Clear Sky Science · en
SLC46A1 deficiency-mediated folate restriction suppresses colorectal cancer progression through epigenetic-transcriptional reprogramming
Why vitamin B9 and gut cancer make an unexpected pair
Many people know folate, or vitamin B9, as a nutrient important for pregnancy and heart health. This study shows that folate also plays a surprising role in colon and rectal cancer. The twist is that the benefit of folate depends on a single gatekeeper protein on cancer cells, called SLC46A1, which decides how much folate actually gets inside the tumor cells. Understanding this hidden gate may help explain why past research on folate and cancer has been so confusing, and could guide safer use of folate in diet and treatment.
A nutrient gatekeeper in the gut
Cells in our intestine cannot make folate on their own; they must pull it in from their surroundings using special transport proteins. The authors show that SLC46A1 is the main folate transporter in the human colon, far more active than other known folate carriers. When they examined large public cancer datasets and tissue samples from patients, they found that SLC46A1 levels were sharply reduced in colorectal tumors compared with nearby healthy tissue. Tumors with less SLC46A1 tended to be more advanced, more likely to spread, and linked to worse patient survival, suggesting that this transporter behaves more like a brake than an accelerator in this cancer.
How losing the gate changes cancer behavior
To test what SLC46A1 actually does, the researchers dialed its levels up and down in colorectal cancer cell lines. When they lowered SLC46A1, cancer cells grew faster, moved more easily, and invaded through artificial barriers in the lab. When they increased SLC46A1 in cells that started with low levels, the cells became less invasive. Similar patterns appeared in mice: tumors grown from SLC46A1-deficient cells were larger and produced more lung metastases. At the same time, tumors and cultured cells with less SLC46A1 contained less folate inside, confirming that this protein really functions as a key entry point for the vitamin.
From vitamin shortage to scrambled gene control
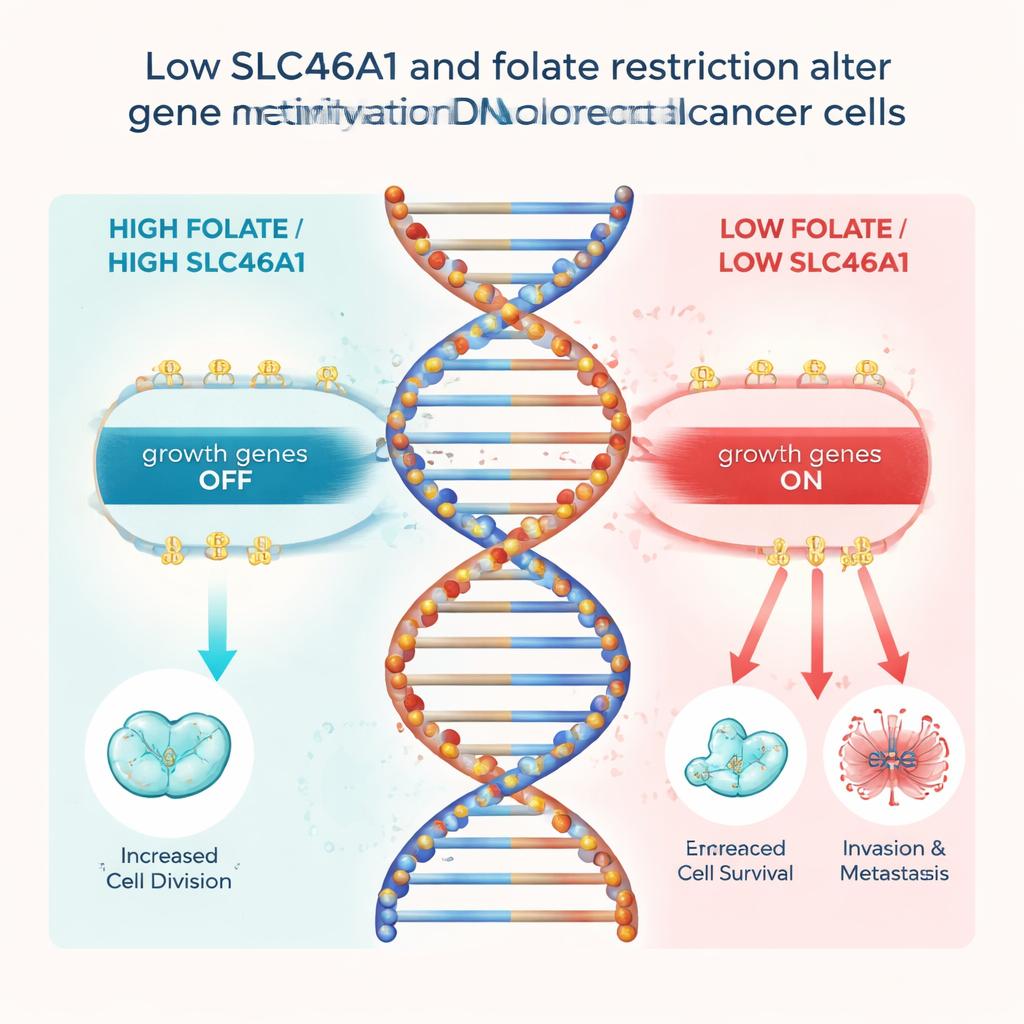
Folate does more than help make DNA building blocks; it also feeds a chemical system that places tiny methyl “tags” on DNA, which help switch genes on or off. The team showed that when SLC46A1 is lost and folate inside the cell drops, the overall ability to place these tags declines. This, in turn, selectively removes methyl tags from the control region of a gene called FOS, a well-known driver of cell growth. Without those tags, FOS switches on and activates several “growth and spread” genes, including CCND1, BCL2 and PLAU, which help cancer cells divide, avoid death, and break through surrounding tissue. Blocking FOS or PLAU largely erased the extra growth and invasiveness caused by losing SLC46A1, tying this chain of events together.
When folate helps only if the door is open
The study also clarifies when folate itself is good or bad for tumors. In cell cultures grown in folate-poor conditions, adding back folate at levels similar to those found in human blood slowed cancer cell growth and movement—but only if SLC46A1 was present to bring folate inside. In mouse tumors, direct injections of folate into the tumor mass shrank growth and reduced signs of cell division, again only when the cancer cells still made SLC46A1. Tumors lacking the transporter barely responded. In patient samples, the researchers saw an inverse pattern: tumors with high SLC46A1 had lower folate in the surrounding fluid, as if they were efficiently soaking it up, and they showed lower levels of FOS and PLAU. Tumors with low SLC46A1 left more folate unused and had higher levels of these aggressive markers.
What this means for patients and prevention
For a layperson, the main message is that folate’s impact on colorectal cancer depends on whether tumor cells still express the SLC46A1 “door” that lets folate in. When that door is present, realistic amounts of folate can support healthy DNA tagging and help keep growth-driving genes in check. When the door is missing, the tumor cannot use folate in this protective way, and instead shifts toward a more aggressive state driven by unleashed genes like FOS. This work suggests that future screening of colorectal cancers for SLC46A1 levels could help doctors predict prognosis and decide whether folate-related diets or treatments are likely to help, forming a step toward more personalized, nutrient-aware cancer care.
Citation: Zhou, Y., Liu, Y., Liu, Y. et al. SLC46A1 deficiency-mediated folate restriction suppresses colorectal cancer progression through epigenetic-transcriptional reprogramming. Cell Death Dis 17, 189 (2026). https://doi.org/10.1038/s41419-026-08423-8
Keywords: folate, colorectal cancer, epigenetics, SLC46A1, DNA methylation