Clear Sky Science · en
TRPA1 promotes overactive bladder progression by activating the NLRP3 inflammasome and driving pyroptosis
Why an irritable bladder matters
Needing to rush to the bathroom, getting up several times a night, or worrying about leaks are common problems grouped under “overactive bladder.” This condition affects about one in five people worldwide and can quietly erode sleep, work, travel, and social life. Yet most current medicines offer only modest relief and can cause bothersome side effects. This study explores a newly uncovered chain of events inside the bladder that may explain why it becomes so jumpy—and points to fresh treatment options that go beyond simply relaxing the muscles.
A hidden trigger in the bladder wall
Deep in the bladder lining, cells carry a sensor protein called TRPA1 that reacts to physical and chemical stress. The researchers examined urine cells from women with overactive bladder and bladder tissue from animal models that mimic both short-term and long-lasting disease. They found that TRPA1 levels were consistently higher in overactive bladder than in healthy controls, and the more TRPA1 a person’s cells made, the worse their symptom scores. In animals, raised TRPA1 went hand in hand with a touchy bladder that contracted more often, needed less urine to trigger emptying, and showed swelling and structural changes in its wall.
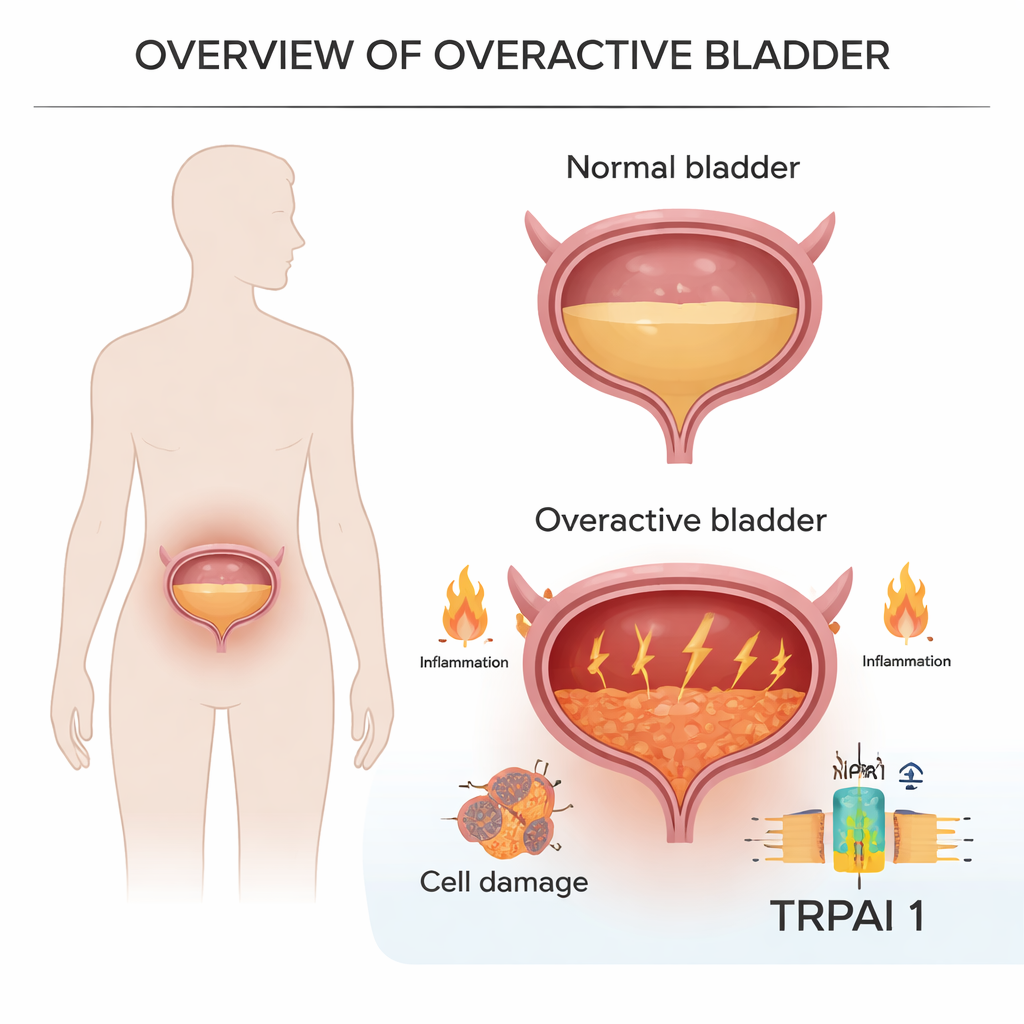
Inflammation links nerves, injury, and urgency
The team then looked at which genes were switched on in overactive bladders. Many were tied to inflammation—the body’s chemical alarm system—including well-known messengers such as IL-6 and TNF. These changes were seen not only in their own mouse models but also in public datasets from people with bladder problems. In both mice and rats with overactive bladder, inflammatory genes were turned up sharply in the bladder tissue, suggesting that the condition is not just a mechanical muscle issue but also an inflammatory disease in which the lining cells are stressed, damaged, and sending constant distress signals.
A fiery form of cell death
One key pathway stood out: a molecular machine called the NLRP3 inflammasome. When activated, NLRP3 switches on an enzyme (Caspase-1) that chops another protein, Gasdermin D, into a form that punches holes in cell membranes. This process, known as pyroptosis, causes cells to swell and burst, spilling inflammatory molecules like IL-1β and IL-18 into surrounding tissue. The study showed that in overactive bladder models, NLRP3, active Caspase-1, and cleaved Gasdermin D were all elevated, especially in the bladder’s inner lining. In simple terms, stressed bladder cells were not just malfunctioning—they were dying explosively and fueling a cycle of irritation and overactivity.
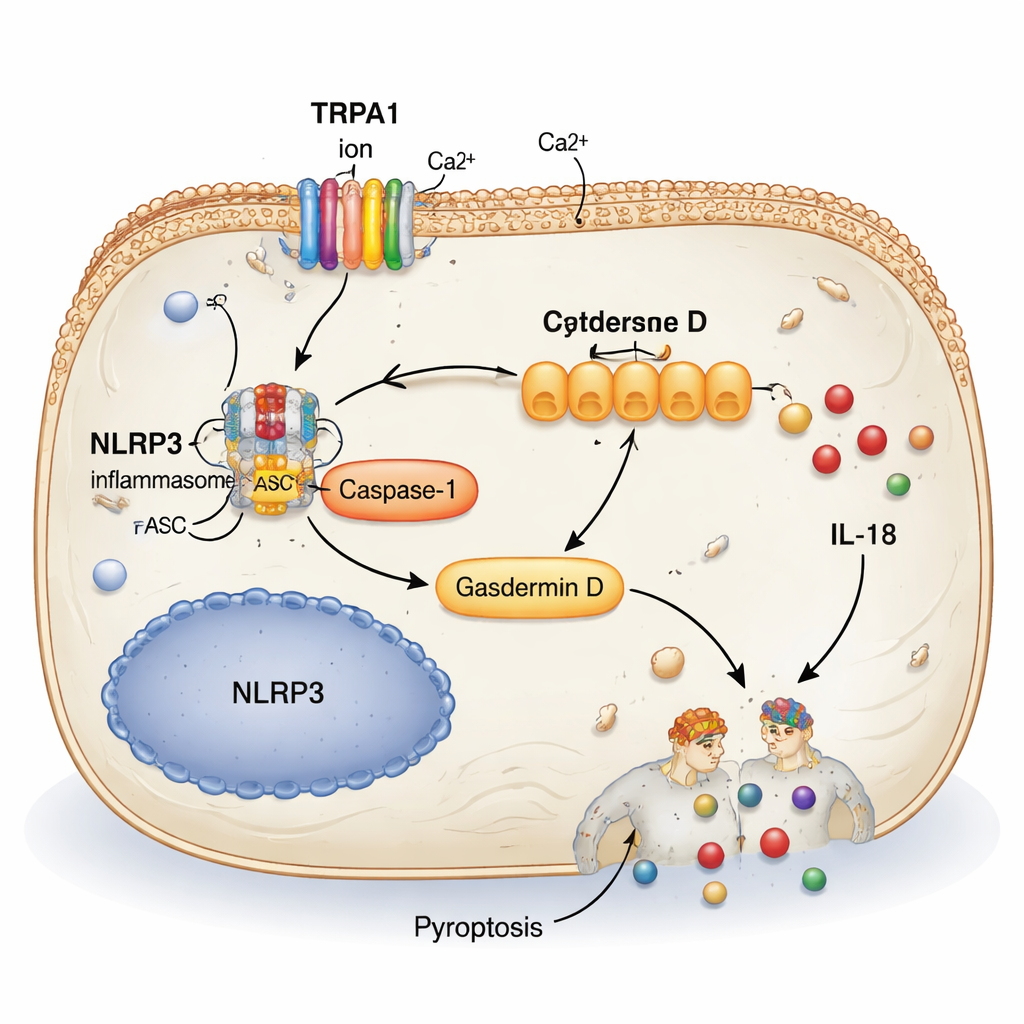
Switching off the chain reaction
To test whether TRPA1 was driving this damaging cascade, the researchers used a drug called HC-030031, which blocks TRPA1 channels. In bladder cells exposed to a harmful chemical, the blocker reduced TRPA1 levels, dampened inflammatory genes, and improved cell survival. In mice with overactive bladder, the same blocker eased inflammation, reduced markers of pyroptosis, and markedly improved bladder behavior—fewer urine spots, longer intervals between voids, and better bladder flexibility. When the team artificially boosted NLRP3 levels in the bladder, these benefits largely disappeared, showing that TRPA1’s harmful effects travel mainly through the NLRP3–pyroptosis route. They also discovered that TRPA1 boosts NLRP3 production by working with two DNA-binding proteins, MAZ and SMAD3, which help turn the NLRP3 gene on.
What this means for people with bladder urgency
Put simply, the study proposes that an overactive bladder is driven in part by an oversensitive sensor (TRPA1) on bladder-lining cells that cranks up an inflammatory death program (NLRP3 inflammasome and pyroptosis). This makes the bladder wall leaky, inflamed, and overly responsive, so it signals “go now” even when only a small amount of urine is present. By blocking TRPA1 or interrupting the NLRP3 pathway, it may be possible to calm this internal alarm system, protect the bladder lining, and reduce urgency and frequency. While the work is still at the experimental stage, it opens the door to a new class of treatments that target the root inflammatory circuitry of overactive bladder rather than just its muscle contractions.
Citation: Rao, Y., Wang, Y., Gao, J. et al. TRPA1 promotes overactive bladder progression by activating the NLRP3 inflammasome and driving pyroptosis. Cell Death Dis 17, 226 (2026). https://doi.org/10.1038/s41419-026-08426-5
Keywords: overactive bladder, TRPA1, inflammation, NLRP3 inflammasome, pyroptosis