Clear Sky Science · en
Targeting glycerophospholipid biosynthesis overcomes chemoresistance driven by SLFN11 loss in Ewing sarcoma
Why this research matters for childhood cancer
Ewing sarcoma is a rare but aggressive cancer that mostly strikes children and teenagers. Many patients initially respond well to chemotherapy, but for those whose cancer comes back, current treatments often fail. This study asks a pressing question: when Ewing sarcoma cells become resistant to chemotherapy, what changes inside them help them survive—and can those changes be turned into new weaknesses doctors can target?
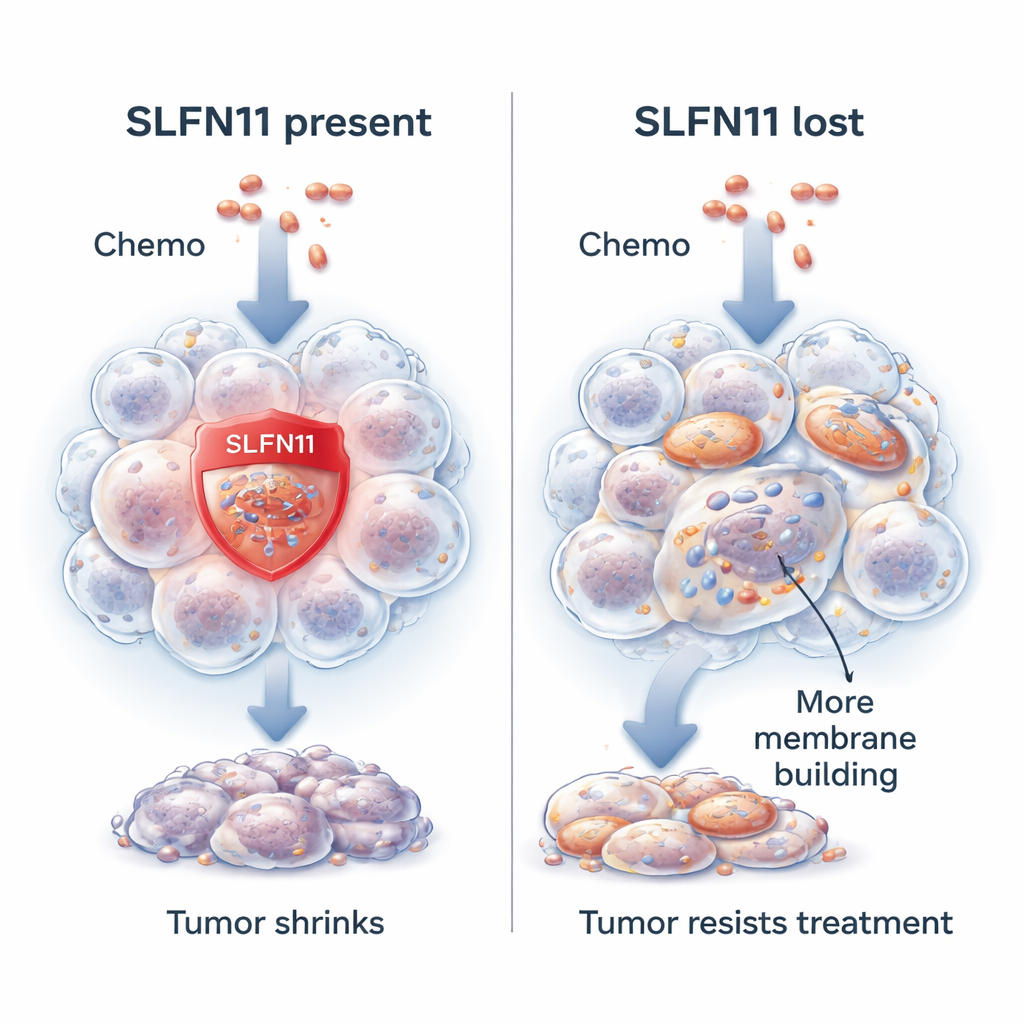
A gene that makes cancer cells easier to kill
The researchers focus on a gene called SLFN11. In many Ewing sarcoma tumors, SLFN11 is highly active and makes cancer cells much more sensitive to drugs that damage DNA, the cell’s genetic blueprint. When these drugs are given, SLFN11 helps shut down DNA repair and pushes the cancer cell toward death. Patients whose tumors have higher SLFN11 levels tend to live longer and respond better to treatment. However, about one in ten tumors either start out with little or no SLFN11 or lose it over the course of therapy. When that happens, the same chemotherapy is far less effective, yet the cancer cells remain perfectly capable of growing.
How cancer cells rewire their use of fuel and fats
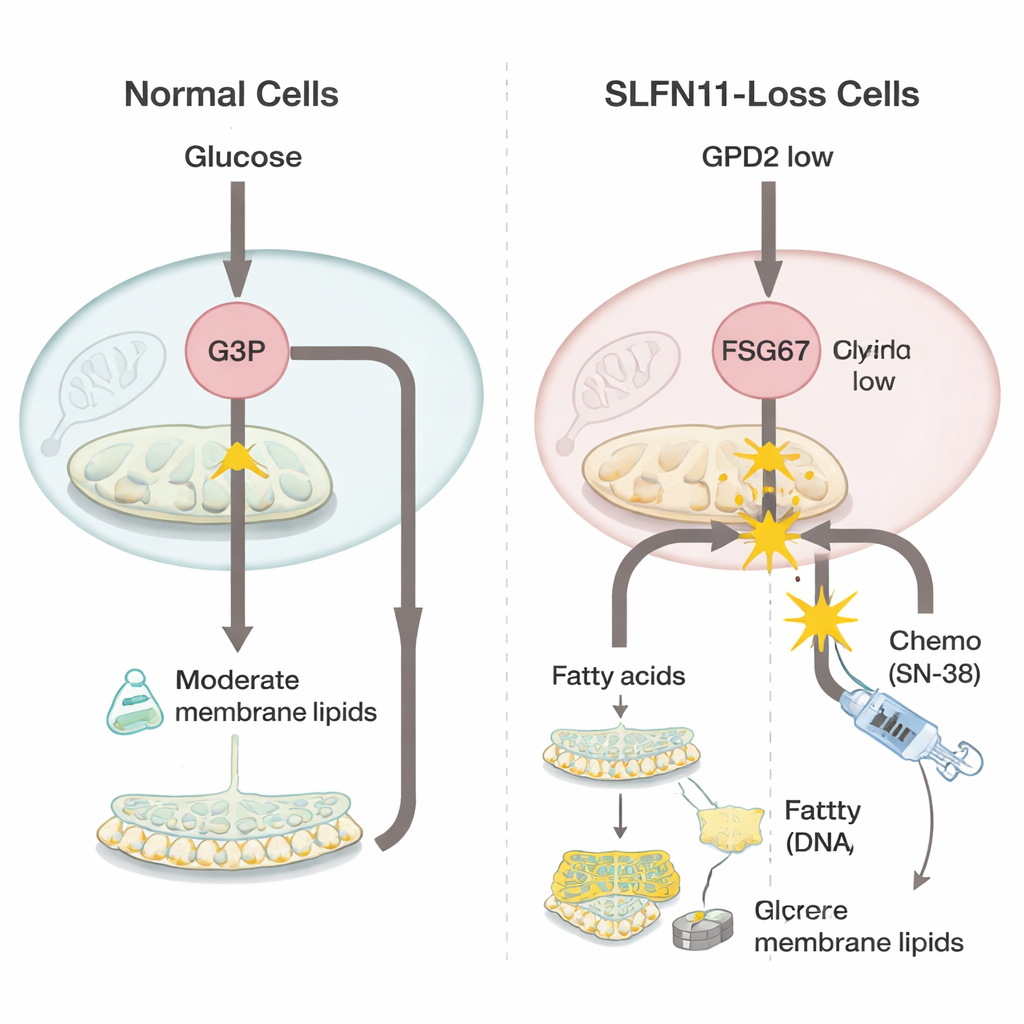
To understand what changes when SLFN11 is lost, the team compared Ewing sarcoma cells with and without this gene using powerful “omics” tools that measure thousands of genes and small molecules at once. They discovered that cells lacking SLFN11 dial down an enzyme in their mitochondria called GPD2, which normally helps burn a molecule called glycerol‑3‑phosphate as part of energy production. When GPD2 is reduced, glycerol‑3‑phosphate builds up. Rather than going to waste, this extra building block is diverted into making more fatty molecules that form cell membranes, a group known as glycerophospholipids. The cells also showed signs of making more flexible, unsaturated fats, which can help fast‑growing tumors adapt to stress.
Turning a survival trick into a weak point
Because these SLFN11‑deficient cells lean more heavily on membrane building, the scientists tested whether blocking this process could restore chemotherapy sensitivity. They used a compound called FSG67, which inhibits a key step in glycerophospholipid production. On its own, the standard DNA‑damaging drug SN‑38 had become much less potent in SLFN11‑deficient cells, mirroring the clinical problem of resistance. But when SN‑38 was combined with FSG67, the formerly resistant cells were hit much harder, and the two drugs worked together better than expected from their individual effects. In contrast, in cells that still had SLFN11 and were already highly sensitive to SN‑38, adding FSG67 offered little extra benefit and could even be slightly counterproductive. This pattern suggests that the newfound dependence on fat and membrane production is specific to the resistant, SLFN11‑low state.
A potential noninvasive signal of hard‑to‑treat tumors
The researchers then asked whether this metabolic rewiring could be detected in actual tumors, not just cells in a dish. They grew Ewing sarcoma tumors in mice, with or without SLFN11, and used nuclear magnetic resonance (NMR) to examine the chemical makeup of the tumor extracts. Tumors lacking SLFN11 showed a higher ratio of two choline‑containing molecules linked to membrane turnover: phosphocholine and glycerophosphocholine. A high phosphocholine/glycerophosphocholine ratio has been associated in other cancers with more aggressive behavior and poorer response to treatment. Because choline signals can be measured with advanced imaging techniques, this kind of shift may one day serve as a noninvasive marker to flag Ewing sarcoma tumors that have adopted this resistant, membrane‑building mode.
What this could mean for future treatments
Overall, the study shows that when Ewing sarcoma cells lose SLFN11 and become less sensitive to DNA‑damaging chemotherapy, they compensate by rewiring their metabolism toward making more membrane fats. This shift does not just help the cells survive; it also creates a new Achilles’ heel. Blocking glycerophospholipid production with a drug like FSG67 can partially restore the killing power of chemotherapy in these resistant cells. While FSG67 itself is not yet a clinical drug, the work points toward a strategy in which doctors might one day match patients’ tumors by SLFN11 status and metabolic features, then pair DNA‑damaging therapies with targeted blockers of fat and membrane synthesis to overcome resistance.
Citation: Chakraborty, K., Burman, R., Satheesh, S. et al. Targeting glycerophospholipid biosynthesis overcomes chemoresistance driven by SLFN11 loss in Ewing sarcoma. Cell Death Dis 17, 190 (2026). https://doi.org/10.1038/s41419-026-08432-7
Keywords: Ewing sarcoma, chemoresistance, SLFN11, cancer metabolism, lipid biosynthesis