Clear Sky Science · en
HSPA5 promotes YAP/TAZ stability independently of the Hippo pathway and induces proneural-to-mesenchymal transition in glioblastoma
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers, in part because many tumors shift over time into a tougher, more treatment‑resistant state. This study uncovers how a common stress‑response protein, HSPA5, helps drive that shift and keeps tumor‑promoting molecules alive longer than they should be. Understanding this "survival trick" suggests new ways to make glioblastoma less aggressive and more responsive to existing therapies.
A tumor that changes its personality
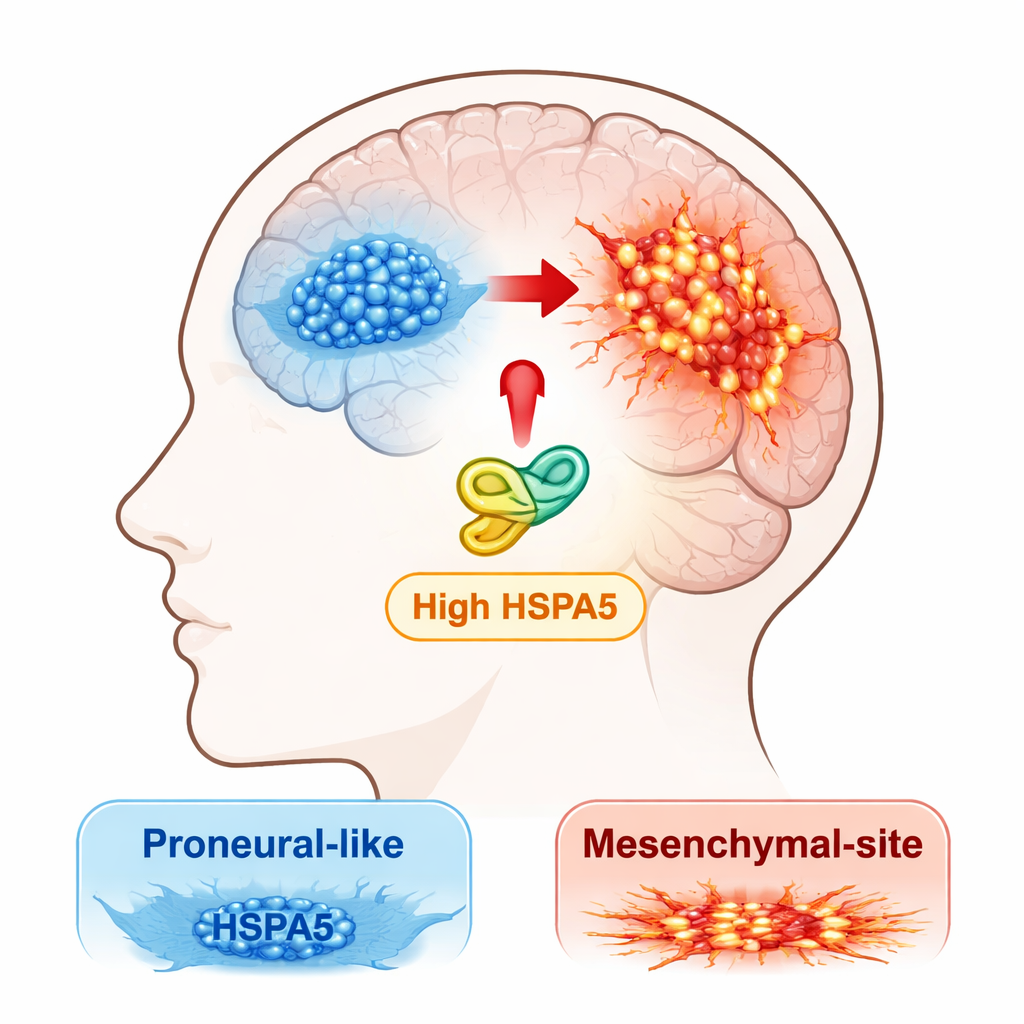
Doctors and scientists now recognize that glioblastoma is not a single disease, but a mix of molecular subtypes. Two major flavors are a “proneural” form, which tends to be somewhat less aggressive, and a “mesenchymal” form, which is more invasive, resistant to treatment, and linked to earlier relapse. Tumors can evolve from the proneural to the mesenchymal state, a process the authors call proneural‑to‑mesenchymal transition, or PMT. The team set out to find which stress‑response proteins might be pushing tumors along this dangerous path.
A stress helper protein in the spotlight
The researchers started by scanning large public cancer datasets and their own patient samples for members of the HSP70 family, a group of “chaperone” proteins that help other proteins fold and survive under stress. One chaperone, HSPA5, stood out. Its activity was highest in the mesenchymal subtype of glioblastoma and markedly higher in tumors than in normal brain tissue. Patients whose tumors carried more HSPA5 tended to have poorer overall survival, highlighting it as a potential driver of aggressive disease rather than a passive bystander.
Forcing cells to become more aggressive
To explore what HSPA5 actually does inside tumor cells, the team grew primary glioblastoma cells taken directly from patients and separated them into proneural‑like and mesenchymal‑like groups. When they forced proneural cells to make extra HSPA5, those cells began to behave more like mesenchymal cells: they divided faster, migrated and invaded more readily, and switched on hallmark mesenchymal markers such as CD44 and c‑MET, while turning down proneural markers like SOX2 and OLIG2. The reverse was also true—reducing HSPA5 levels in mesenchymal cells dampened their growth and invasiveness and partially pushed them back toward a less aggressive profile.
Protecting key growth switches from the cellular trash can
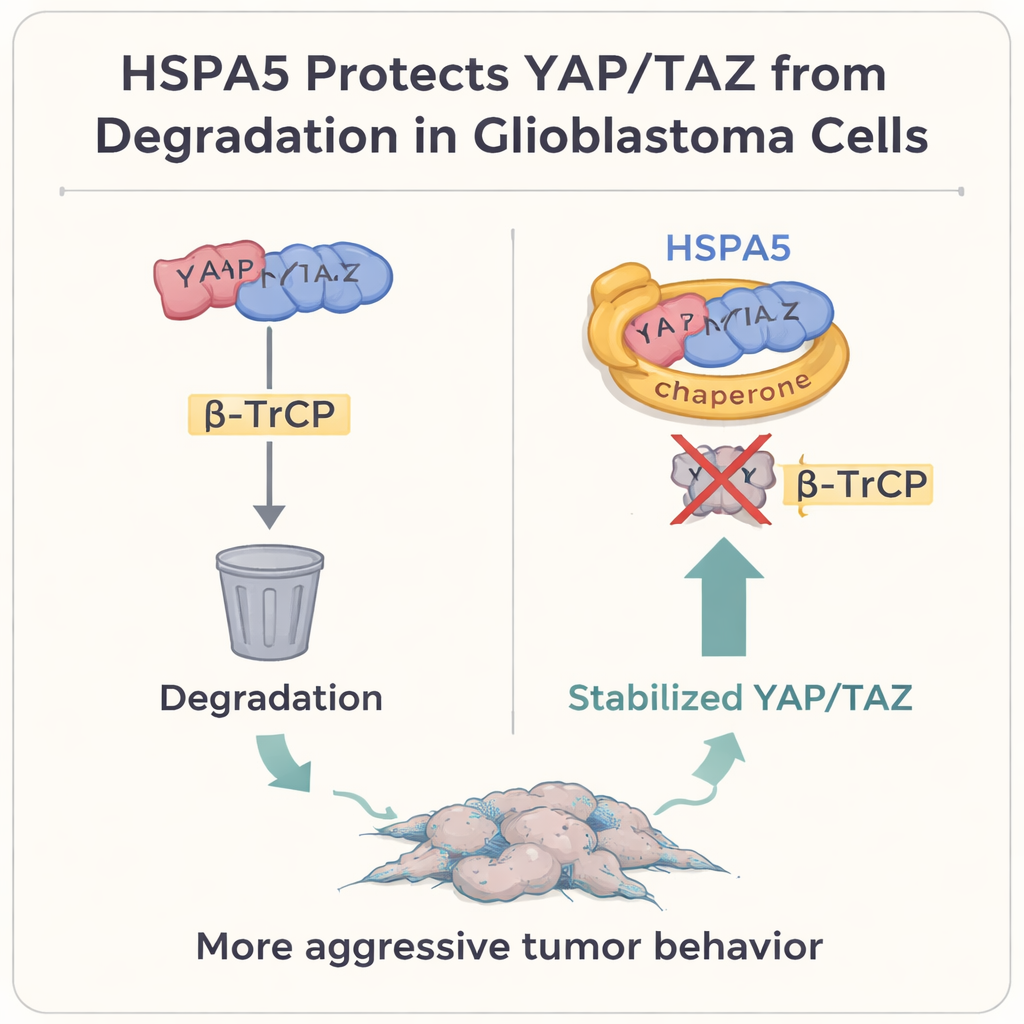
Diving deeper, the authors focused on two powerful growth regulators, YAP and TAZ, which sit at the heart of the Hippo signaling pathway. In many solid tumors, including glioblastoma, these proteins act as master switches that promote cell growth, plasticity, and the mesenchymal state. Normally, when cells want to rein in this program, they tag YAP and TAZ for destruction through a protein called β‑TrCP, which escorts them to the cell’s “trash can,” the proteasome. The study shows that HSPA5 physically binds YAP and TAZ, using its substrate‑binding domain like a protective glove. By doing so, it blocks β‑TrCP from attaching, prevents YAP/TAZ from being ubiquitinated and destroyed, and allows them to accumulate in the nucleus where they turn on mesenchymal genes, including CD44 and c‑MET themselves.
From dishes and mice to patient tumors
The team confirmed this mechanism in several ways. Blocking HSPA5 made YAP and TAZ proteins disappear faster, unless the proteasome was chemically inhibited, and increased the “trash tag” signals on them. Re‑activating YAP and TAZ rescued the loss of growth and invasion seen when HSPA5 was reduced, while silencing YAP/TAZ wiped out the pro‑tumor effects of HSPA5 overexpression. In mouse models where human glioblastoma cells were implanted into the brain, tumors with high HSPA5 grew larger and killed animals sooner; turning down HSPA5, or interrupting its support of YAP/TAZ, slowed growth and extended survival. Finally, in matched human samples taken from the same patients before and after recurrence, recurrent, more mesenchymal tumors showed higher levels of HSPA5, YAP, TAZ, and mesenchymal markers than the original proneural‑like tumors.
What this means for future brain cancer treatment
In plain terms, this work suggests that glioblastoma cells exploit a stress‑response helper, HSPA5, to shield crucial growth switches (YAP and TAZ) from being broken down. That protection helps tumors shift into and maintain a more aggressive, treatment‑resistant identity. Because small‑molecule inhibitors of HSPA5 already exist and tumor cells may depend on this chaperone more than normal cells do, the HSPA5–YAP/TAZ axis offers an appealing new target. Therapies that weaken this protective shield could make glioblastoma cells less adaptable, less invasive, and more vulnerable to standard treatments such as chemotherapy and radiotherapy.
Citation: Gui, S., Yu, W., Song, Z. et al. HSPA5 promotes YAP/TAZ stability independently of the Hippo pathway and induces proneural-to-mesenchymal transition in glioblastoma. Cell Death Dis 17, 208 (2026). https://doi.org/10.1038/s41419-026-08428-3
Keywords: glioblastoma, HSPA5, YAP/TAZ, tumor plasticity, mesenchymal transition