Clear Sky Science · en
The long noncoding RNA AC093895.1 promotes ovarian cancer formation and metastasis through a positive feedback network dependent on the transcription factor SOX4
Why this research matters
Ovarian cancer is often called a “silent killer” because it tends to be diagnosed late, when it has already spread and is harder to treat. Many women relapse even after surgery and chemotherapy, and current blood tests are not always reliable at predicting who will do poorly. This study uncovers a new molecular “control loop” inside ovarian cancer cells that helps tumors grow and spread, and suggests a fresh target that might one day improve both diagnosis and treatment.
A hidden RNA with a loud impact
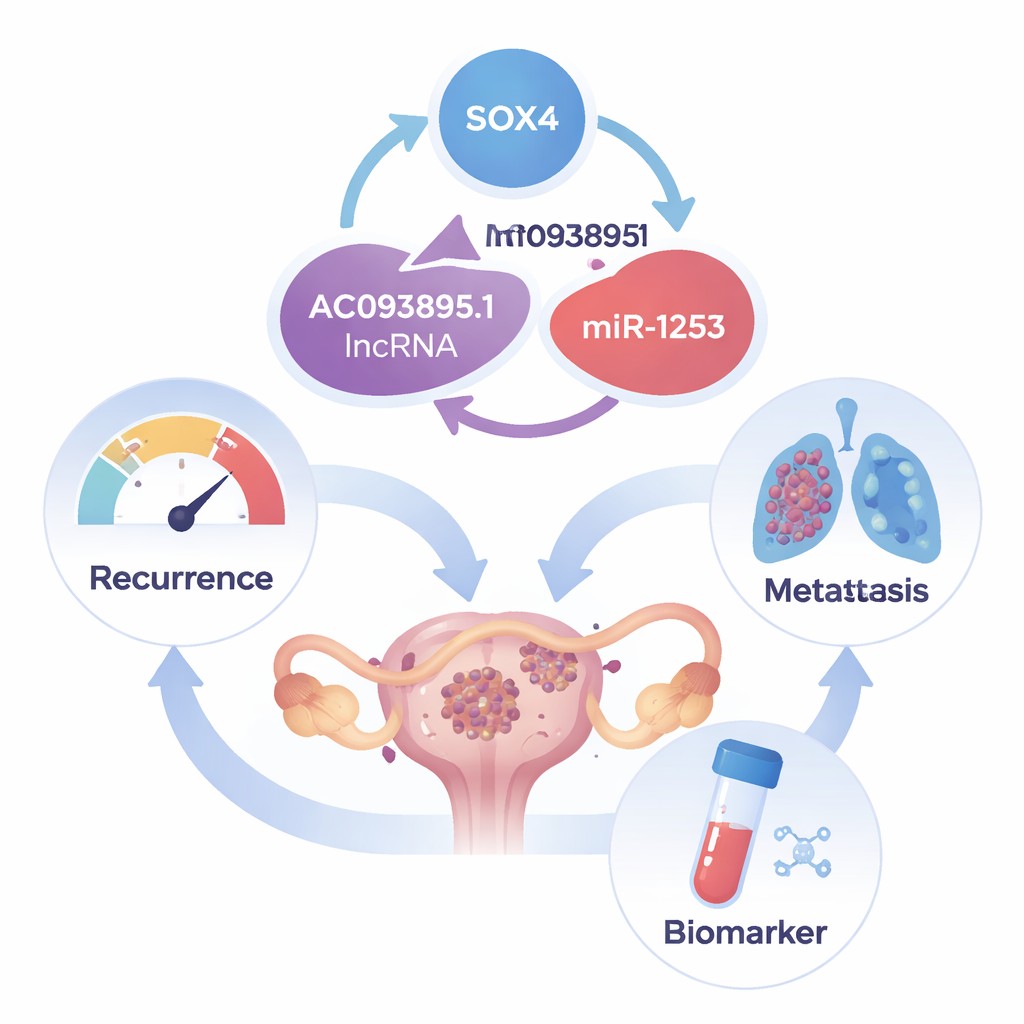
Our DNA carries instructions not only for making proteins, but also for many RNA molecules that never become proteins. These so‑called long noncoding RNAs act more like conductors than instruments in a cellular orchestra, coordinating how other genes behave. The researchers focused on one such molecule, called AC093895.1, and found that it is much more abundant in ovarian cancer tissue than in healthy ovary samples. Using large public datasets and tumor samples from patients, they showed that high AC093895.1 levels are especially common in metastatic tumors and are linked to worse survival, suggesting that this RNA tracks with aggressive disease.
Turning growth up and self‑destruct down
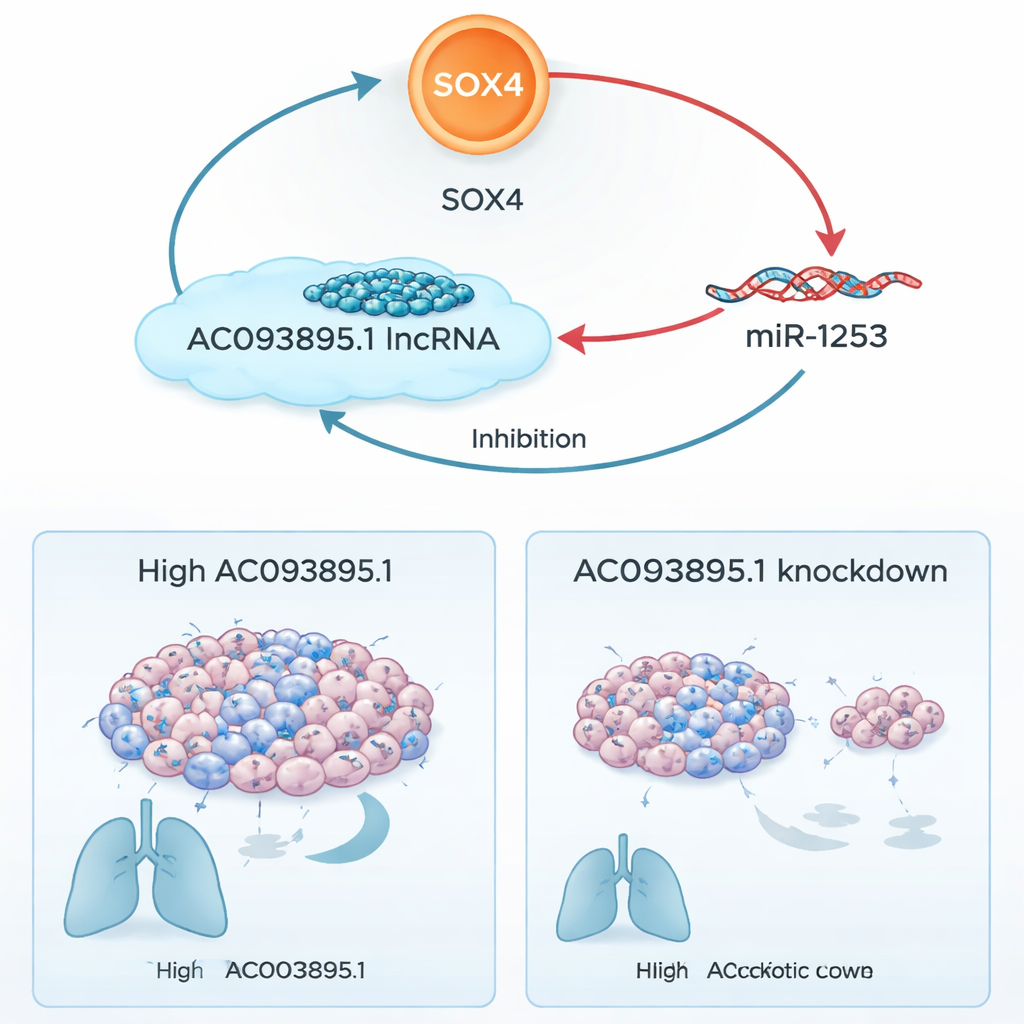
To understand what AC093895.1 actually does, the team used ovarian cancer cell lines grown in the lab and reduced the amount of this RNA using genetic tools. When AC093895.1 was knocked down, cancer cells grew more slowly, got stuck before entering the active division phase of the cell cycle, and were more likely to undergo programmed cell death. Their ability to migrate and invade through barriers—key steps in forming metastases—also dropped sharply. At the molecular level, markers associated with cell death went up, while proteins that drive cell division and cell movement went down. In mouse models, tumors formed from AC093895.1‑depleted cells were smaller, had fewer dividing cells, more dying cells, and produced fewer lung metastases.
An RNA “sponge” that frees a cancer‑promoting gene
Digging deeper, the scientists discovered that AC093895.1 mainly resides in the cytoplasm of cancer cells, where it can interact with small regulatory RNAs called microRNAs. They identified one such partner, miR‑1253, which normally acts as a tumor suppressor in several cancer types. AC093895.1 and miR‑1253 bind each other and pull in opposite directions: when AC093895.1 is high, miR‑1253 is low, and vice versa. By acting as a molecular “sponge,” AC093895.1 soaks up miR‑1253 and prevents it from restraining its targets. One of those targets is SOX4, a transcription factor protein known to promote cancer cell survival, invasion, and spread. When miR‑1253 is blocked, SOX4 levels rise; when AC093895.1 is reduced, SOX4 levels fall. Functionally, blocking miR‑1253 can undo many of the beneficial effects of AC093895.1 knockdown, confirming that this pathway is central to how the RNA drives malignancy.
A self‑reinforcing cancer circuit
Strikingly, the relationship does not end there. SOX4 itself can switch on the gene that produces AC093895.1. The authors showed that SOX4 physically binds to the promoter region—the DNA “on switch”—for AC093895.1 and boosts its activity. This creates a closed circuit: SOX4 increases AC093895.1, AC093895.1 suppresses miR‑1253, and the loss of miR‑1253 allows even more SOX4 to accumulate. In tumor samples from 14 patients, regions with high AC093895.1 almost always had high SOX4 and low miR‑1253, and these patterns were associated with more advanced stages and greater lymph node and organ spread. Breaking the loop by lowering AC093895.1 weakened SOX4‑driven tumor growth and metastasis in both cell culture and mice.
What this could mean for patients
To a non‑specialist, the SOX4/AC093895.1/miR‑1253/SOX4 loop can be thought of as a stuck accelerator pedal in ovarian cancer cells: once pressed, it keeps telling cells to grow, survive, and travel to new organs. This study shows that AC093895.1 is a key part of that mechanism and that dialing it down can slow tumors and reduce spread in experimental models. While therapies and tests based on this RNA are not yet available, AC093895.1—and the loop it controls—may form the basis of future blood‑ or tissue‑based prognostic tools and targeted treatments aimed at preventing recurrence and metastasis in women with ovarian cancer.
Citation: Huang, B., An, H., Qiu, Y. et al. The long noncoding RNA AC093895.1 promotes ovarian cancer formation and metastasis through a positive feedback network dependent on the transcription factor SOX4. Cell Death Dis 17, 202 (2026). https://doi.org/10.1038/s41419-026-08429-2
Keywords: ovarian cancer, long noncoding RNA, metastasis, SOX4, biomarker