Clear Sky Science · en
Loss of MLKL impairs abdominal aortic aneurysm development by attenuating smooth muscle cell necroptosis
Why this matters for your heart and arteries
Abdominal aortic aneurysms are silent bulges in the body’s main artery that can suddenly rupture and be fatal. Today, the only real protection is surgery once the vessel is already dangerously enlarged. This study explores a very different angle: instead of waiting for the artery to fail, can we protect the vessel wall by stopping a specific, highly inflammatory form of cell death in its muscle layer?
When a key artery quietly weakens
The abdominal aorta carries blood from the heart to the lower body. In some people, its wall slowly stretches and forms an aneurysm, often without symptoms until it is large or ruptures. Scientists know that the smooth muscle cells in the vessel wall die off and that chronic inflammation erodes the supporting tissue, but the exact type of cell death involved has been unclear. The authors focus on a process called necroptosis—a “messy” form of programmed cell death driven by a protein named MLKL that bursts cells open and spills their contents, stirring up inflammation.
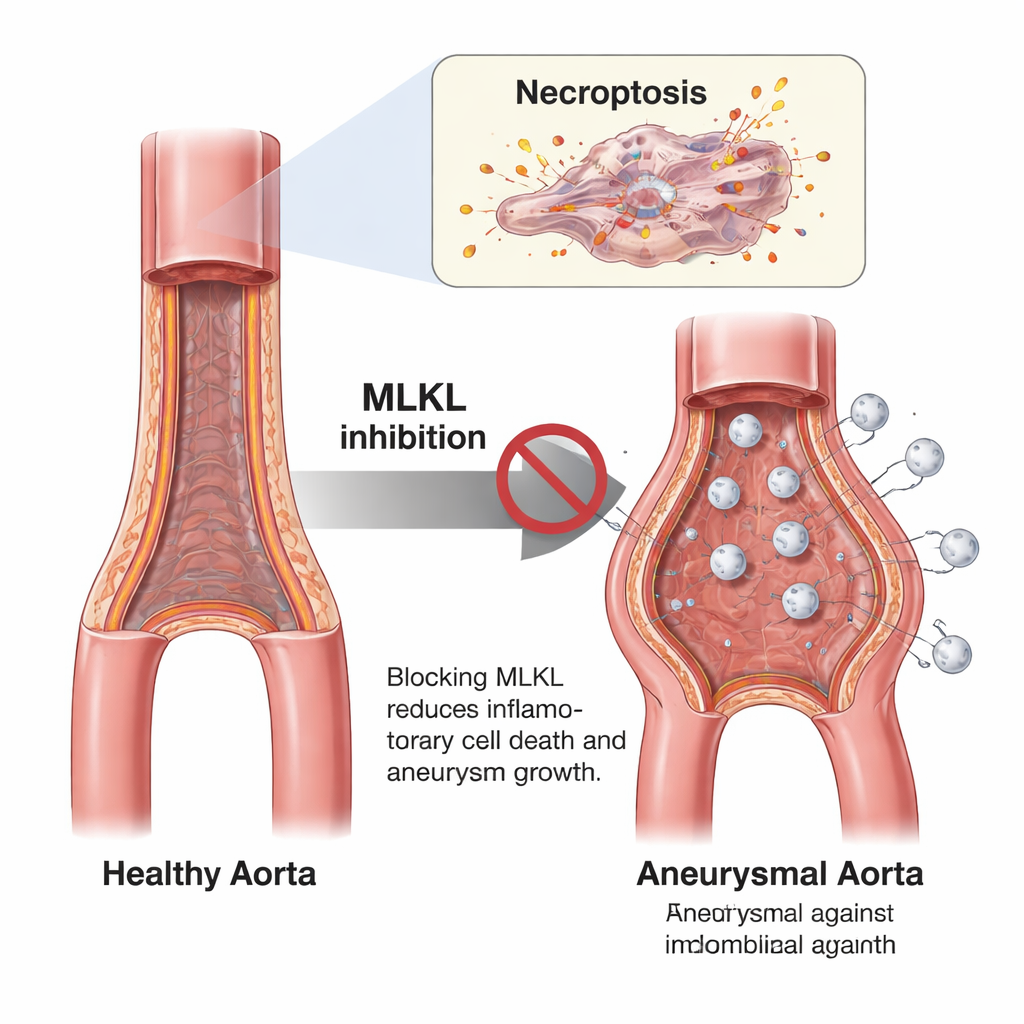
Blocking a death switch in mice protects the aorta
To test how important necroptosis is, the team used mice with genetic changes that disable this pathway: some lacked MLKL entirely, some had a mutant version of MLKL that cannot be activated, and others carried a version of an upstream protein, RIPK1, that cannot trigger its usual signals. They then induced aneurysm-like damage in the lower aorta using an elastase solution that weakens the vessel wall. Compared with normal mice, those unable to execute necroptosis developed far smaller aneurysms, kept a more normal aortic shape on ultrasound, and showed less damage to collagen and elastic fibers—the “rebar and rubber bands” that give arteries strength and flexibility.
Fewer inflammatory cells invade the vessel wall
Microscope images revealed why the protected mice fared better. In normal animals, the injured aorta quickly filled with neutrophils and macrophages—frontline white blood cells that release enzymes and oxidants, further breaking down the wall. In mice lacking functional MLKL, many fewer of these cells entered the aortic wall, even though their numbers in the bloodstream were unchanged. At the same time, the smooth muscle cell layer was better preserved. Gene-activity profiling of the aorta confirmed this picture: in ordinary mice, genes linked to inflammation, immune-cell recruitment, and blood clot breakdown were strongly switched on, while in necroptosis-deficient animals these inflammatory and fibrinolysis-related programs were markedly dampened.
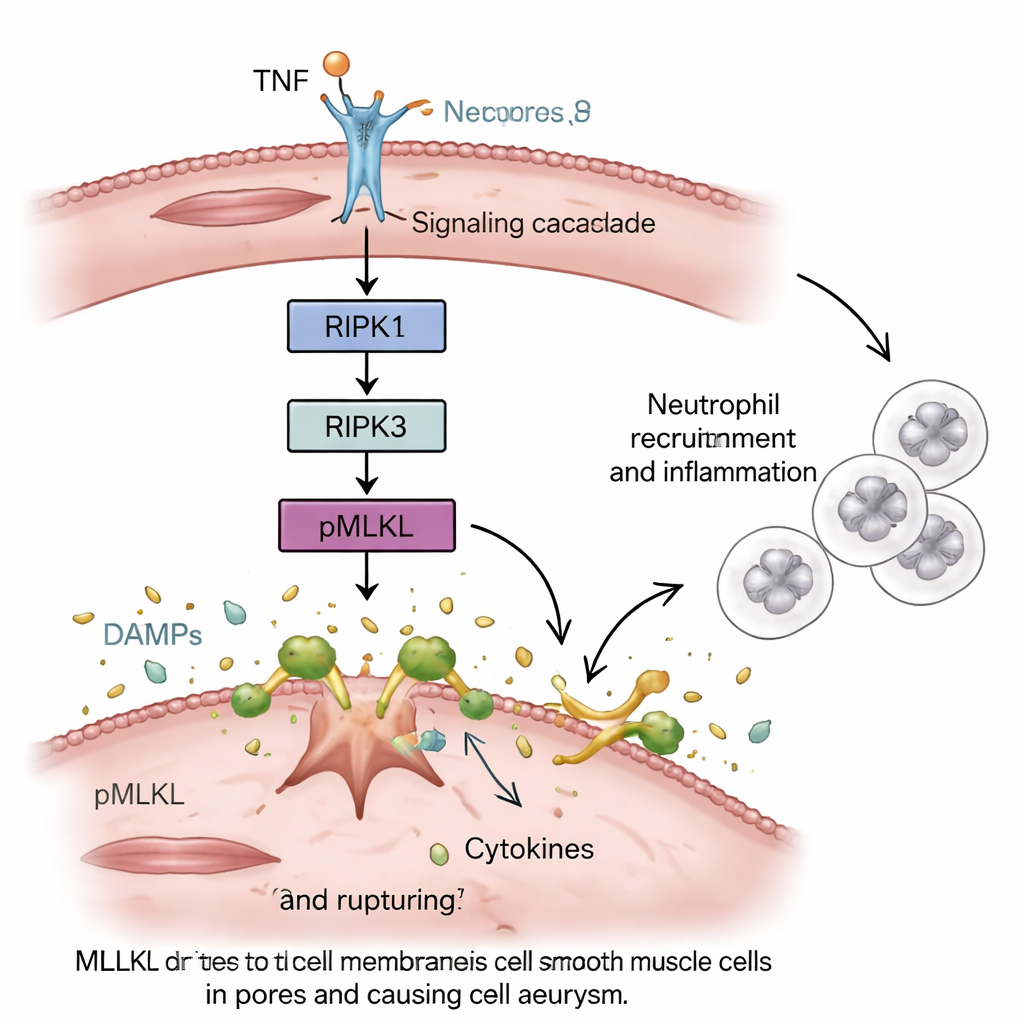
Smooth muscle cells turn from protectors into instigators
Next, the researchers asked where necroptosis mattered most. By swapping bone marrow between normal and mutant mice, they could separate the effects in blood cells from those in the vessel wall. The clearest protection occurred when the aortic smooth muscle cells, not the immune cells, lacked MLKL. In cell-culture experiments with human and mouse smooth muscle cells, forcing MLKL-driven necroptosis caused the cells to burst and release danger signals and inflammatory molecules. These secretions powerfully attracted and activated neutrophil-like cells, which then migrated toward the dying muscle cells. When an MLKL-blocking compound was added, smooth muscle cell death and the resulting neutrophil activation were both reduced.
A potential new way to keep aneurysms from growing
Putting these results together, the study suggests that when smooth muscle cells in the aorta die by necroptosis, they do not simply disappear; they actively call in immune cells and ignite a cycle of inflammation and tissue breakdown that drives aneurysm growth. Removing or disabling MLKL in mice interrupts this cycle, preserving the vessel wall and limiting aneurysm formation. For patients, this raises the possibility that future drugs targeting MLKL could slow or prevent abdominal aortic aneurysms, offering a medical alternative to “watchful waiting” and late-stage surgery. While such treatments are not yet available, this work maps out a concrete molecular target for protecting one of the body’s most vital blood vessels.
Citation: Nemade, H., Mehrkens, D., Lottermoser, H.S. et al. Loss of MLKL impairs abdominal aortic aneurysm development by attenuating smooth muscle cell necroptosis. Cell Death Dis 17, 217 (2026). https://doi.org/10.1038/s41419-026-08427-4
Keywords: abdominal aortic aneurysm, necroptosis, smooth muscle cells, MLKL inhibition, vascular inflammation