Clear Sky Science · en
Modulation of immune cells and metabolic reprogramming in efferocytosis
How Our Bodies Quietly Clean Up Dying Cells
Every day, billions of our cells reach the end of their lives. Yet we do not feel this microscopic turnover, nor do we become sick from the debris. This quiet housekeeping is handled by immune and tissue cells that recognize and swallow dying cells, preventing harmful inflammation. The reviewed research explains how this clean-up process, called efferocytosis, not only keeps tissues tidy but also rewires the metabolism of the cells doing the cleaning, shaping whether our immune system calms down, fights infection, or—if things go wrong—contributes to disease.
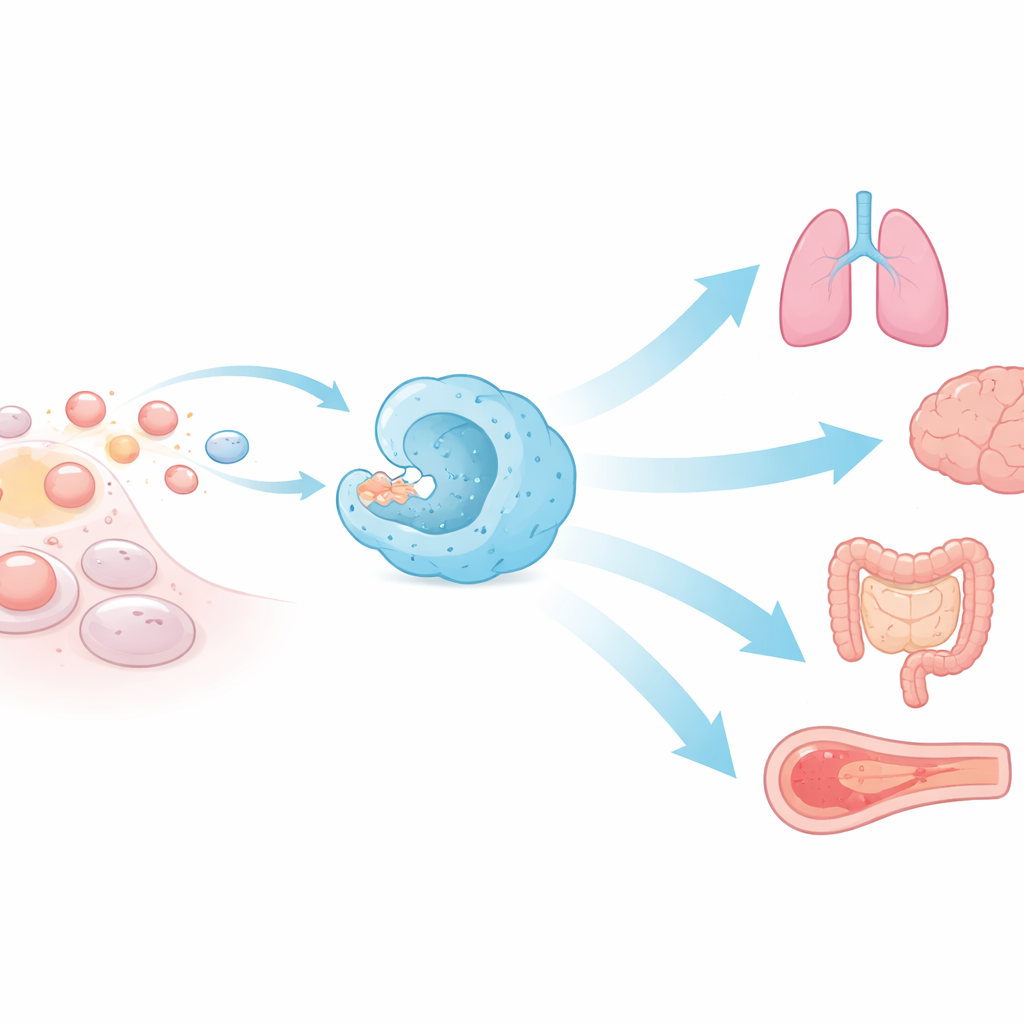
The Body’s Silent Garbage Collectors
Efferocytosis is the name given to the engulfment and removal of apoptotic, or programmedly dying, cells. Specialized immune cells known as macrophages and dendritic cells perform most of this work, but many non-immune cells—such as epithelial cells in the gut, lung, eye, and testes—can also step in. Dying cells send out chemical “find me” cues that attract nearby scavenger cells, then display “eat me” flags on their surface while turning off “don’t eat me” signals. Once recognized, they are wrapped into a membrane pocket, pulled inside, and broken down in enzyme-filled compartments. This swift, multi-step choreography explains why apoptotic cells are rarely seen under the microscope despite massive daily cell loss.
Housekeeping That Protects Organs
Efficient efferocytosis protects the structure and function of many organs. In the eye, failure of retinal cells to clear dying photoreceptor fragments leads to vision loss. In the testes, poor removal of dying germ cells can cause infertility. During pregnancy, efferocytosis of placental cells encourages maternal tolerance by dampening inflammatory molecules and boosting calming ones. In the brain, microglia prune excess neurons and support healthy wiring, while in the gut, Paneth cells and other phagocytes clean up dying cells to prevent chronic intestinal inflammation. In the lungs, airway macrophages continuously remove dead cells, avoiding the release of danger signals that can drive asthma, chronic obstructive pulmonary disease, or severe viral damage.
When Infections Turn Cleanup into a Double-Edged Sword
During infections, efferocytosis becomes more complex. Pathogens often trigger cell death, and the resulting engulfment of infected corpses can either weaken or strengthen host defense. Some viruses and bacteria exploit the anti-inflammatory side of efferocytosis to hide from immune attack or to spread, using engulfed vesicles as “Trojan horses.” In other settings, swallowing infected dead cells delivers microbial fragments into macrophages and dendritic cells, which then present these fragments to T cells and rally protective immune responses. Depending on the microbe and tissue, efferocytosis can bias T helper cells toward different immune profiles, supporting either bacterial clearance or, if self-molecules are presented alongside microbial ones, the emergence of autoimmune T cells that attack the body’s own tissues.
Fuel from the Fallen: How Metabolism Is Rewired
Breaking down a whole cell is metabolically demanding, but it also floods the scavenger with nutrients—lipids, sugars, amino acids, and nucleotides. The authors describe a growing field, sometimes called efferotabolism, that tracks how this nutrient surge reprograms the metabolism of macrophages and dendritic cells. Lipids from dying cells activate nuclear receptors that boost cholesterol export, support mitochondrial fat burning, and promote an anti-inflammatory, tissue-repairing state. Amino acids such as arginine, methionine, and tryptophan are recycled into molecules that power repeated rounds of engulfment and trigger pro-resolving signals. Short bursts of glucose breakdown and related pathways supply energy, maintain redox balance, and help keep receptors and membranes in the right state for continual cleanup. These intertwined metabolic shifts determine whether efferocytic cells stay tolerant, switch to repair mode, or retain strong antimicrobial activity.
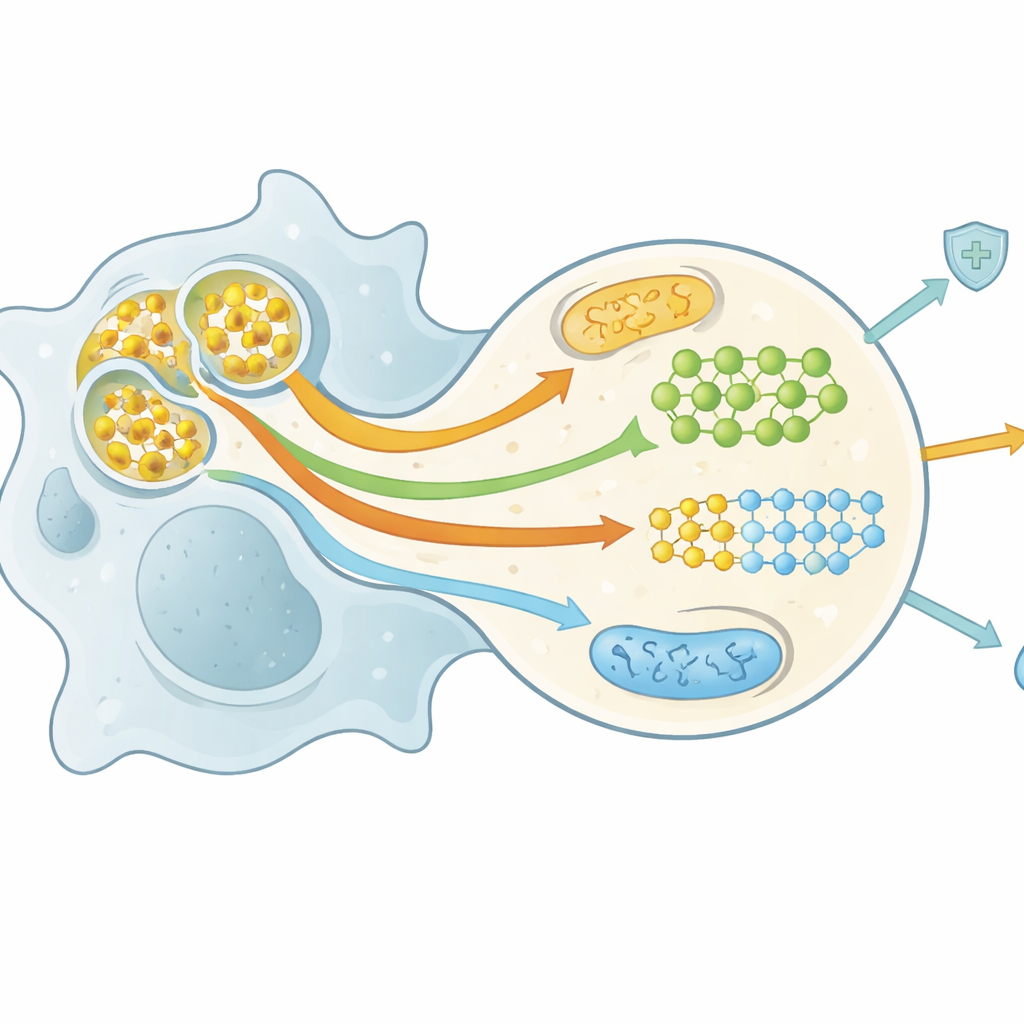
Open Questions and Future Treatment Paths
The review highlights that not all efferocytes, or cell eaters, behave the same. Professional immune cells and tissue cells may rely on distinct metabolic programs, and different forms of cell death—such as necrosis or ferroptosis—likely leave different metabolic and immune footprints. Key unknowns include how mixed signals from both self and microbial components inside the same dying cell shape energy pathways and long-term immunity. By mapping these links between cleanup, metabolism, and immune fate, researchers hope to design therapies that gently steer efferocytosis: enhancing it to resolve chronic inflammation and aid wound repair, or tuning it to preserve antimicrobial strength without tipping into autoimmunity. In essence, understanding how the body feeds on its own cellular leftovers may unlock new ways to keep inflammation, infection, and tolerance in healthy balance.
Citation: Oliveira, K.C., Marcos, C.M., Penteado, L.d.A. et al. Modulation of immune cells and metabolic reprogramming in efferocytosis. Cell Death Dis 17, 257 (2026). https://doi.org/10.1038/s41419-026-08431-8
Keywords: efferocytosis, immune tolerance, innate immunity, immunometabolism, chronic inflammation