Clear Sky Science · en
Targeting GNG4 inhibits tumor progression and restores enzalutamide sensitivity in prostate cancer by suppressing autophagy
Why this research matters
For many men with advanced prostate cancer, powerful hormone-blocking drugs like enzalutamide work well at first but then slowly lose their punch. This study uncovers a hidden survival trick used by tumor cells and points to a specific protein, called GNG4, that helps cancers keep growing and shrug off treatment. Understanding this mechanism could lead to combination therapies that make current drugs work better and longer.
A protein switch hiding in plain sight
Prostate cancer cells are heavily driven by male hormones, so standard care for advanced disease cuts off these signals. Yet most tumors eventually become “castration resistant” and start growing again. The authors looked for genes that were much more active in tumor tissue than in nearby normal prostate from the same patients. Among several candidates, GNG4 stood out: when they reduced its levels in prostate cancer cells, tumor cell growth dropped sharply. Analyses of hundreds of patient samples confirmed that GNG4 is higher in primary prostate cancers than in normal tissue and is especially elevated in more aggressive, higher-grade tumors.
How GNG4 helps tumors thrive
GNG4 is part of a larger signaling family known as G proteins, which sit at the cell membrane and relay signals from the outside world to the cell’s interior. The team showed that turning down GNG4 in prostate cancer cell lines slowed cell division, pushed cells to pause in a checkpoint phase of the cell cycle, and increased programmed cell death. These changes also reduced the ability of cancer cells to migrate and close wounds in lab tests, and tumors formed from GNG4-deficient cells grew more slowly in mice. Together, these findings suggest GNG4 acts like a gas pedal for prostate cancer growth and spread.
Autophagy: the cancer cell’s emergency pantry
The researchers next focused on a process called autophagy, in which cells break down and recycle their own components to survive stress, such as starvation or drug treatment. In many advanced cancers, autophagy serves as an emergency pantry that helps tumor cells cope with harsh conditions. In this study, prostate cancer cells with high GNG4 activity showed more signs of active autophagy, while cells with GNG4 knocked down had fewer recycling structures and lower levels of key autophagy markers. When GNG4 was overproduced, cells tolerated enzalutamide better and underwent less cell death. Blocking autophagy with a chemical inhibitor erased much of this protection, indicating that GNG4 helps tumors endure treatment largely by ramping up this recycling system.
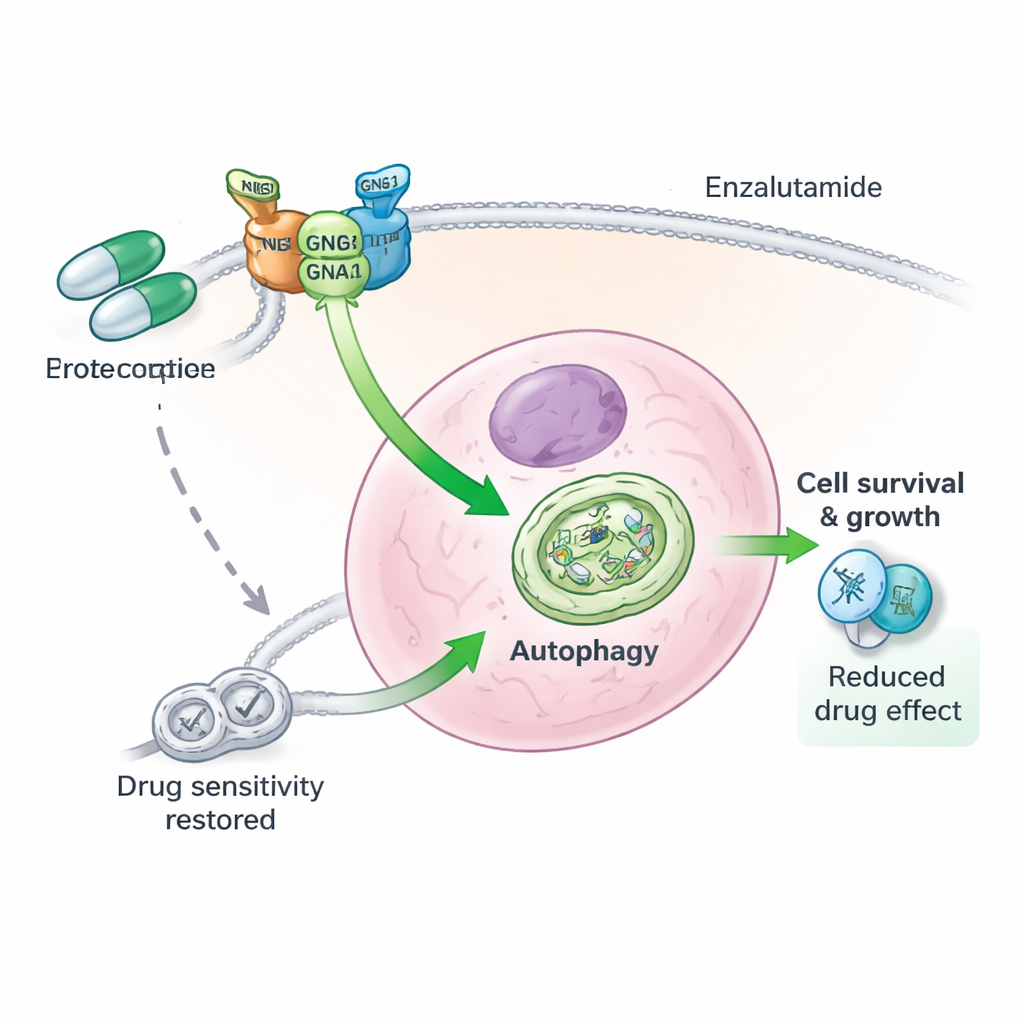
A three-part protein team behind drug resistance
Digging deeper, the authors discovered that GNG4 does not act alone. It physically associates with another G protein component, GNB1, to stabilize a third protein, GNAI3. Together, these three subunits form a functional complex that boosts autophagy inside prostate cancer cells. When GNG4 was reduced, GNAI3 protein was more quickly tagged for destruction and broken down by the cell’s waste-disposal machinery, shutting down the autophagy signal. Removing GNAI3 itself cancelled out the growth and survival advantages conferred by extra GNG4, both in cultured cells and in mouse tumors. In other words, GNG4 drives malignancy and stress resistance largely through this GNG4–GNB1–GNAI3 axis.
Making current drugs work harder
Finally, the team asked what happens when GNG4 targeting is combined with enzalutamide, a leading drug for advanced prostate cancer. In the lab, GNG4 knockdown made cancer cells markedly more sensitive to enzalutamide and reduced their ability to form colonies after treatment. In mice, tumors with lowered GNG4 shrank more when given enzalutamide than tumors with normal GNG4 levels, without obvious added toxicity. Blocking autophagy directly produced similar improvements. These results suggest that interfering with GNG4-driven autophagy can strip tumor cells of a vital coping mechanism, allowing existing hormone-blocking therapies to deliver a stronger and more durable blow.
What this means for patients
To a non-specialist, the key message is that some prostate cancers survive treatment by switching on an internal recycling system that acts like a backup generator. This study identifies GNG4 as a main switch that turns that system on, working together with partner proteins to protect cancer cells from enzalutamide. By turning this switch off—either by directly targeting GNG4 or by safely dampening autophagy—future therapies may be able to slow tumor growth and restore drug sensitivity in men whose cancers have become hard to treat.
Citation: Chen, L., Zhang, J., Hu, Y. et al. Targeting GNG4 inhibits tumor progression and restores enzalutamide sensitivity in prostate cancer by suppressing autophagy. Cell Death Dis 17, 160 (2026). https://doi.org/10.1038/s41419-026-08421-w
Keywords: prostate cancer, drug resistance, autophagy, G proteins, enzalutamide