Clear Sky Science · en
TGFβ signaling promotes cell cycle progression and resistance to the CDK4/6 inhibitor palbociclib through SOX4 transcriptional modulation in breast cancer cells
When Cancer Cells Hijack a Helpful Signal
Our bodies rely on countless chemical signals to keep cells dividing when needed and stopping when they should. One of these signals, called transforming growth factor beta (TGFβ), normally helps prevent tumors. This study shows how, in certain breast cancers, the same TGFβ signal can be twisted to help tumor cells grow and resist an important drug, palbociclib. Understanding this switch could guide better treatment combinations for patients whose tumors stop responding to therapy.
From Growth Brake to Double-Edged Signal
In healthy tissue, TGFβ usually acts as a brake: it slows down cell division and limits inflammation, helping to prevent early tumor formation. But in established cancers, especially in breast tumors that have already acquired cancer-driving mutations, TGFβ can do the opposite—encouraging cells to migrate, invade, and spread. To explore this dual behavior, the researchers used 3D “mini-tumors” grown from human breast cells. They compared normal-like cells to otherwise identical cells carrying an oncogenic HRAS mutation, which pushes them toward cancer. This controlled setup let them ask how the same TGFβ signal plays out in a pre-cancer versus a cancer-like environment. 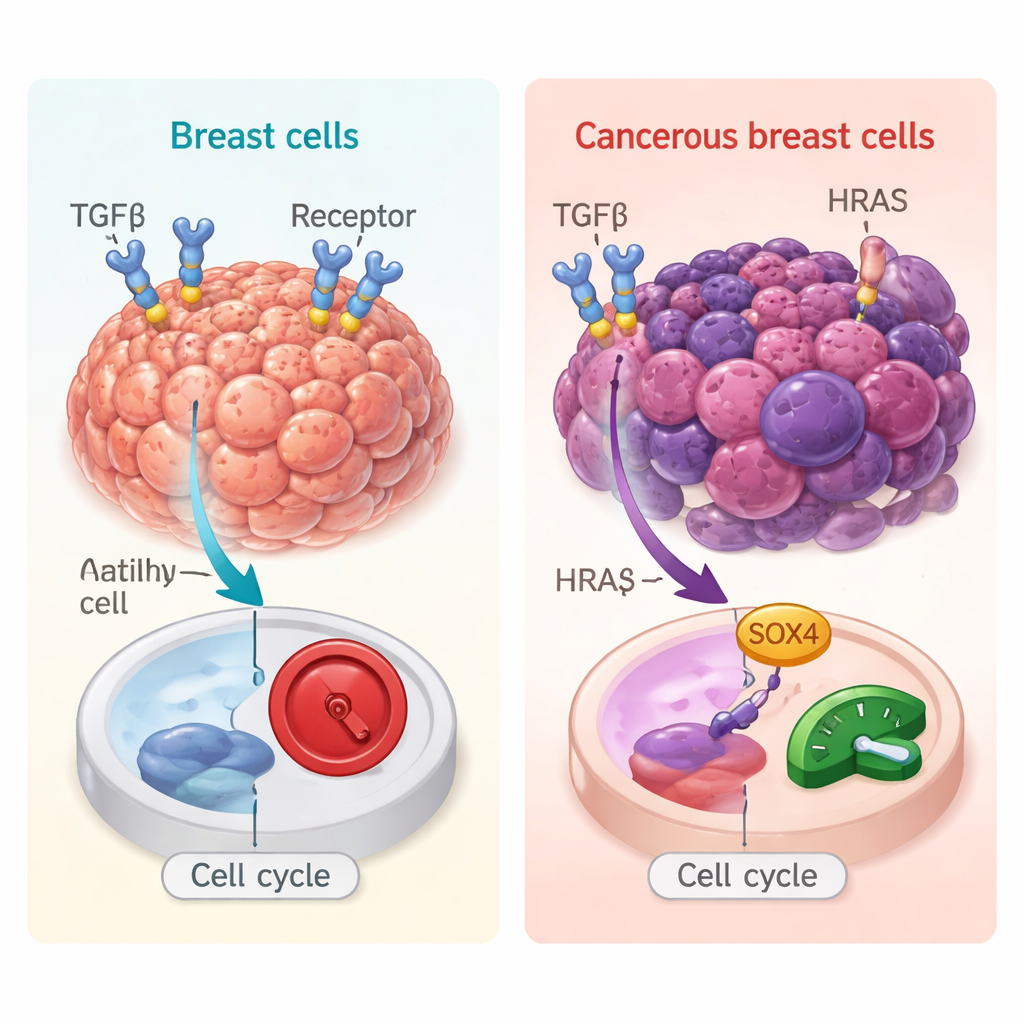
Rewiring the Genome’s On/Off Switches
The team looked at chromatin, the DNA-and-protein packaging that controls which genes are accessible and can be turned on. After treating the 3D cell clusters with TGFβ, they saw widespread opening of chromatin at gene start sites and distant enhancer regions in both normal-like and HRAS-mutant cells. However, the pattern of newly accessible sites differed sharply between the two. In the cancer-like cells, binding sites for a transcription factor called SOX4 were especially enriched. At the same time, gene expression profiling showed that TGFβ was no longer just turning on well-known programs like epithelial–mesenchymal transition (linked to invasion) and metabolic shifts; in the HRAS-mutant cells it also boosted genes involved in DNA replication, DNA repair, and progression through key cell-cycle checkpoints.
SOX4 as a Molecular Middleman
Diving deeper, the researchers showed that SOX4 sits at the heart of this switch. Single-cell RNA sequencing data from primary breast tumors revealed that SOX4 is co-expressed with cell-cycle drivers such as CDK4, CDK6 and cyclin D1 (CCND1), particularly in actively dividing cancer cells. In their 3D models, TGFβ increased SOX4 levels, and SOX4 was found bound near the control regions of these cell-cycle genes, especially CDK4. When SOX4 was reduced using genetic tools, TGFβ could no longer effectively turn on many of its target genes, including those linked to cell division and tissue remodeling, and the cancer-like spheres became less invasive and more stable. Biochemical experiments further demonstrated that SOX4 works together with SMAD3, a key TGFβ signal carrier, forming a cooperative module in the nucleus that amplifies gene activation.
Undermining a Front-Line Breast Cancer Drug
Palbociclib is a widely used drug that blocks CDK4 and CDK6, proteins that drive cells from a resting state into active division. Many patients eventually develop resistance to this treatment. The authors combined large drug-response datasets with lab experiments in multiple breast cancer cell lines and 3D cultures. They found that stronger TGFβ activity, along with SOX4 and related gene signatures, is associated with higher palbociclib doses needed to inhibit growth. In the lab, adding TGFβ made cancer cells less sensitive to palbociclib, while blocking TGFβ signaling or knocking down SOX4 restored sensitivity. Long-term exposure to palbociclib produced resistant cell lines that showed heightened TGFβ signaling, more SOX4, and stronger marks of active chromatin at the SOX4 gene itself. In zebrafish models, inhibiting TGFβ substantially reduced growth of tumors derived from palbociclib-resistant cells. 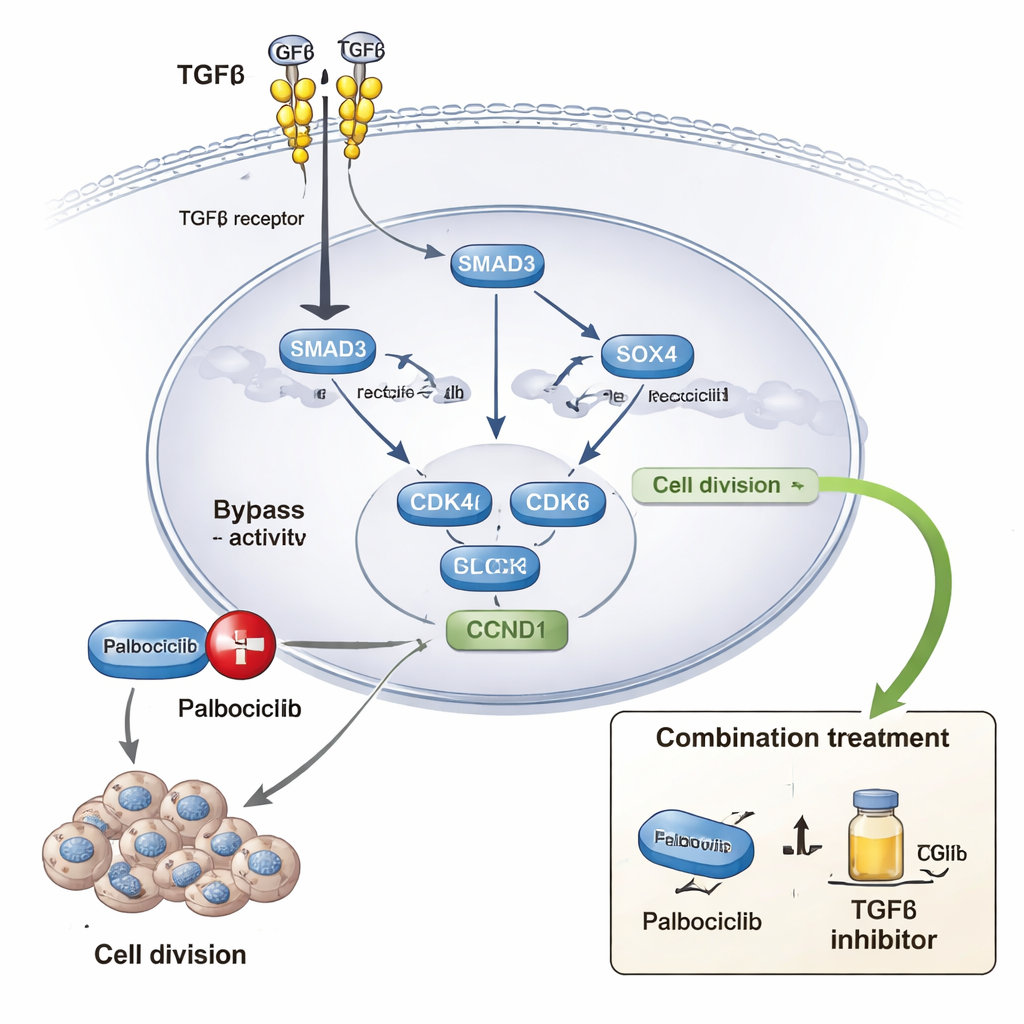
What This Means for Patients and Future Therapies
To a non-specialist, the key message is that a signal meant to protect us from cancer can, in the wrong context, be co-opted to help tumors survive and outsmart drugs. Here, TGFβ teams up with SOX4 to open up crucial regions of the genome, switch on cell-cycle engines like CDK4 and CDK6, and blunt the impact of palbociclib. The findings suggest that, for some breast cancers, combining CDK4/6 inhibitors with drugs that block TGFβ signaling—or that target SOX4-driven programs—could delay or overcome resistance. While more clinical work is needed, this study maps a detailed molecular escape route that cancer cells use and points to new ways to cut it off.
Citation: Ali, M.M., Itoh, Y., Badji, A.M.P. et al. TGFβ signaling promotes cell cycle progression and resistance to the CDK4/6 inhibitor palbociclib through SOX4 transcriptional modulation in breast cancer cells. Cell Death Dis 17, 209 (2026). https://doi.org/10.1038/s41419-026-08435-4
Keywords: breast cancer, TGF-beta signaling, SOX4, palbociclib resistance, cell cycle