Clear Sky Science · en
Sec8: a novel positive regulator of RIG-I in anti-RNA viral defense
How Our Cells Spot Sneaky Viruses
Many dangerous viruses, including influenza and emerging animal viruses, carry their genetic material as RNA. Our cells rely on internal alarm systems to notice these invaders quickly and launch an antiviral response. This paper explores a little‑known cellular protein called Sec8 and shows that it quietly plays a central role in keeping one of our key viral sensors, RIG‑I, alive and active long enough to protect us.
A Cellular Burglar Alarm Under Threat
RIG‑I is a molecular “burglar alarm” that patrols the inside of cells for bits of viral RNA. When it detects suspicious RNA, it changes shape, switches on, and sends signals that ultimately trigger type I interferons—potent warning molecules that help nearby cells resist infection and activate immune defenses. Because an overactive alarm can cause harmful inflammation or autoimmune disease, cells tightly control RIG‑I levels. Several other proteins can tag RIG‑I for destruction by attaching small ubiquitin chains, leading it to the cellular “shredder” called the proteasome. Until now, it was unclear which additional cellular factors protect RIG‑I from being prematurely destroyed, especially during active RNA virus infection.
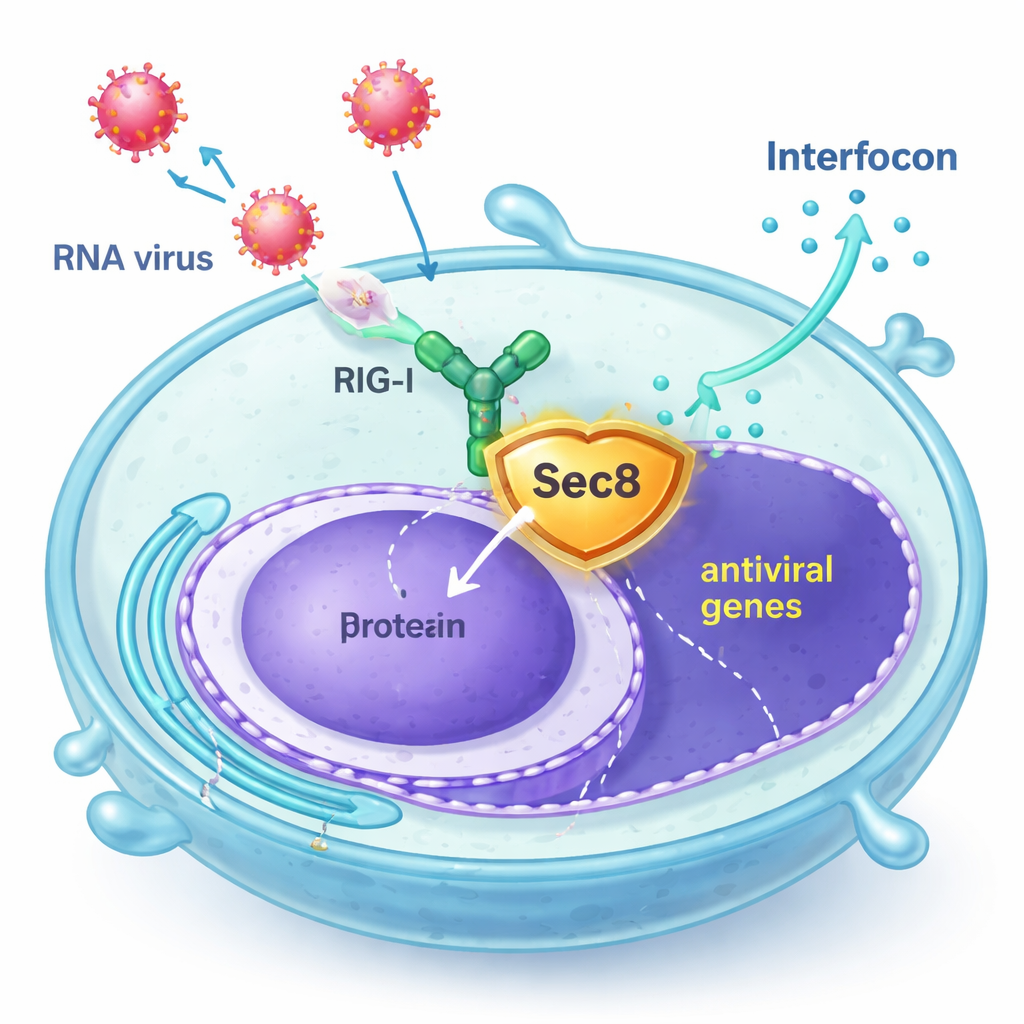
Sec8 Steps In as a Bodyguard
The authors discovered that Sec8, best known as part of an eight‑protein complex that helps secretory vesicles fuse with the cell surface, actually boosts antiviral signaling. In human cell lines and in primary mouse immune cells, increasing Sec8 levels led to stronger activation of interferon‑related genes after infection with RNA viruses such as vesicular stomatitis virus (VSV) and Sendai virus, or after exposure to synthetic viral RNA. Conversely, reducing Sec8 lowered the production of interferon and interferon‑stimulated genes, leaving cells less able to resist viral replication. These findings suggested that Sec8 acts not just in secretion, but as a positive regulator within the antiviral alarm pathway.
Blocking the Cellular Shredder
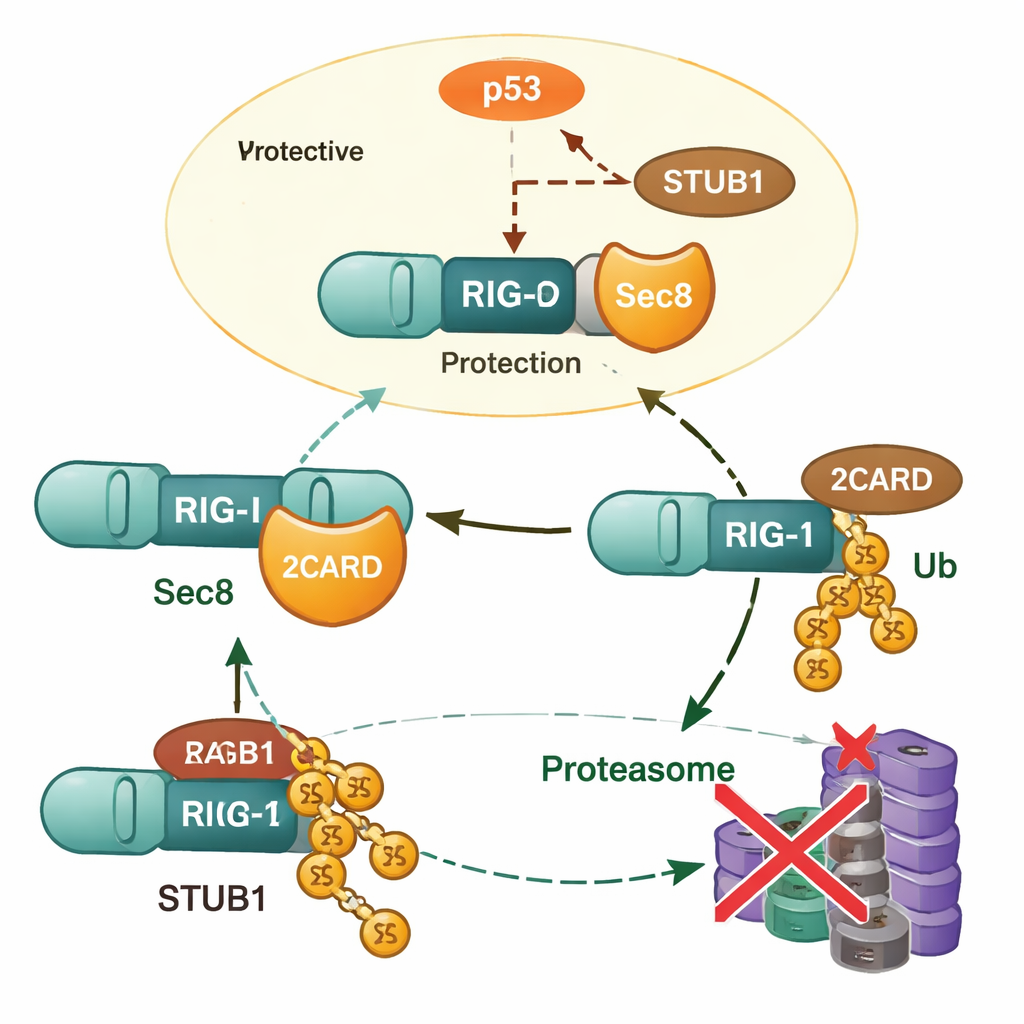
Digging deeper, the researchers found that Sec8 does not increase RIG‑I’s gene expression; instead, it keeps the RIG‑I protein from being degraded. When Sec8 was removed, RIG‑I levels dropped more quickly, and this loss could be prevented by drugs that block the proteasome, implicating the cell’s protein‑recycling machinery. The team identified another protein, STUB1, as a key “tagger” that attaches a specific type of ubiquitin chain (K48‑linked) to RIG‑I at an amino acid called lysine 190. This tag marks RIG‑I for destruction. Sec8 interferes with this process in two ways: it physically competes with STUB1 for binding to the same activation region on RIG‑I, and it lowers the amount of STUB1 the cell makes in the first place.
Sec8, p53, and Control of the Tagger
To explain how Sec8 reduces STUB1 production, the authors turned to gene regulation. They pinpointed a short stretch of DNA that acts as the core on‑switch (promoter) for the STUB1 gene and showed that the famous tumor‑suppressor protein p53 binds this region and raises STUB1 levels. Sec8 dampens both the amount and the activating phosphorylation of p53, so p53 can no longer drive STUB1 production as effectively. When p53 was blocked or STUB1 was silenced, the harmful effects of losing Sec8 on interferon production and viral growth were largely reversed. This places Sec8 at the top of a regulatory chain that runs from p53 to STUB1 and finally to RIG‑I stability.
From Petri Dish to Living Animals
The team then tested the importance of Sec8 in mice engineered to lack the Sec8 gene specifically in certain immune cells. After infection with VSV, these animals produced less interferon, carried higher amounts of virus in organs such as the spleen, liver, and lungs, and showed worse lung damage, greater weight loss, and lower survival than control mice. These in‑vivo results confirmed that Sec8 is not a minor player but a significant protector against RNA virus infection in a whole organism.
Why This Matters for Future Therapies
In plain terms, this study shows that Sec8 acts like a bodyguard for the antiviral sensor RIG‑I. By keeping the “tagger” protein STUB1 in check and directly shielding RIG‑I from being sent to the cellular shredder, Sec8 allows cells to mount a timely interferon response and better control RNA viruses. Understanding this newly described p53–STUB1–RIG‑I axis opens the door to future antiviral strategies that stabilize RIG‑I or mimic Sec8’s protective actions, potentially enhancing our defenses against a wide range of RNA‑based viral infections.
Citation: Wang, L., Ma, W., Hou, P. et al. Sec8: a novel positive regulator of RIG-I in anti-RNA viral defense. Cell Death Dis 17, 165 (2026). https://doi.org/10.1038/s41419-026-08414-9
Keywords: innate immunity, RNA viruses, RIG-I, ubiquitination, interferon signaling