Clear Sky Science · en
Preclinical profiling of antibody drug conjugates targeting oncofetal chondroitin sulfate
Why this research matters for future cancer treatment
Cancer drugs often work like carpet bombing: they hit fast-growing cells across the body, not just in tumors, causing harsh side effects and sometimes failing to reach all cancer cells. This study explores a smarter approach that combines the precision of antibodies with the power of chemotherapy drugs, and targets a structure that appears widely in tumors but barely in healthy tissues. For a lay reader, it offers a glimpse of how future cancer therapies might be both more effective and less damaging.
A special cancer-only “flag” on tumor tissue
The researchers focus on a molecule called oncofetal chondroitin sulfate, or ofCS. This is a sugar-like coating that appears during fetal development, disappears in most healthy adult tissues, and then reappears in many cancers. It is found not only on cancer cells themselves, but also in the surrounding tumor scaffolding and support cells. That makes it an unusually attractive target: hitting ofCS could mean striking both the tumor and its “soil” while mostly sparing normal organs. The team previously discovered an antibody fragment, named Vartumab, that recognizes ofCS on a wide variety of solid tumors with very little binding to normal tissues.
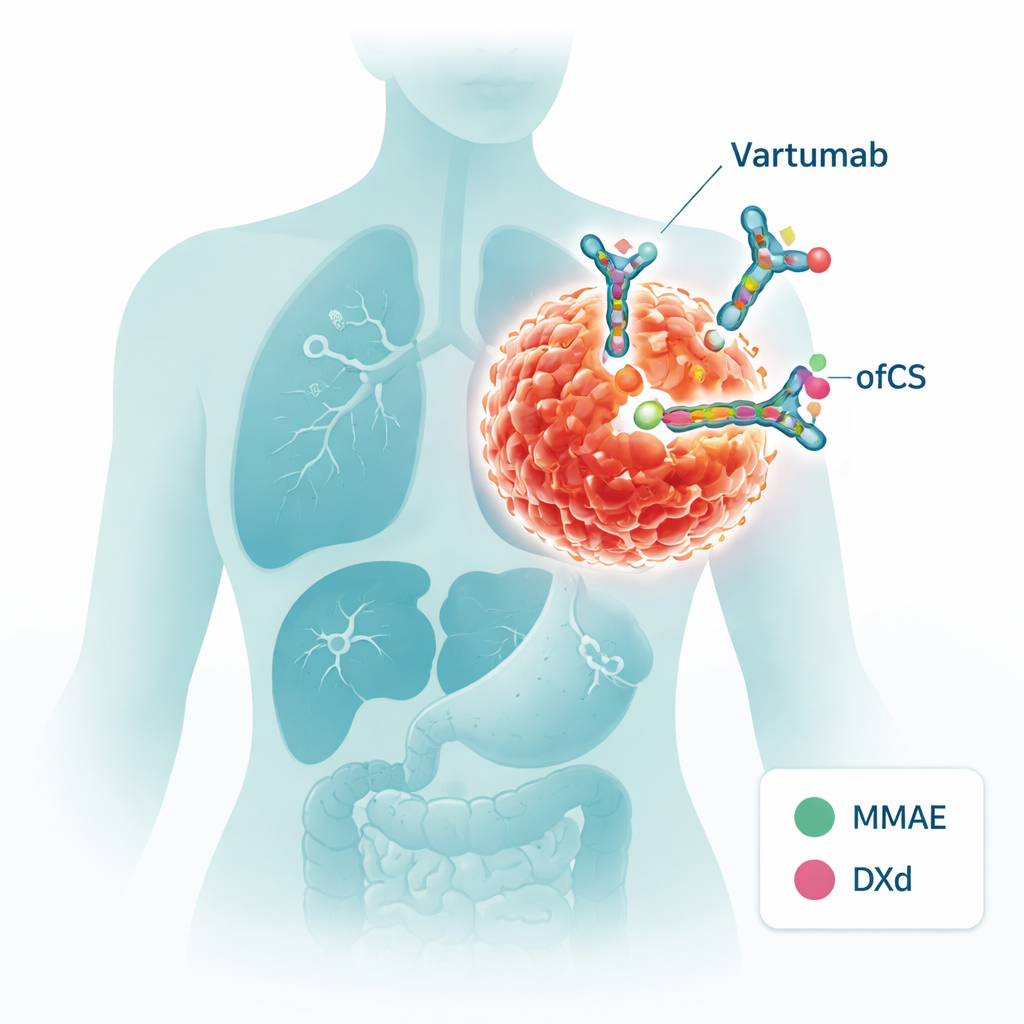
Designing a guided missile against tumors
In this work, Vartumab is turned into an antibody–drug conjugate (ADC) — essentially a guided missile carrying a toxic warhead. The scientists attached two different chemotherapy payloads, MMAE and DXd, each through a chemical “linker” that can be cut by enzymes commonly found in the tumor environment. When Vartumab binds to ofCS, these linkers can be cleaved, releasing the drug exactly where it is needed. The team carefully verified that the modified antibodies stayed stable in blood-like conditions, released the payload when exposed to tumor-related enzymes, and, crucially, still recognized ofCS just as well as the original Vartumab.
Hitting cancer cells and their neighbors
A key feature of the chosen payloads is the “bystander effect.” Once released, MMAE and DXd are able to diffuse out of the first cell they kill and enter nearby cells, even if those neighbors do not display the ofCS target. The researchers tested this by mixing ofCS-positive melanoma cells with genetically engineered ofCS-negative cells. ADCs carrying MMAE or DXd killed not only the targeted cells, but also the target-negative neighbors in a manner that depended on how many ofCS-positive cells were present. In contrast, a control ADC with a less permeable payload (MMAF) killed only the directly targeted cells and failed to produce a strong bystander effect.
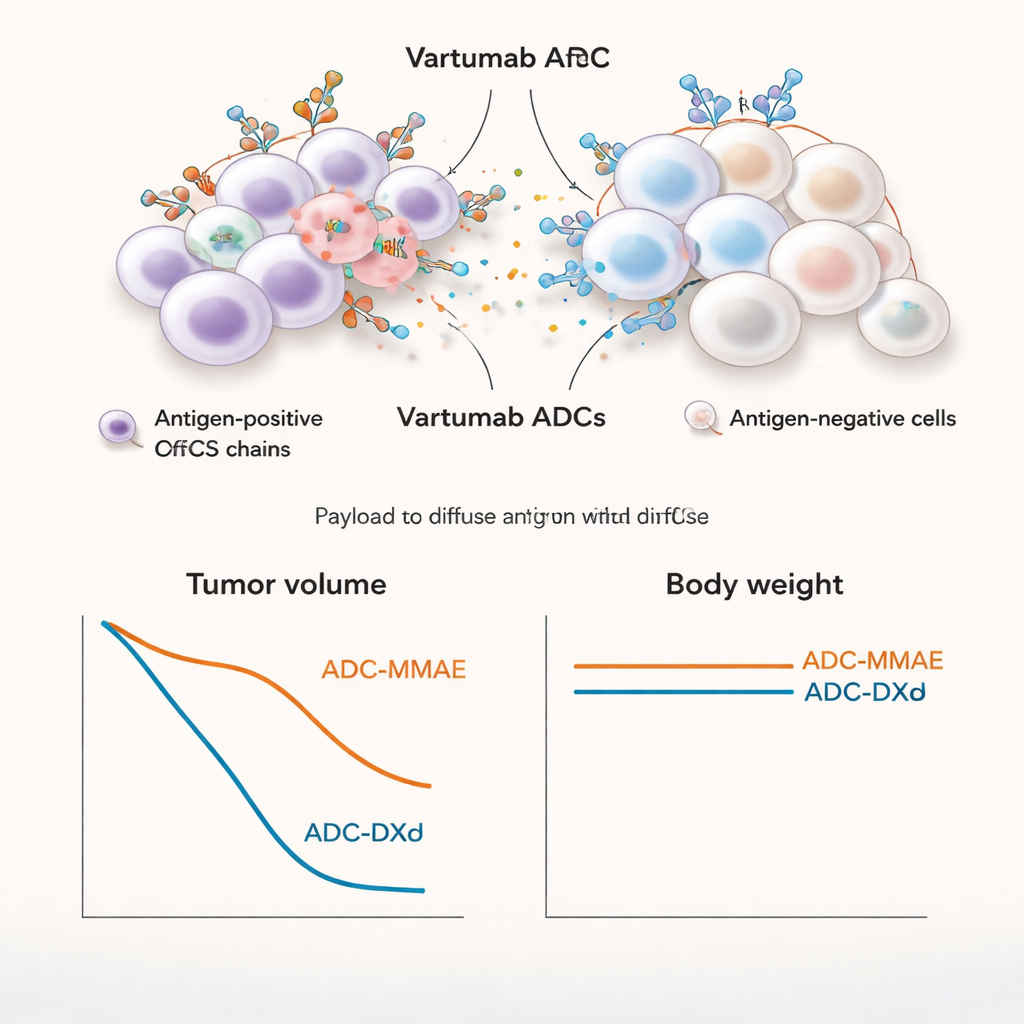
Strong tumor shrinkage with limited off-target damage
Moving into mouse models of melanoma and lung cancer, the Vartumab ADCs accumulated strongly in tumors while showing much lower levels in other organs. The MMAE version was especially potent: at relatively low doses, it completely shrank established tumors, including in a lung cancer model with lower levels of ofCS. The DXd version also produced strong tumor regression, though it generally required higher doses. When the team compared MMAE to the non-bystander MMAF in mice, only the bystander-capable MMAE ADC achieved full and lasting tumor clearance, underscoring the importance of killing both cancer cells and supportive stromal cells. In rats, repeated dosing of the MMAE ADC up to 5 mg/kg was well tolerated, with weight, blood tests, and tissue examinations showing only mild, reversible changes similar to known effects of related approved drugs.
What this could mean for patients
For non-specialists, the message is that this study refines a promising “seek-and-destroy” cancer strategy. By homing in on ofCS — a marker broadly present in tumors but scarce in normal tissues — and using payloads that can spill over to nearby cells, the Vartumab ADCs attack not just the visible tumor mass, but also its supportive environment and hidden cancer cells that might otherwise escape. The work is still preclinical, but it suggests a path toward future drugs that are more tumor-focused, work across many cancer types, and may avoid some of the most severe side effects of conventional chemotherapy.
Citation: Skafte, A., Vidal-Calvo, E.E., Choudhary, S. et al. Preclinical profiling of antibody drug conjugates targeting oncofetal chondroitin sulfate. Cell Death Dis 17, 162 (2026). https://doi.org/10.1038/s41419-026-08420-x
Keywords: antibody-drug conjugates, oncofetal chondroitin sulfate, bystander effect, targeted cancer therapy, tumor microenvironment