mapping, the authors found that KDM5B and CUL4B often sit together at gene promoters, where they alter chemical tags on histone proteins—the spools around which DNA is wrapped—to turn genes down.
Releasing the brakes on cholesterol
Among many affected pathways, cholesterol metabolism stood out. Cancer cells need extra cholesterol to build membranes and survive stress, and ER+ tumors also use cholesterol‑derived molecules to mimic estrogen. The study shows that the KDM5B–CRL4B complex binds directly to the control regions of two key “brake” genes, INSIG1 and INSIG2. These genes normally help keep a master cholesterol regulator, SREBP2, in check. KDM5B–CRL4B adds a repressive tag (H2AK119ub1) and removes an activating tag (H3K4me3) on histones at the INSIG1/2 promoters, dialing these genes down. With fewer INSIG1/2 proteins, SREBP2 is freed to switch on cholesterol‑making genes, raising cholesterol levels inside ER+ breast cancer cells and boosting their invasive behavior. When the researchers disrupted KDM5B or CRL4B, INSIG1/2 rose, SREBP2 activity fell, and total cholesterol in cells dropped.
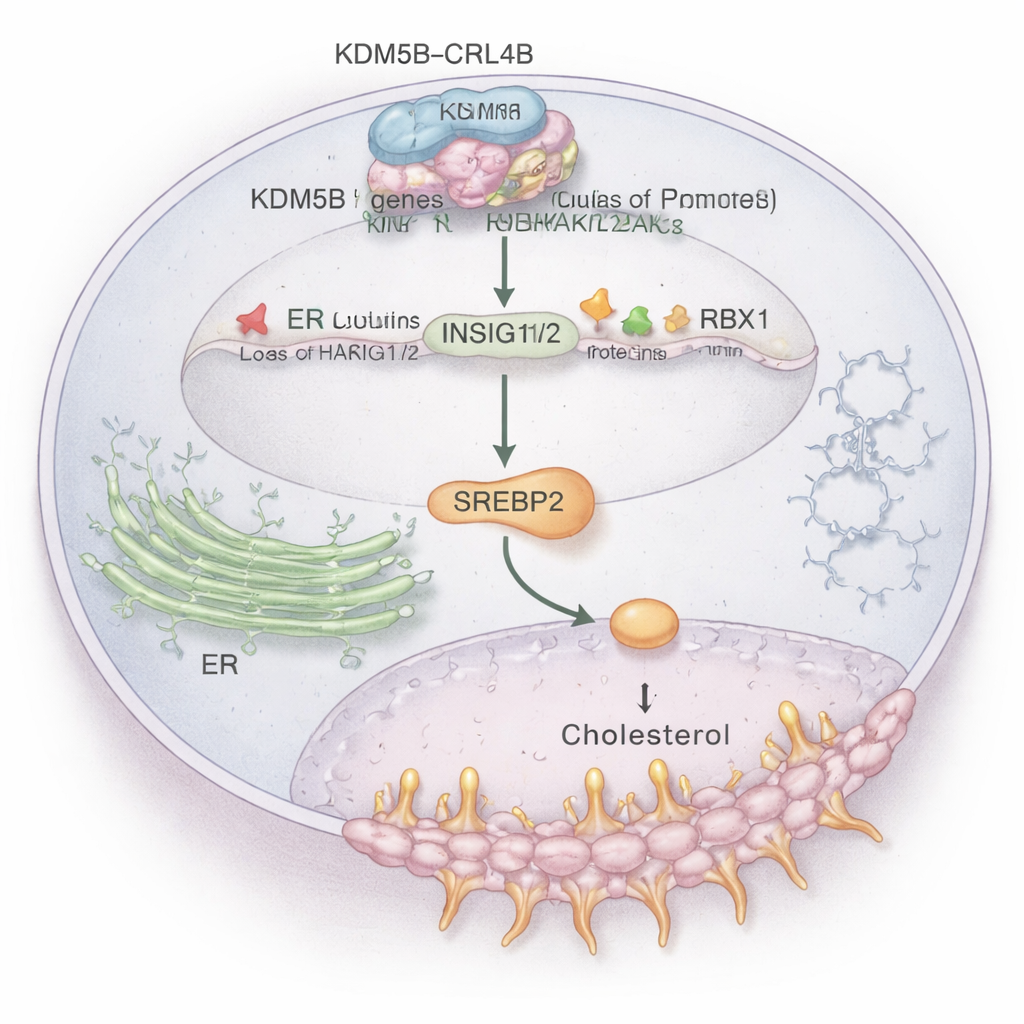
Cholesterol drugs and cancer signals intersect
The work also ties this pathway to a cholesterol‑derived signal called 27‑hydroxycholesterol (27‑HC), a molecule already known to promote ER+ breast cancer. Treating ER+ cells with 27‑HC increased KDM5B levels and further suppressed INSIG1/2, pushing cells toward more growth and invasion. Importantly, blocking KDM5B or CRL4B blunted these aggressive effects, suggesting that 27‑HC partly acts by feeding into the KDM5B–CRL4B axis. Separately, the researchers showed that simvastatin, a widely used cholesterol‑lowering statin, slowed breast cancer cell growth, and when combined with a KDM5B inhibitor, the anti‑tumor effect was stronger. This hints that pairing drugs that target cholesterol production with drugs that target KDM5B’s gene‑controlling activity could be a promising therapeutic strategy.
What this means for patients and future treatments
This study reveals a new chain of events inside ER+ breast cancer cells: a cholesterol‑related signal (27‑HC) boosts KDM5B; KDM5B teams up with the CRL4B complex to shut down INSIG1 and INSIG2; this unleashes SREBP2, ramps up cholesterol production, and helps tumors grow, invade, and maintain stem‑like cells. Because KDM5B is also elevated and linked to poor survival in several other cancers, blocking this protein—or restoring the INSIG1/2 brakes—could offer new ways to control tumor growth. While more work is needed before this can translate into routine care, the findings highlight how closely cancer behavior is tied to everyday molecules like cholesterol, and how existing drugs such as statins might one day be combined with epigenetic therapies to improve outcomes.
Citation: Yang, Y., Gao, T., Yuan, B. et al. KDM5B cooperates with CRL4B complex to promote the tumorigenesis of ER+ breast cancer via regulating cholesterol metabolism. Cell Death Dis 17, 207 (2026). https://doi.org/10.1038/s41419-026-08438-1
Keywords: ER-positive breast cancer, cholesterol metabolism, KDM5B, INSIG1/INSIG2, epigenetic therapy