Clear Sky Science · en
Targeting ENO1 reprograms macrophage polarization to trigger antitumor immunity and improves the therapeutic effect of radiotherapy
Turning Tumor Shields into Targets
Cancer often survives not just by growing fast, but by quietly disarming the body’s defenses. This study explores a hidden molecule on cancer cells, called ENO1, that helps tumors evade the immune system and resist radiation therapy. By developing a new antibody drug that targets ENO1, the researchers show it is possible to drain the tumor of its protective chemicals, wake up immune cells, and make radiation work better in models of colorectal and triple-negative breast cancer. 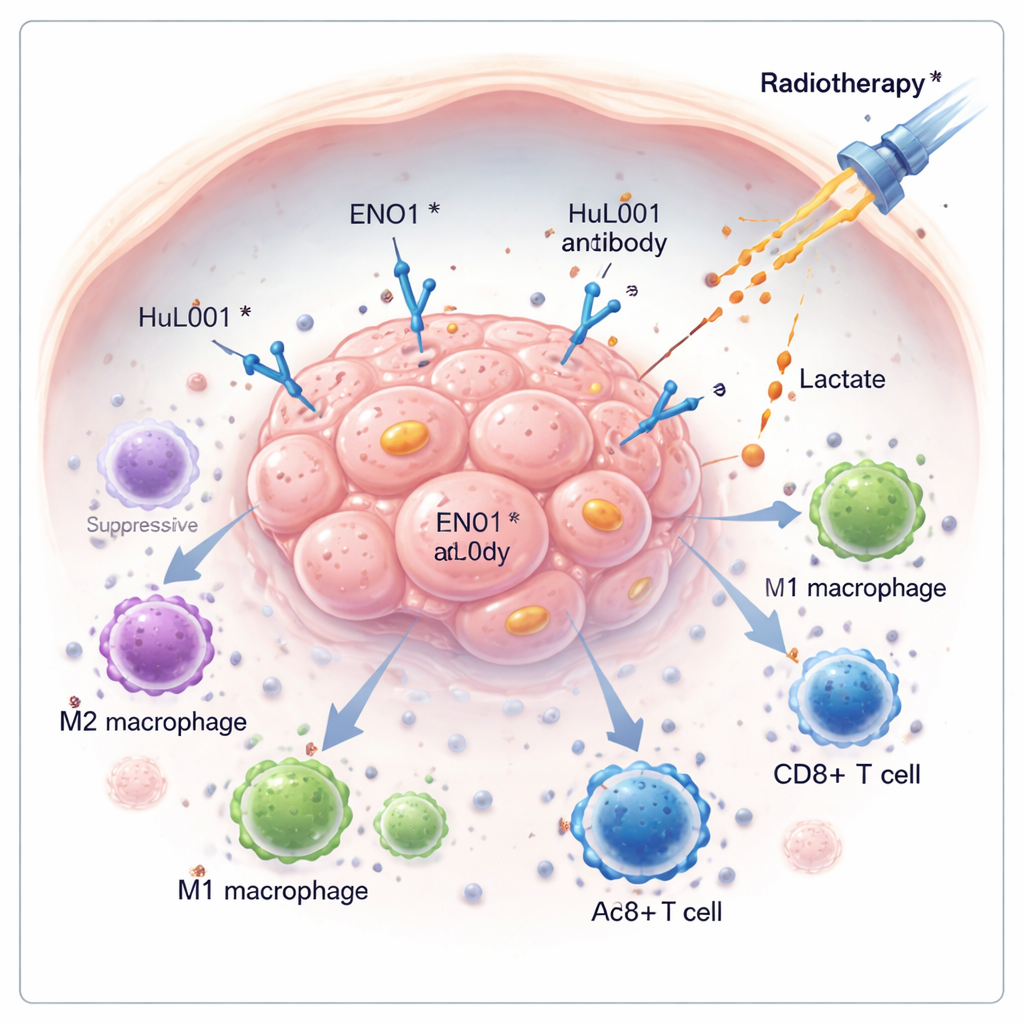
A Metabolic Trick That Silences Immunity
Many tumors rewire how they use energy, breaking down sugar in a way that produces large amounts of lactic acid even when oxygen is available. This lactic acid is pumped out of the cancer cells and builds up around the tumor, turning the surrounding area into a harsh, acidic niche that weakens immune attack. In this work, the authors focus on ENO1, an enzyme involved in sugar breakdown that can also appear on the surface of cancer cells. When located on the cell surface, ENO1 partners with another protein, a transporter called MCT4, to help export lactate—fueling an environment that favors tumor growth and suppresses immune cells.
How Radiation Can Backfire
Radiation therapy is designed to damage tumor DNA and trigger immune responses, but it also has side effects inside the tumor microenvironment. The team discovered that signals driven by a molecule called TGFβ1, which are boosted by radiation, activate an enzyme (PRMT5) that chemically modifies ENO1 and moves it to the cancer cell surface. Once there, ENO1 physically interacts with MCT4 to stabilize it and promote lactate export. Higher surface ENO1 levels were found in patients with advanced colorectal and triple-negative breast cancers and were linked to more metastasis and poorer survival, suggesting that radiation can unintentionally strengthen a lactate-driven, immune-suppressive shield around tumors.
Reprogramming Friendly and Foe Immune Cells
In the acidic halo around tumors, immune cells called macrophages are pushed into a “healer” state (often called M2) that actually helps tumors by damping inflammation and aiding repair, rather than killing cancer cells. The researchers created a humanized antibody, HuL001, that recognizes ENO1 on both mouse and human cancer cells. In cell culture and mouse models, HuL001 disrupted ENO1’s support of MCT4, reduced lactate release, and shifted macrophages away from the tumor-promoting M2 state toward an M1 state, which is more aggressive against cancer and better at gobbling up tumor cells. Importantly, HuL001 did not simply kill macrophages; instead, it re-educated them while directly increasing cancer cell death.
Making Radiation Work Harder
When HuL001 was combined with radiation therapy in mouse models of colorectal and triple-negative breast cancer, tumors shrank more, regrew more slowly, and in many animals disappeared entirely. 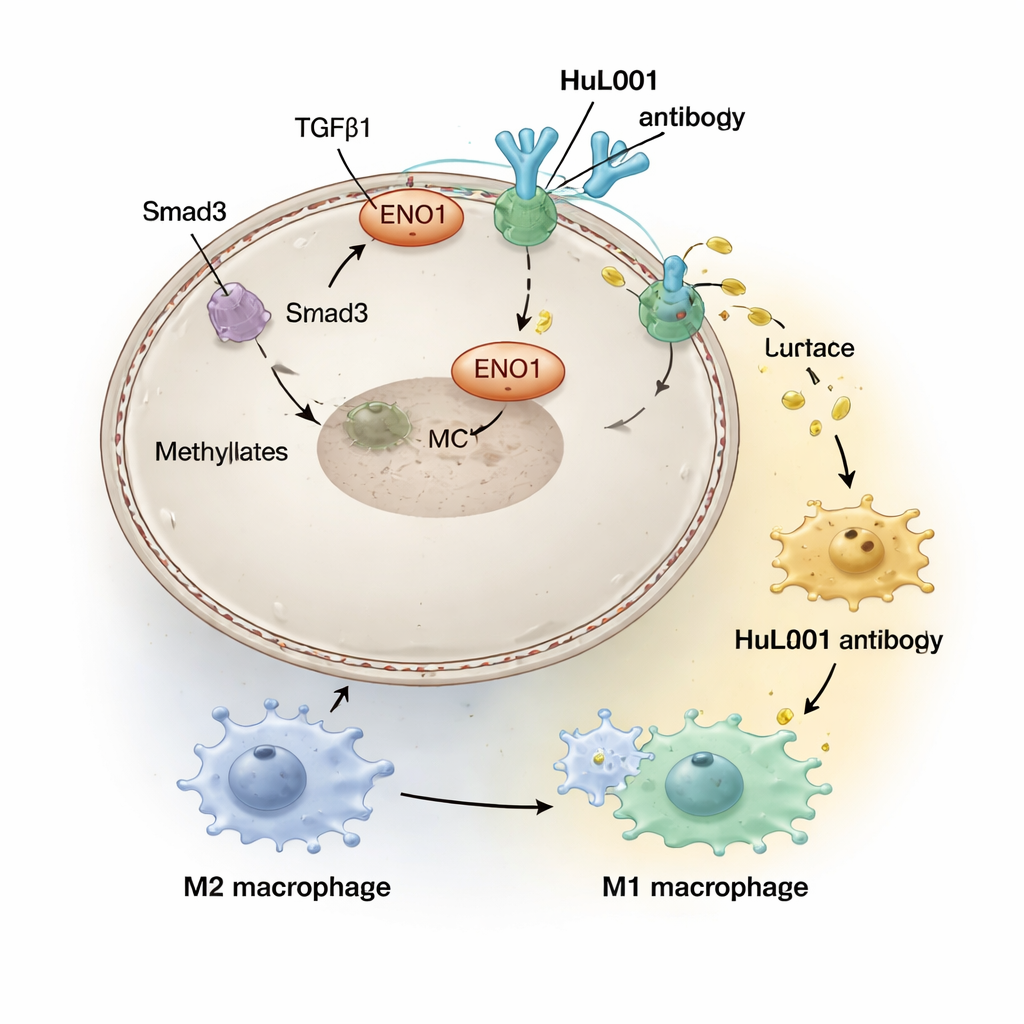
From Complex Pathways to Practical Promise
For non-specialists, the message of this study is that a tumor’s chemistry and its neighborhood of immune cells are deeply intertwined. ENO1 sits at a crossroads: it helps tumors burn sugar, export lactic acid, and recruit the wrong kind of immune help. By blocking surface ENO1 with a targeted antibody, the researchers were able to reduce harmful lactic acid buildup, flip macrophages from tumor helpers to tumor fighters, and make radiation more effective in difficult-to-treat cancer models. While HuL001 is not yet a clinical drug, the work suggests that aiming at metabolic gatekeepers like ENO1 could become a powerful way to turn a tumor’s own defenses against it and enhance existing therapies such as radiotherapy.
Citation: Lin, YS., Chang, HY., Hong, WZ. et al. Targeting ENO1 reprograms macrophage polarization to trigger antitumor immunity and improves the therapeutic effect of radiotherapy. Cell Death Dis 17, 194 (2026). https://doi.org/10.1038/s41419-026-08416-7
Keywords: tumor metabolism, macrophage polarization, radiotherapy, immunotherapy, lactate