Clear Sky Science · en
Targeting hypoxic exosomal IGFBP2 overcomes CD47-mediated immune evasion in glioblastoma
Why starving tumors of oxygen can backfire
Doctors have long known that glioblastoma, a deadly brain cancer, often grows in regions starved of oxygen. These hypoxic pockets make the tumor harder to treat. This study reveals how low oxygen helps cancer cells hide from the immune system, and suggests a new combination treatment that could make them easier for the body to attack.
A deadly brain tumor that dodges our defenses
Glioblastoma is the most aggressive common brain tumor in adults, with typical survival measured in months. Even though immune cells infiltrate these tumors, cancer cells frequently escape destruction. One key escape route is a surface protein called CD47, sometimes called a "don’t eat me" signal, which tells nearby immune cells such as macrophages not to engulf and destroy the tumor cell. Drugs that block CD47 are already being tested, but results in solid tumors like glioblastoma have been mixed, suggesting that other factors in the tumor environment may be undermining these therapies.
Hypoxic zones and tiny cancer parcels
Using single-cell RNA sequencing, the researchers mapped thousands of cells from different regions of glioblastoma samples, comparing the low-oxygen core of the tumor with its outer edge. They found a particularly aggressive cell subtype in the hypoxic core, called mesenchymal-like GBM cells, that strongly expressed CD47 and a protein called IGFBP2. At the same time, they discovered that these core cells released large numbers of nano-sized vesicles known as exosomes, which carried IGFBP2 on their surface. Because exosomes can travel through the brain and even into the bloodstream, they can spread signals far beyond where they are made. 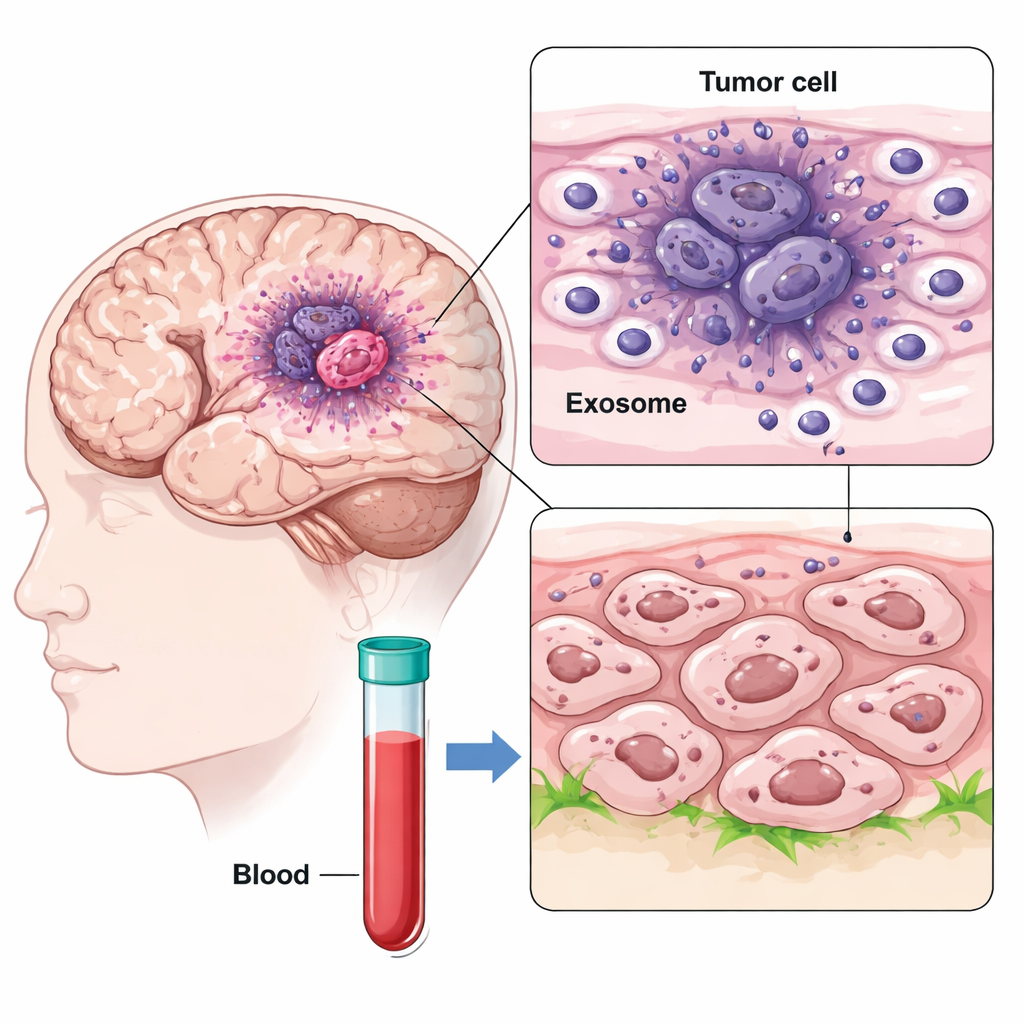
How a partner protein boosts the “don’t eat me” signal
The team then dug into what IGFBP2 was doing. They showed that under low oxygen, a hypoxia sensor protein called HIF-2α switches on the IGFBP2 gene in tumor cells. IGFBP2 then binds to specific receptors called integrins on the surface of exosomes, decorating their outer membrane. When these IGFBP2-coated exosomes fuse with other tumor cells, they activate a chain of signals inside the cell involving proteins known as FAK and STAT3. This cascade ultimately increases the amount of CD47 on the tumor cell surface, strengthening the "don’t eat me" message to macrophages and further shielding cancer cells from immune attack. 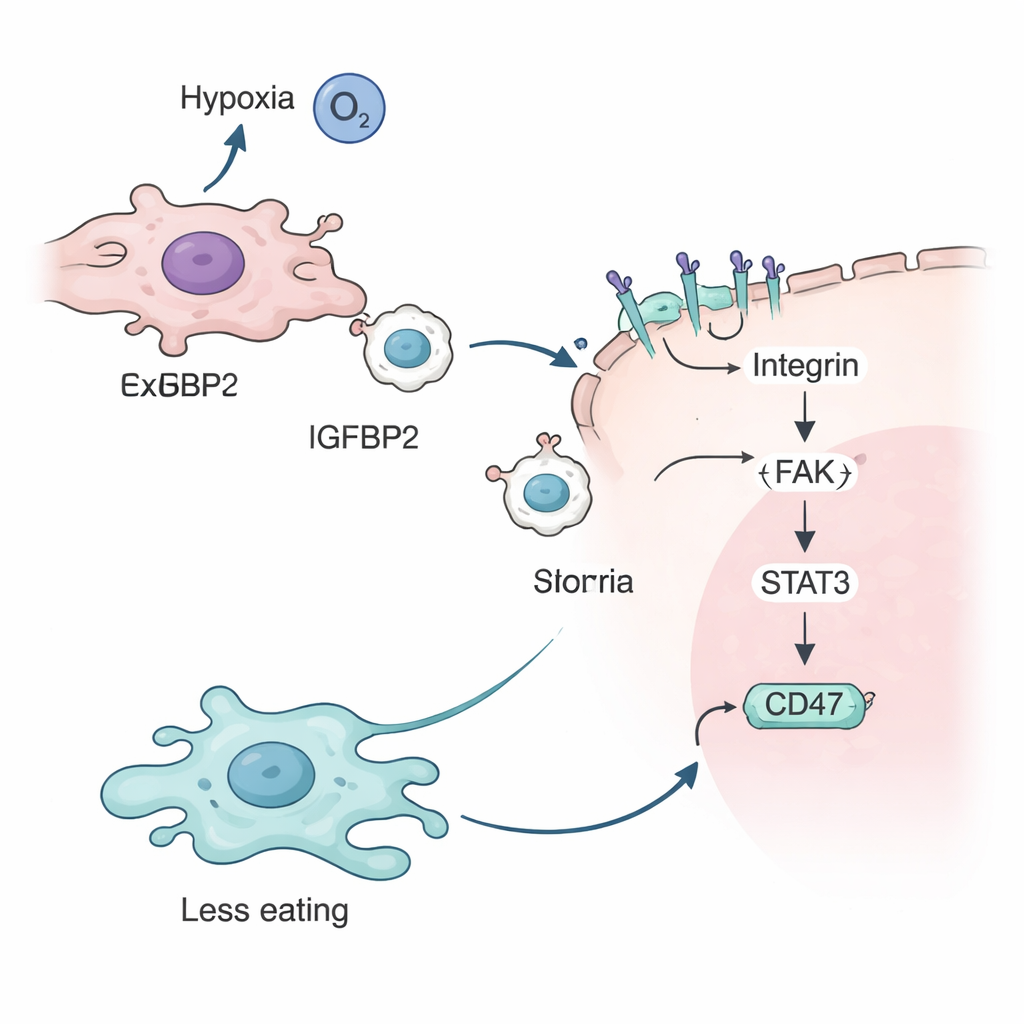
Evidence from patient samples and animal models
To connect these lab findings to real disease, the researchers examined tumor tissue and blood from people with glioma. They found that IGFBP2 levels were higher in both tumors and blood exosomes from patients with more advanced disease, suggesting IGFBP2-positive exosomes could serve as a blood-based marker of tumor grade. In mouse models, lowering IGFBP2 in glioblastoma cells made them more vulnerable to being eaten by macrophages, slowed tumor growth, and extended survival. Conversely, adding extra IGFBP2-rich exosomes boosted CD47 levels, reduced immune cell eating, and sped tumor progression.
A promising one-two punch for therapy
Finally, the team tested whether blocking IGFBP2 could make CD47-targeted therapy more effective. In mice with brain tumors, treatment with antibodies against both IGFBP2 and CD47 led to stronger macrophage activity, smaller tumors, and longer survival than either treatment alone. By cutting off the hypoxia-driven IGFBP2 signal and directly blocking the CD47 "don’t eat me" signal, this combination therapy attacked the tumor’s immune camouflage from two directions. For patients, these findings suggest that measuring IGFBP2 in blood exosomes might help identify those most likely to benefit from CD47-based immunotherapy, and that pairing IGFBP2 and CD47 inhibitors could be a more powerful strategy against glioblastoma than either drug by itself.
Citation: Qi, Y., Zhao, R., Zhang, X. et al. Targeting hypoxic exosomal IGFBP2 overcomes CD47-mediated immune evasion in glioblastoma. Cell Death Dis 17, 192 (2026). https://doi.org/10.1038/s41419-026-08430-9
Keywords: glioblastoma, exosomes, immune evasion, CD47, hypoxia