Clear Sky Science · en
Lactylation of SLC26A3 in the acidic tumor microenvironment promotes malignant progression of colorectal carcinoma
Why Tumor Acidity Matters
Colorectal cancer remains one of the deadliest cancers worldwide, in part because tumors often come back, spread, or stop responding to drugs. This study explores an underappreciated culprit behind that behavior: the acidic, lactic-acid–rich environment inside tumors. By uncovering how this harsh environment destabilizes a protective membrane protein called SLC26A3, the authors reveal a new chain of events that helps cancer cells become more stem cell–like, invasive, and drug resistant—offering fresh ideas for treatment.
A Harsh Neighborhood Around Cancer Cells
Rapidly growing tumors consume large amounts of sugar and oxygen, releasing lactic acid and hydrogen ions that make their surroundings acidic. Using large patient datasets and tissue samples, the researchers show that this acidic state, tracked by a marker protein called CA9, goes hand in hand with aggressive colorectal tumors. In more advanced cancers, CA9 levels are high, while SLC26A3 levels are consistently low. SLC26A3 normally sits on the surface of colon cells and helps balance salt and bicarbonate, which in turn helps control acidity. In tumor samples, cancer cell lines, and single-cell analyses, the team finds that when acidity rises, SLC26A3 levels fall—especially in malignant, stem cell–like cancer cells. 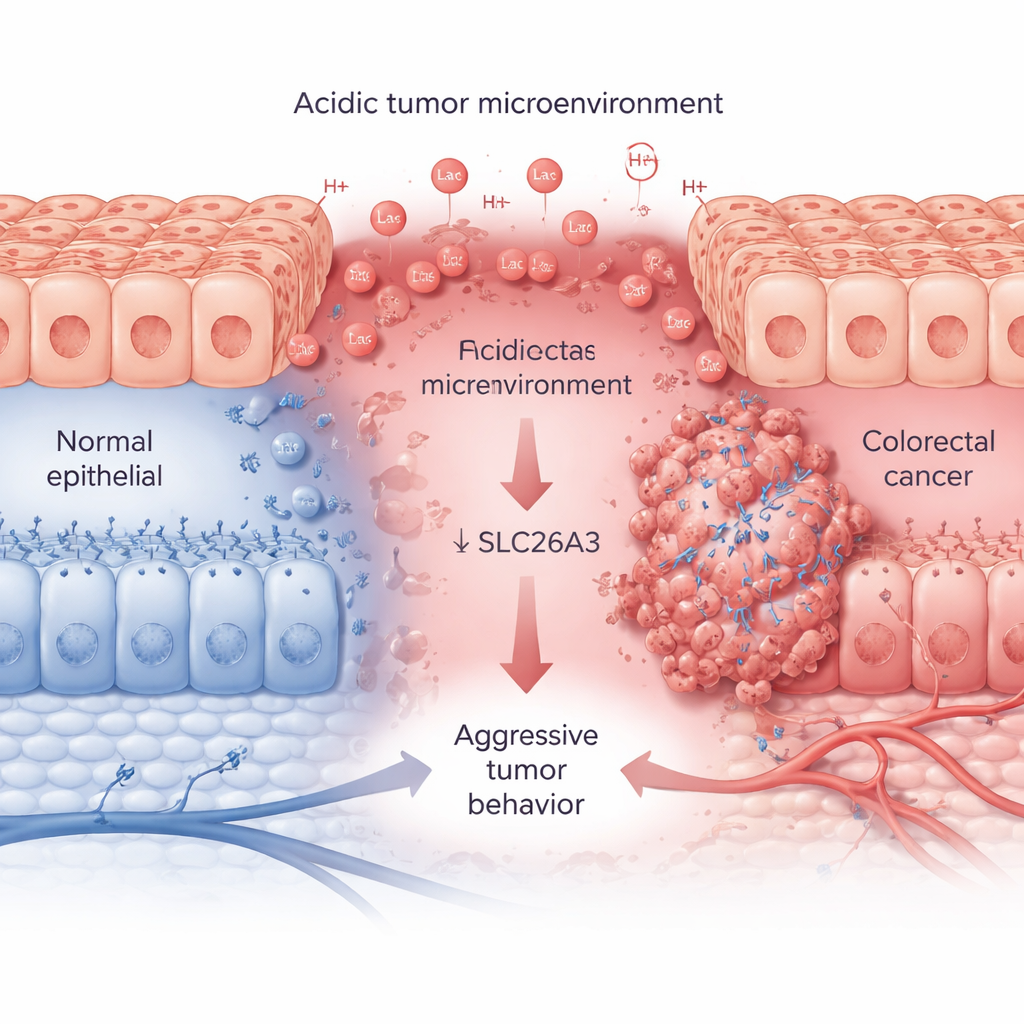
How Lactic Acid Marks a Target for Destruction
The authors then ask why SLC26A3 drops in acidic tumors. They focus on lactylation, a newly discovered chemical tag formed from lactic acid that can be added to proteins. When colorectal cancer cells are exposed to lactic acid or grown in acidic conditions, SLC26A3 becomes heavily lactylated. Detailed protein analysis pinpoints one key site, an amino acid called K536, where this tag is added. Lactylation at K536 makes SLC26A3 less stable: the protein is more likely to be tagged with ubiquitin, a cellular “dispose of this” signal, and sent to the cell’s recycling machinery. Blocking the enzyme p300, which helps add lactylation marks, partially protects SLC26A3 from breakdown. As a result, acidity and lactate set up a damaging feedback loop—more lactate leads to more lactylation, which reduces SLC26A3, further upsetting pH balance and favoring an even more acidic microenvironment.
Turning Off a Brake on Cancer Stemness and Spread
With SLC26A3 depleted, cancer cells behave more aggressively. In cell culture, colorectal cancer stem-like cells naturally express less SLC26A3 than their non–stem-like counterparts. When researchers intentionally lower SLC26A3 levels, tumor cells form more spheres (a hallmark of stemness), renew themselves more efficiently, invade through artificial membranes, and migrate faster across scratch wounds. They also become less sensitive to common chemotherapy drugs like oxaliplatin and 5‑fluorouracil. Conversely, boosting SLC26A3 reduces stemness markers and slows invasion and migration—but this benefit is blunted when extra lactate is added, underscoring how acidity can override the protective effect.
A Hidden Link to Gene Control
Beyond helping regulate acidity, SLC26A3 also serves as a scaffold that binds two RNA‑binding proteins, HuR and CUGBP1. These proteins control the lifespan of many messenger RNAs that encode cancer‑relevant proteins. When SLC26A3 is plentiful, it sequesters some HuR and CUGBP1, limiting how strongly they can stabilize or destabilize certain cancer-related messages. When SLC26A3 is lost through lactylation and degradation, HuR and CUGBP1 are freer to bind oncogenic mRNAs that promote stemness, epithelial‑mesenchymal transition (EMT), invasion, and survival. Experiments show that HuR helps stabilize mRNAs for stemness markers and cell junction proteins, while CUGBP1 can oppose HuR for some targets, reducing junction integrity and encouraging EMT—both favor tumor spread.
Rebalancing the Environment to Fight Cancer
To test whether these insights can be turned into treatment strategies, the researchers use mouse models of colorectal cancer. Neutralizing tumor acidity with sodium bicarbonate, blocking lactate transport with an MCT1 inhibitor, or overexpressing SLC26A3 all slow tumor growth. Combining SLC26A3 restoration with chemotherapy further shrinks tumors. In patient data, higher SLC26A3 levels correlate with better survival, less recurrence, fewer metastases, and better responses to chemotherapy, suggesting this protein could serve as both a biomarker and a therapeutic target. 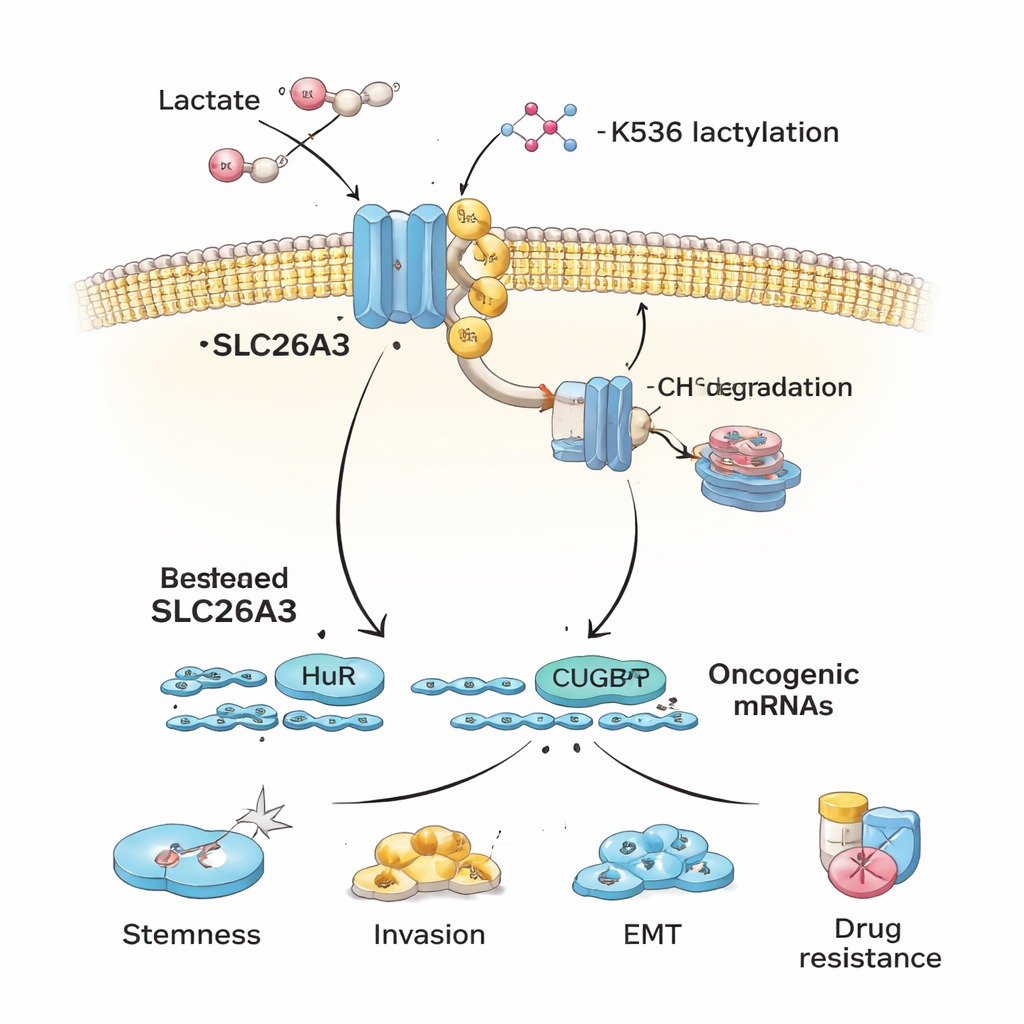
What This Means for Patients
In everyday terms, this study shows that colorectal tumors thrive in a sour, lactic-acid–rich bath that chemically “tags” and destroys a protective gatekeeper, SLC26A3. Once this gatekeeper is gone, cancer cells more easily adopt stem cell–like traits, spread, and resist treatment, aided by powerful RNA regulators inside the cell. By buffering tumor acidity, blocking lactate handling, or restoring SLC26A3, it may be possible to weaken that malignant feedback loop and make standard therapies work better. While such approaches still need rigorous clinical testing, they open a promising new angle on how managing the tumor’s chemical surroundings could help control colorectal cancer.
Citation: Chen, C., Cai, D., Liu, X. et al. Lactylation of SLC26A3 in the acidic tumor microenvironment promotes malignant progression of colorectal carcinoma. Cell Death Dis 17, 164 (2026). https://doi.org/10.1038/s41419-026-08422-9
Keywords: colorectal cancer, tumor microenvironment, lactylation, cancer stem cells, chemotherapy resistance