Clear Sky Science · en
LAP2α drives breast tumorigenesis by mitigating replication stress
Why this matters for breast cancer
Breast tumors grow in part because cancer cells find ways to survive constant stress as they copy their DNA. This study uncovers how a lesser-known protein, called LAP2α, helps breast cancer cells protect their DNA during replication, thereby encouraging tumor growth and making cancers harder to treat. Understanding this hidden support system suggests new ways to weaken tumors and improve the effects of existing drugs.
A hidden helper inside tumor cell nuclei
Every time a cell divides, it must accurately copy its DNA. In cancer cells, this process is especially strained: growth signals are stuck in the “on” position, and the replication machinery is pushed to its limits. This pressure, known as replication stress, leaves stretches of single-stranded DNA that must be quickly coated and stabilized by a protein complex called RPA. The authors previously found that LAP2α, a protein linked to the internal scaffolding of the nucleus, can bind RPA and help it reach damaged DNA. In the present work, they asked whether this LAP2α–RPA partnership actually fuels breast tumor development and affects how tumors respond to treatment.
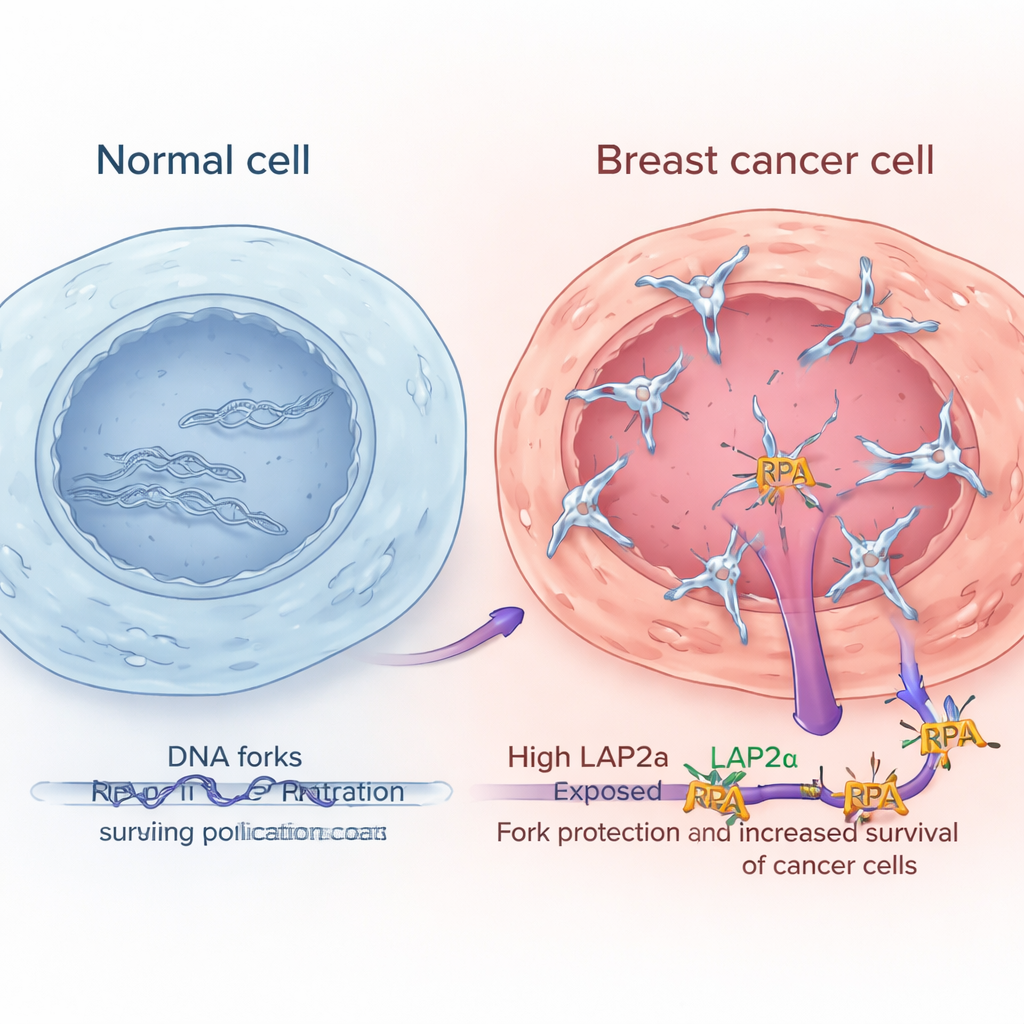
High LAP2α levels mark more aggressive breast tumors
By analyzing large public gene expression datasets and examining tumor samples under the microscope, the researchers showed that LAP2α is consistently higher in breast cancer tissue than in nearby normal breast tissue. Its levels rise with tumor grade, meaning more abnormal and aggressive cancers tend to have more LAP2α. This pattern appeared across multiple major breast cancer subtypes, including hormone receptor–positive, HER2-enriched, and triple-negative tumors. Importantly for patients, those whose tumors had high LAP2α tended to have worse survival. In contrast, the core RPA components themselves did not show similar changes or clear links to outcome, suggesting that it is the boosted function of LAP2α, rather than simply more RPA, that helps cancer cells better cope with replication stress.
Turning off LAP2α slows tumors and exposes weak points
To move beyond correlations, the team used mouse models of breast cancer in which LAP2α could be selectively removed. Once mammary tumors had formed, genetically deleting LAP2α caused tumors to grow more slowly and extended the animals’ survival. Tumor cells lacking LAP2α divided less and carried more signs of DNA damage, as shown by increased staining for DNA break markers and reduced RPA coating on DNA. When these LAP2α-deficient tumor cells were transplanted into new mice, they again formed smaller tumors and showed heightened sensitivity to DNA-damaging chemotherapy, including the platinum drug cisplatin and a PARP inhibitor. Similar experiments in human breast cancer cell lines confirmed that reducing LAP2α made cells more vulnerable to several genotoxic drugs, while restoring normal LAP2α—but not a mutant form that cannot bind RPA—rescued both DNA protection and drug resistance.
How LAP2α helps protect fragile DNA strands
To dissect the mechanism, the researchers recreated the DNA binding steps in test-tube assays. They mixed purified RPA, single-stranded DNA, and either normal LAP2α or a variant unable to interact with RPA. They found that LAP2α directly increased how efficiently RPA coated single-stranded DNA and helped RPA stretch and stabilize these fragile regions, acting much like a loading helper or chaperone. When DNA was present, RPA tended to leave LAP2α and fully commit to binding the single strand, showing that LAP2α is not part of the final protective coat but instead hands RPA off to DNA. Without a functional LAP2α–RPA interaction, more replication forks collapsed, DNA breaks accumulated, and cancer cells were more likely to die, especially when additional damage was induced by chemotherapy.
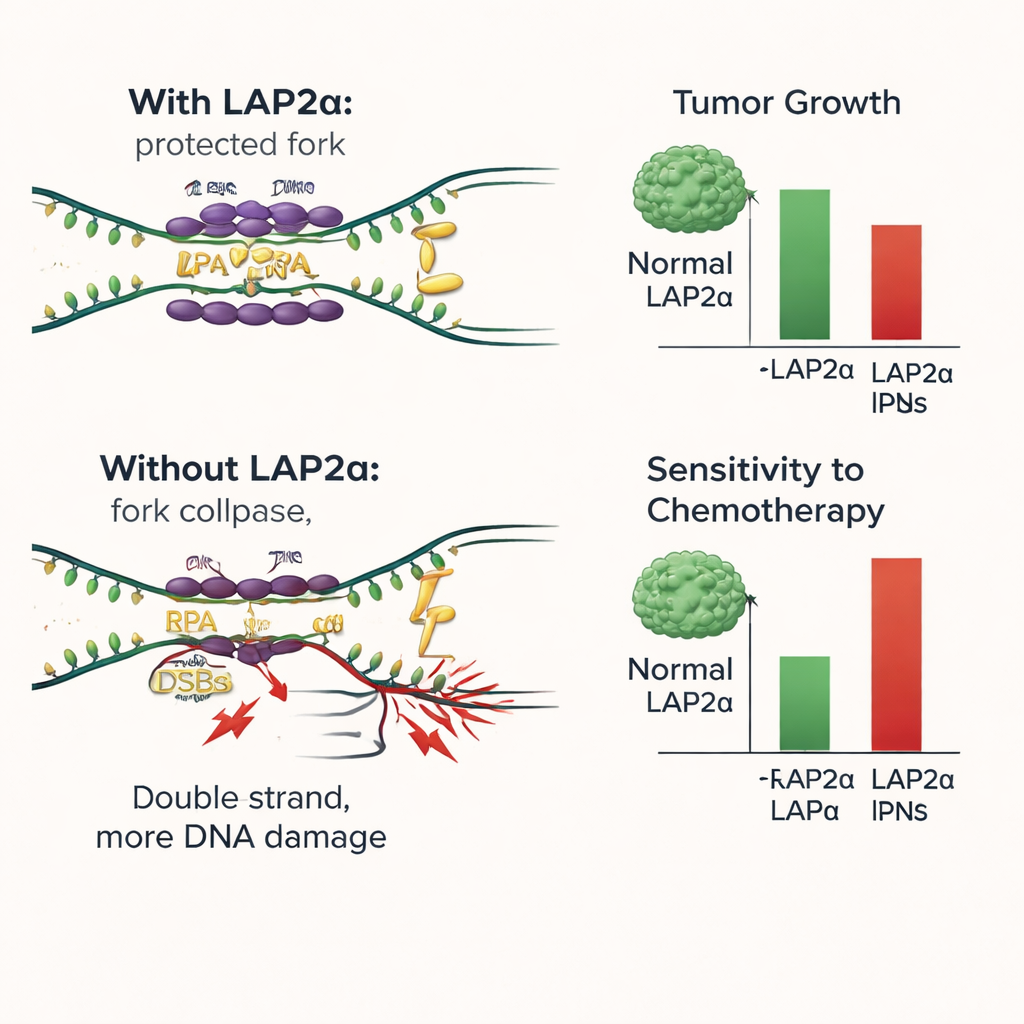
What this means for future breast cancer treatment
In plain terms, this study shows that LAP2α serves as a backstage assistant that helps breast cancer cells survive the strain of copying their DNA. By efficiently loading RPA onto vulnerable DNA, LAP2α reduces damage and supports continued tumor growth. Removing or disabling LAP2α tips the balance: DNA damage piles up, cells stop dividing, and conventional drugs that attack DNA become more effective. These findings suggest that LAP2α could be used as a marker of poor prognosis and as a new therapeutic target. Drugs that block LAP2α or its grip on RPA might make existing treatments such as platinum agents and PARP inhibitors work better, especially in tumors that rely heavily on this stress-buffering system.
Citation: Ma, Y., Qin, Y., Bao, P. et al. LAP2α drives breast tumorigenesis by mitigating replication stress. Cell Death Dis 17, 201 (2026). https://doi.org/10.1038/s41419-026-08433-6
Keywords: breast cancer, DNA replication stress, LAP2 alpha, replication protein A, chemotherapy sensitivity