Clear Sky Science · en
DNA-PKcs orchestrates CTLA-4 depletion-induced senescence in cancer cells
Turning Cancer’s Growth Switch into a Stop Signal
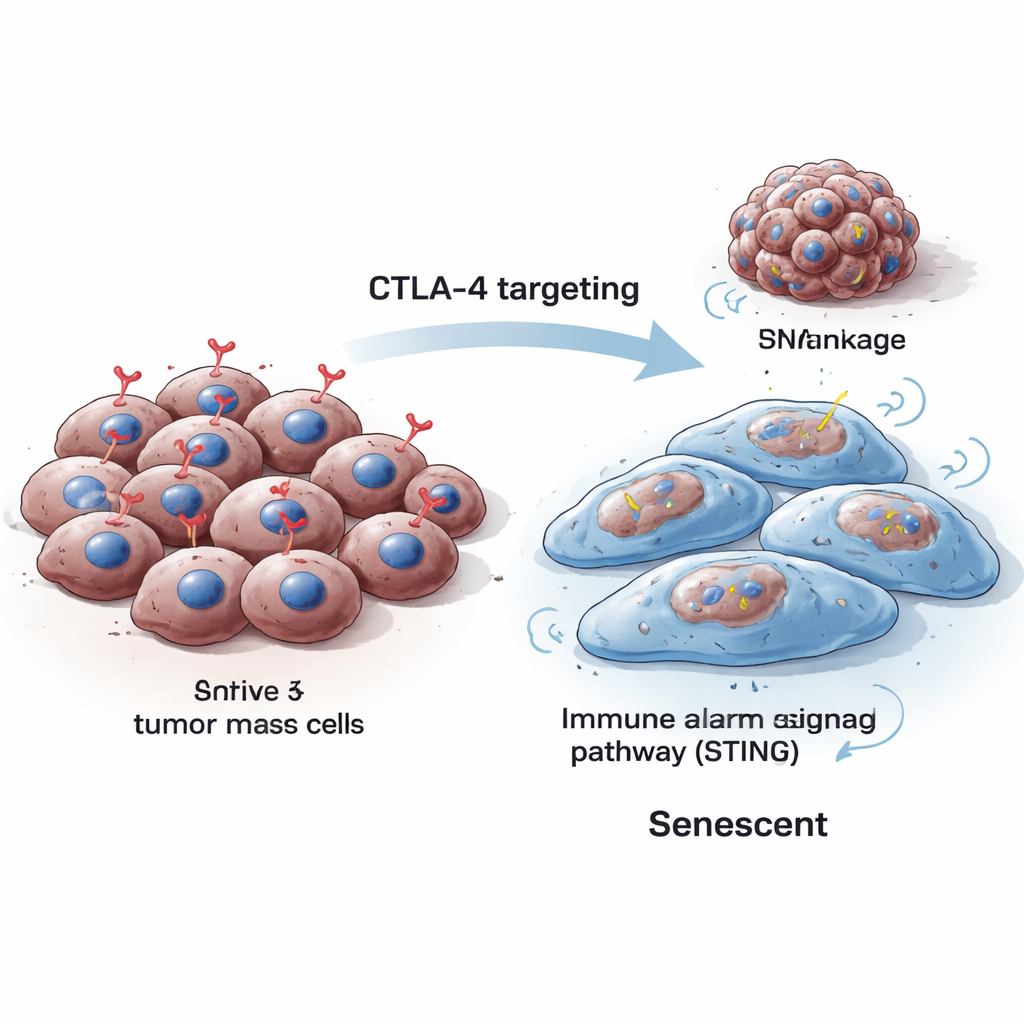
Immune-based cancer drugs often focus on a molecule called CTLA-4, found on immune cells, to help the body attack tumors. This study reveals a surprising twist: CTLA-4 is also found inside cancer cells themselves and can act like a hidden growth helper. When scientists deliberately removed this internal CTLA-4 from melanoma cells, the cancer cells stopped multiplying and slipped into a long-term “retirement” state known as cellular senescence. Understanding this unexpected role opens up new ways to slow or even shrink tumors by targeting CTLA-4 inside cancer cells, not just on immune cells.
When Cells Choose Retirement Over Runaway Growth
Cells normally divide, repair, and—when damaged beyond repair—either die or enter senescence, a permanent halt in their growth. Senescent cells typically grow larger, change shape, and produce specific markers that scientists can detect in the lab. The researchers found that when they reduced CTLA-4 inside mouse and human melanoma cells, the cells became bigger, their growth dropped sharply, and classic senescence signals rose, including enzymes detectable by special staining tests and proteins like p16 and p21 that block the cell cycle. These changes were seen both when CTLA-4 was temporarily silenced and when it was permanently knocked out using gene-editing tools.
Cracks in the Genetic Blueprint Lead to a Standstill
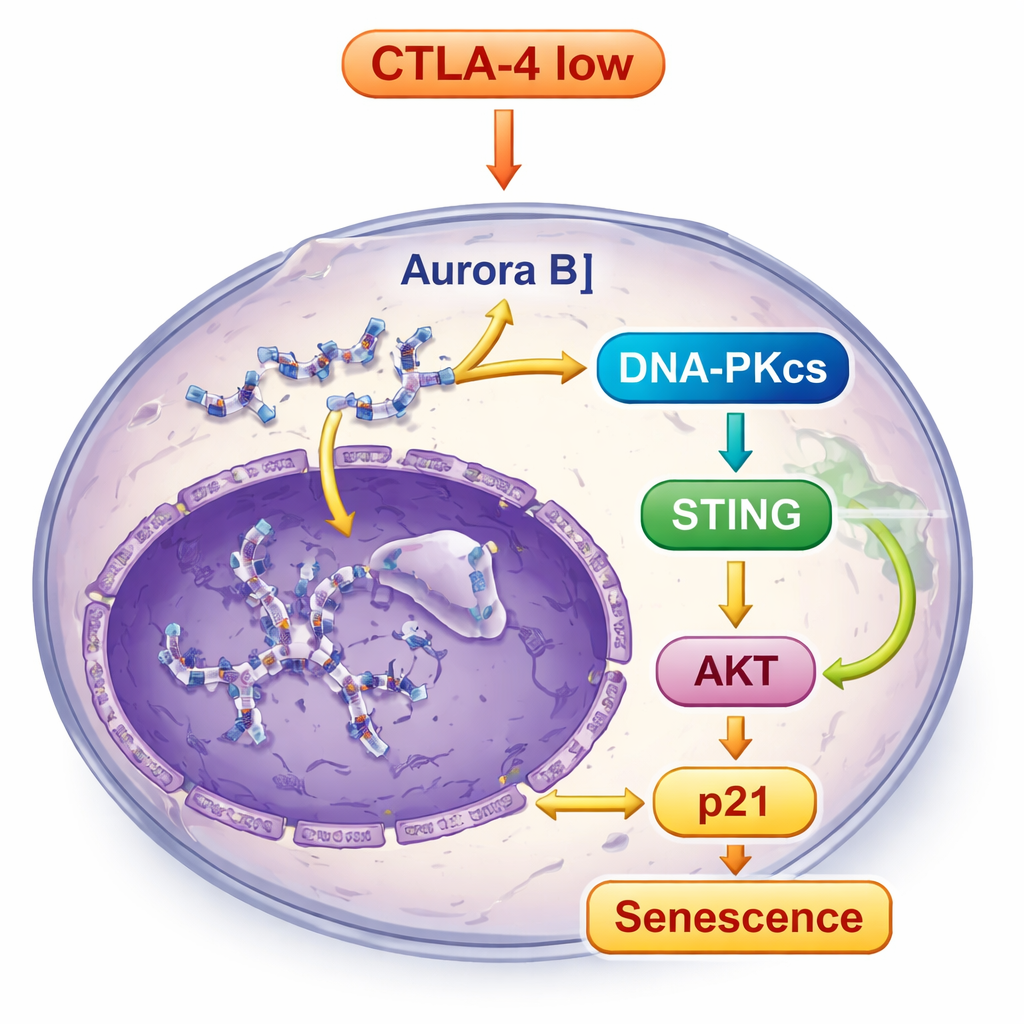
To understand why CTLA-4 loss pushes cancer cells into senescence, the team looked at how these cells handle their DNA during division. They discovered that CTLA-4 depletion reduced levels of Aurora B, a protein that helps chromosomes separate cleanly when cells divide. When Aurora B dropped, cells developed genomic instability: tiny extra DNA-containing bodies called micronuclei appeared, and DNA damage markers increased. This kind of DNA stress is known to push cells toward senescence instead of unchecked growth. Importantly, restoring Aurora B helped reduce these micronuclei, tying CTLA-4 loss directly to faulty chromosome handling and DNA damage.
DNA Damage Sets Off an Internal Alarm Network
The broken and misplaced DNA in CTLA-4-depleted cells did not go unnoticed. It activated DNA-PKcs, a DNA damage sensor, which in turn triggered the STING pathway, a built-in alarm system that responds to stray DNA inside cells. Once STING was switched on, it signaled downstream molecules, including TBK1 and IRF3, and also boosted another key growth-control route, the AKT pathway. Rather than promoting growth, this combined signaling ultimately increased p21, a strong brake on the cell cycle, reinforcing the senescent state. When the scientists blocked DNA-PKcs, STING activation and the senescence features were greatly reduced, showing that DNA-PKcs sits at the heart of this alarm relay.
From Cell Culture to Tumors in Living Animals
Laboratory dishes only tell part of the story, so the researchers tested whether CTLA-4 loss would affect real tumors in mice. They implanted melanoma cells with or without CTLA-4 into opposite sides of the same animals. Tumors lacking CTLA-4 grew more slowly, weighed less, and were smaller in volume than their CTLA-4–containing counterparts. These CTLA-4–deficient tumors also showed stronger senescence staining and higher levels of DNA damage and STING pathway activation. Analysis of patient cancer data from large public databases further supported the link: across several cancer types, CTLA-4 expression tended to be inversely related to DNA-PKcs and other DNA repair components, echoing the lab findings in human disease.
What This Means for Future Cancer Treatments
Overall, the study shows that CTLA-4 inside cancer cells helps them maintain genetic stability and continue dividing. When CTLA-4 is removed, chromosomes become unstable, DNA breaks accumulate, and an internal alarm pathway—centered on DNA-PKcs and STING—drives the cells into permanent growth arrest. For a layperson, this means that turning off CTLA-4 in tumor cells nudges cancers away from dangerous, fast-growing behavior and toward a safer, “retired” state. These insights suggest that future cancer therapies could be designed not only to unleash the immune system by blocking CTLA-4 on immune cells, but also to directly disarm tumors by targeting CTLA-4 inside cancer cells and harnessing senescence as a built-in brake on cancer growth.
Citation: Lee, JJ., Rhee, W.J., Kim, S.Y. et al. DNA-PKcs orchestrates CTLA-4 depletion-induced senescence in cancer cells. Cell Death Dis 17, 204 (2026). https://doi.org/10.1038/s41419-026-08419-4
Keywords: cellular senescence, melanoma, CTLA-4, DNA damage, STING pathway