Clear Sky Science · en
Cytoplasm-nucleus shuttling of TET2: an intrinsic brake in colorectal cancer progression
How Cancer Cells Carry Their Own Brake
Colorectal cancer is one of the most common cancers worldwide, and it often turns deadly when tumor cells learn to invade nearby tissue and spread to other organs. This study reveals that many of these cancer cells actually carry an internal “brake system” — a protein called TET2 — that can slow their advance when it moves into the cell’s nucleus. Understanding how this built‑in brake is turned on and off could open new ways to hold tumor growth in check rather than only trying to kill cancer cells outright.
Watching a Key Protein Move Inside Tumor Cells
The researchers focused on TET2, a protein that helps control which genes are switched on or off by removing certain chemical marks from DNA. In tissue samples from hundreds of colorectal cancer patients, they tracked where TET2 sits inside tumor cells. They found four main patterns: mostly in the nucleus, mostly in the cytoplasm, or strongly biased to one or the other. Patients whose tumors had more TET2 in the nucleus tended to live longer, while those whose tumors kept TET2 out in the cytoplasm fared worse. As cancers advanced to later clinical stages, nuclear TET2 became rarer, suggesting that losing this nuclear protection is part of how tumors become more aggressive.
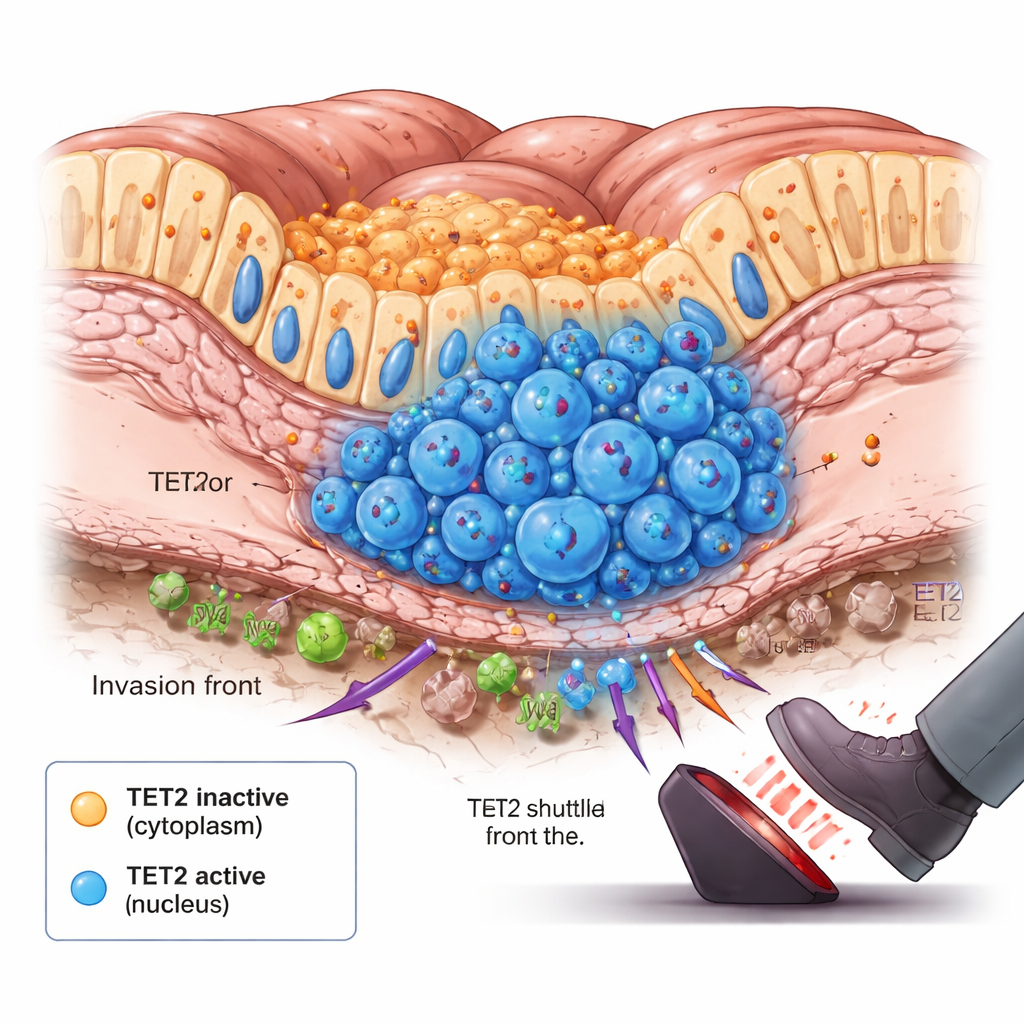
The Tumor’s Front Line and the Moment of Change
Digging deeper, the team noticed something surprising: in tumors that were just beginning to spread, there was a slight rise in cells where TET2 had moved into the nucleus. These cells clustered at the bottom of the intestinal lining, the invasion front where cancer cells first push into deeper tissue. Animal experiments and long‑term cell culture models that mimic tumor growth showed a similar pattern over time. Early on, TET2 stayed in the cytoplasm and was largely inactive. As tumors grew and inner regions became starved of oxygen and nutrients, signals inside the cells triggered TET2 to shuttle into the nucleus in specific subpopulations. Once there, TET2 started removing DNA methylation marks and slowing tumor growth, acting like an emergency brake when the cancer crossed a dangerous threshold.
When “Go” Signals Also Trigger a Brake
Colorectal tumors often rely on two powerful “go” programs: a shape‑shifting process called epithelial–mesenchymal transition (EMT), which helps cells become more mobile and invasive, and the WNT signaling pathway, which boosts growth and survival. These pathways are usually viewed as purely harmful in cancer. However, this study shows that when EMT and WNT fire strongly, they also help drive TET2 into the nucleus. Using drugs and genetic tools in cell cultures, the scientists showed that turning on EMT or WNT made more cells move TET2 into their nuclei, while blocking these pathways did the opposite. Once in the nucleus, active TET2 then dialed down EMT and WNT‑related genes, reducing cell migration and slowing proliferation. In other words, the same signals that help tumors grow also awaken an internal counterforce.
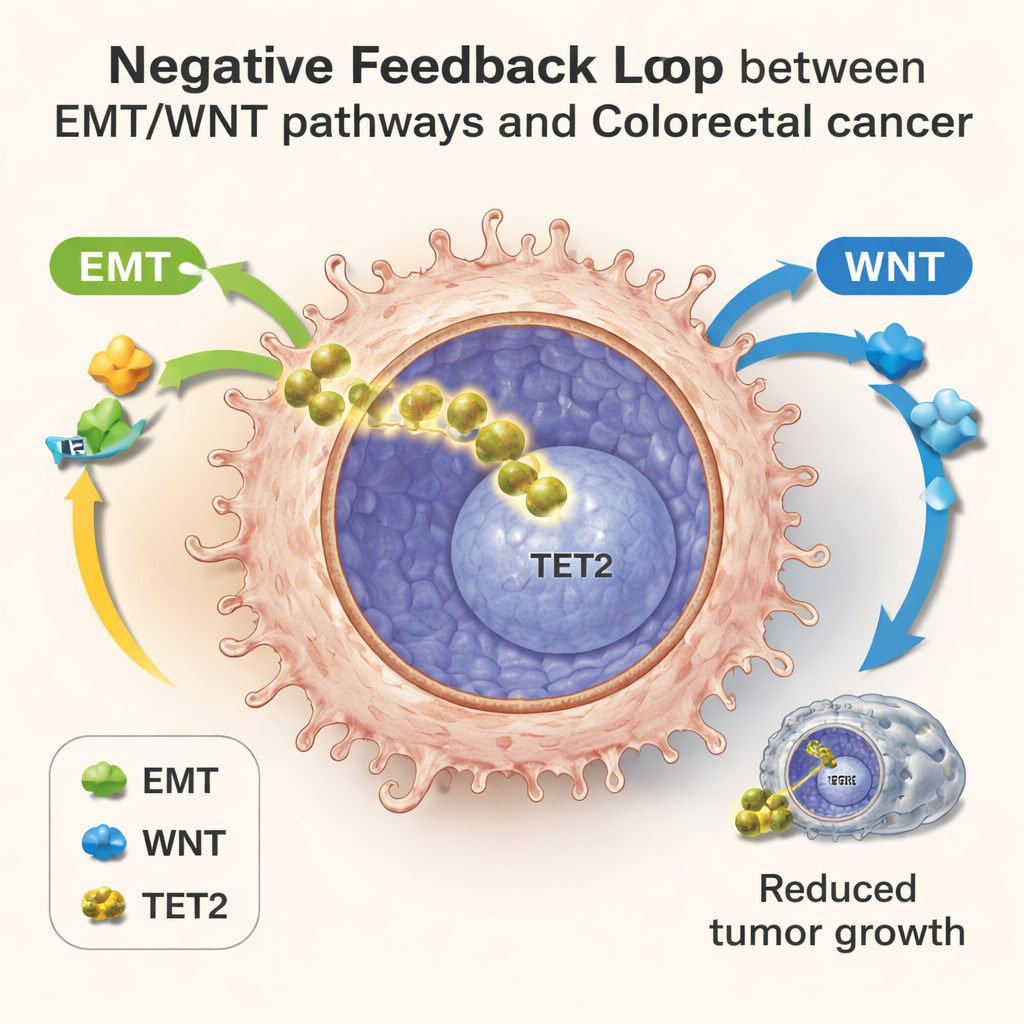
Single Cells Reveal a Hidden Feedback Loop
To map this tug‑of‑war in detail, the team used single‑cell RNA sequencing, which reads out which genes are active in thousands of individual cells at once. In lab‑grown colonies, in mouse tumors, and in samples from colorectal cancer patients, they saw cells spread along a progression timeline. Early cells showed normal energy use and weak EMT/WNT activity; later cells showed metabolic re‑wiring, strong EMT/WNT signals, and then rising TET2 target genes. At these later steps, cells with higher TET2 activity had lower levels of invasion‑related genes and were associated with better patient outcomes, even among tumors that had already started to spread. This pattern supports the idea of a negative feedback loop: metabolic stress and EMT/WNT activation push TET2 into the nucleus, and nuclear TET2 in turn restrains those same aggressive programs.
What This Means for Future Cancer Treatments
For non‑specialists, the key message is that colorectal cancer progression is not a simple “on/off” switch between good and bad behavior. Instead, there is a built‑in, delayed safety mechanism: as tumor cells become more invasive, they also activate TET2 in their nuclei, which partially reins them in. Over time, many tumors escape by losing nuclear TET2 or overwhelming its effects. By finding ways to keep TET2 in the nucleus or boost its activity — possibly in combination with drugs that modulate EMT or WNT — doctors might strengthen this natural brake and slow cancer spread. Rather than only attacking cancer cells from the outside, future therapies could work by restoring and amplifying this internal control system.
Citation: Li, C., Meng, F., He, J. et al. Cytoplasm-nucleus shuttling of TET2: an intrinsic brake in colorectal cancer progression. Cell Death Dis 17, 163 (2026). https://doi.org/10.1038/s41419-026-08418-5
Keywords: colorectal cancer, TET2, epigenetics, EMT WNT signaling, cancer progression