Clear Sky Science · en
p16-mediated G0/G1 cell cycle arrest leads to SASP and fibrosis in Fuchs endothelial corneal dystrophy
Why this eye disease matters
As people age, one of the lesser-known threats to vision is a condition called Fuchs endothelial corneal dystrophy (FECD), which slowly clouds the normally clear front window of the eye. This paper explores why certain cells on the inner surface of the cornea wear out and scar over time, and why women are affected more often. Understanding these hidden changes could open the door to medicines that delay or even prevent the need for corneal transplants.
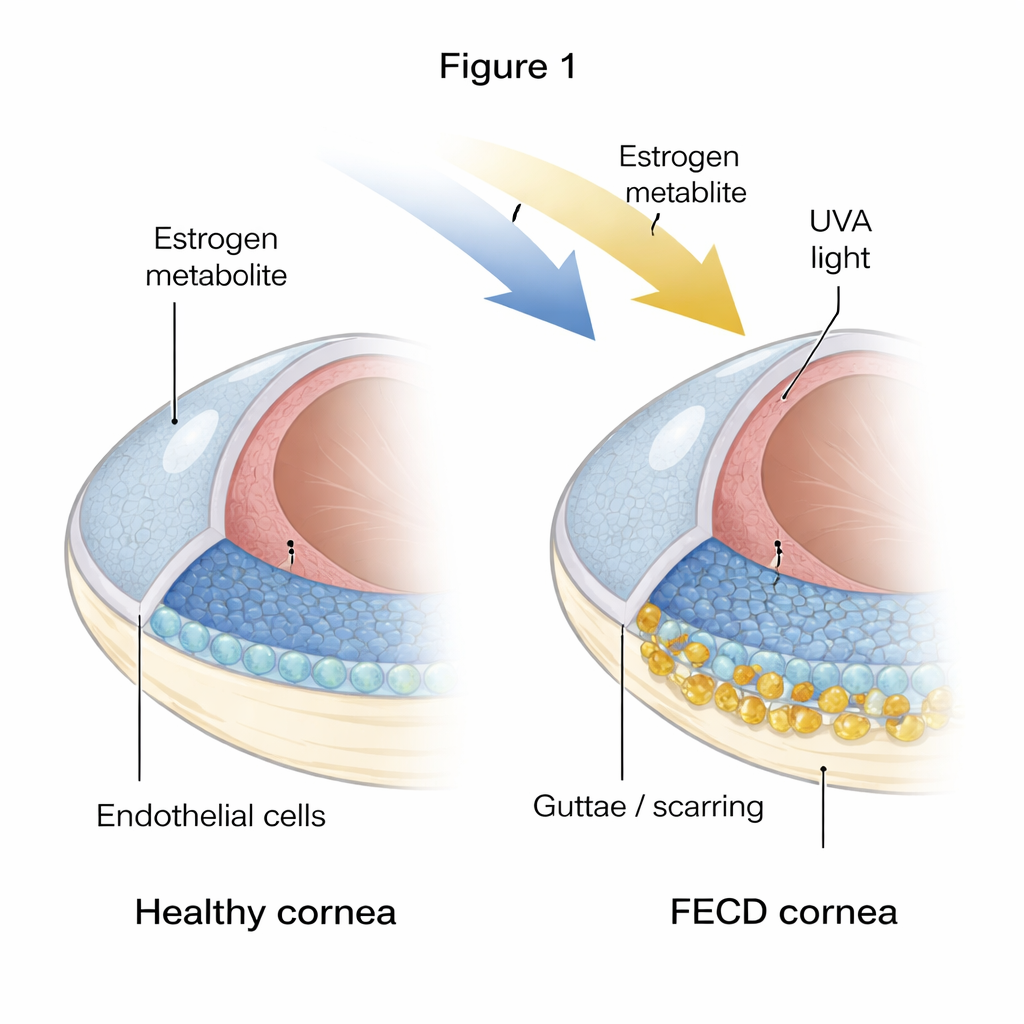
The cornea’s fragile inner lining
The cornea must stay precisely dehydrated to remain transparent, a job handled by a single layer of hexagon-shaped cells called corneal endothelial cells. In FECD, many of these cells die and the layer becomes patchy and irregular. At the same time, hard bumps and thickened material, known as guttae and excess extracellular matrix, build up on the inner surface. The authors compared healthy donor corneas to corneas from FECD patients and found strong signs that the diseased tissue is both “aged” and fibrotic: cells showed markers of senescence (a permanent shutdown state) and of fibrosis (cells behaving more like scar-forming cells and laying down extra tissue).
Chronic light and hormone stress push cells into a dead end
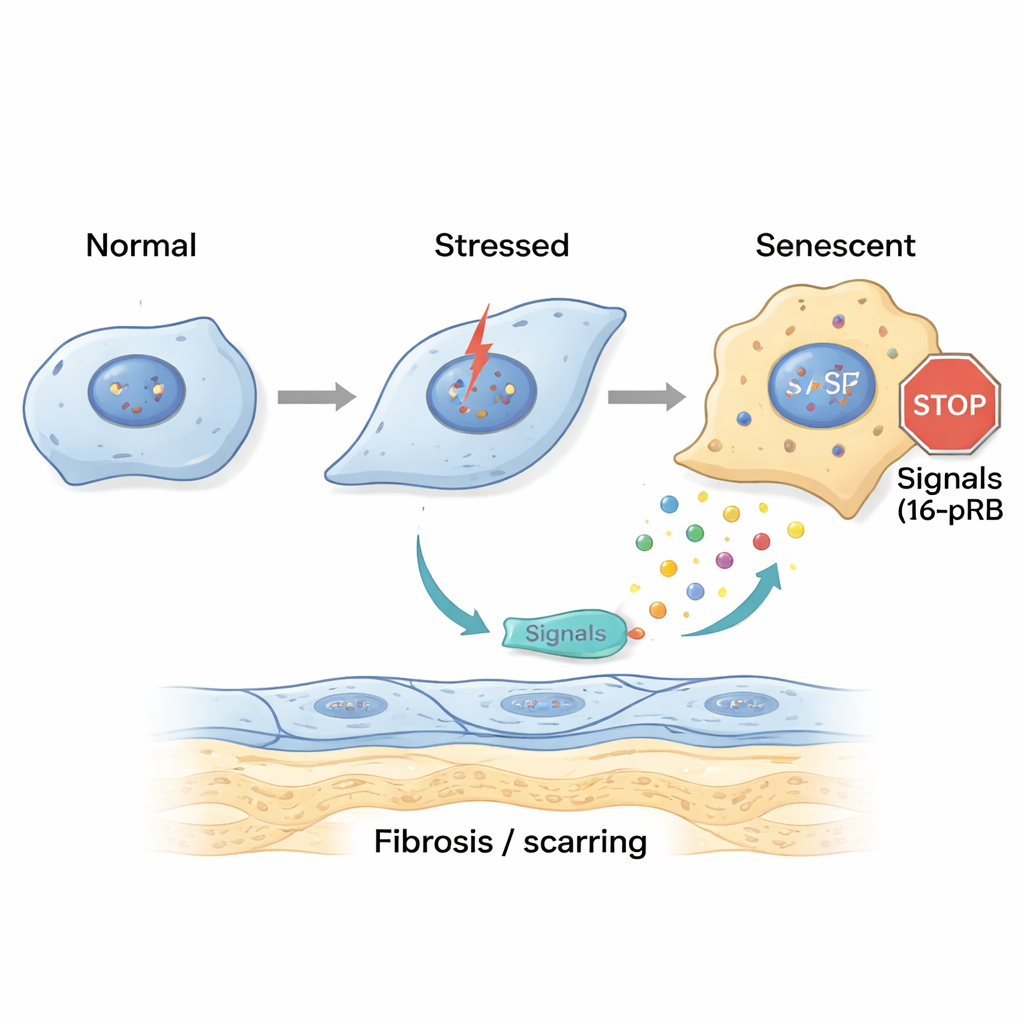
To probe how this damage starts, the researchers recreated FECD-like stress in the lab. They exposed healthy corneal endothelial cells to ultraviolet A (UVA) light and to 4-hydroxyestradiol, an oxidized estrogen byproduct linked to DNA damage. A short, single hit of stress made cells temporarily arrest in a late stage of the cell cycle and begin an early transition toward a more fibroblast-like state, but some capacity for recovery remained. In contrast, repeated, chronic exposure drove cells into a different state: they piled up in an early cell-cycle stage called G0/G1 and switched on a gene called p16, which locks the cell cycle through its partner protein pRB. These cells became large, flat, and unmistakably senescent, while also strongly expressing fibrosis markers and proteins that make up the thickened guttae in FECD.
Senescent cells send harmful signals and spread scarring
Senescent cells do not just sit quietly; they secrete a stew of signaling molecules known as the senescence-associated secretory phenotype (SASP). The team collected fluid from stressed corneal cells and applied it to fresh cells and to healthy donor corneas. The “acute” SASP caused early signs of cell shape change, while “chronic” SASP pushed many cells into senescence and increased genes tied to fibrosis and extra matrix deposition. Detailed testing showed that chronic SASP was rich in inflammatory messengers such as IL-8 and IL-17, which are known to promote cell aging, fibrotic scarring, and immune cell recruitment. When the researchers blocked the IL-17 pathway or the IL-8 receptor CXCR2, cells exposed to chronic SASP showed fewer senescence markers and less fibrosis, suggesting these signals are key drivers of disease.
Culling worn-out cells to protect vision
Because p16-positive senescent cells were so tightly linked to scarring, the authors tested whether eliminating them could protect the cornea. In a mouse model where UVA light triggers FECD-like changes, they treated animals with a senolytic drug combination, Dasatinib plus Quercetin, which selectively kills senescent cells. Compared to untreated mice, senolytic-treated mice kept a more regular endothelial cell mosaic, preserved more cells overall, and showed fewer markers of senescence, fibrosis, and excess matrix. In other words, clearing out the most damaged cells lightened the burden of harmful secretions and helped maintain a healthier corneal surface.
What this means for people with FECD
For patients, FECD currently often leads to corneal transplant when vision becomes too cloudy. This study suggests a different path: the disease may be driven by a slow accumulation of stressed, non-dividing cells that both fail to do their job and actively poison their neighbors with inflammatory and fibrotic signals. Targeting those signals with drugs against IL-17 or IL-8 pathways, or using senolytic therapies to selectively remove the worst-off cells, could one day delay scarring, preserve more of the eye’s natural tissue, and postpone or reduce the need for surgery.
Citation: Parekh, M., Adhikari, Y., Deshpande, N. et al. p16-mediated G0/G1 cell cycle arrest leads to SASP and fibrosis in Fuchs endothelial corneal dystrophy. Cell Death Dis 17, 197 (2026). https://doi.org/10.1038/s41419-026-08425-6
Keywords: Fuchs dystrophy, corneal endothelium, cellular senescence, fibrosis, senolytic therapy