Clear Sky Science · en
Lactate transmission from hypoxic tumor cells promotes macrophage senescence and M2 polarization via the DNMT1-NHE7 axis to accelerate endometrial cancer progression
Why starving tumors of oxygen matters
Endometrial cancer, which arises from the lining of the uterus, is becoming more common worldwide. Many advanced tumors are difficult to treat because they grow back, spread, or resist standard therapies. This study looks at what happens inside these tumors when they run low on oxygen—a common feature of fast‑growing cancers—and shows how a simple waste product, lactate, can reprogram nearby immune cells to help the cancer grow instead of fighting it.
Low oxygen turns tumors into lactate factories
As endometrial cancer cells multiply, their blood supply cannot keep up, creating pockets of low oxygen, or hypoxia. The cells respond by switching on a master regulator called HIF1A that rewires how they make energy. Instead of relying mainly on efficient energy production, the cells ramp up glycolysis, a quick but wasteful process that produces large amounts of lactate. The researchers analyzed tumor data from hundreds of patients and found that higher HIF1A levels went hand in hand with increased activity of many glycolysis‑related genes and transporters that pump lactate out of the cancer cells. Patients whose tumors had more HIF1A tended to fare worse, suggesting this metabolic shift is closely linked to aggressive disease.
How tumor waste reshapes helpful immune cells
Macrophages are immune cells that can either attack tumors or support them, depending on their state. In this work, the team showed that under hypoxia, endometrial cancer cells release far more lactate into their surroundings. This lactate is exported through a transporter called MCT3 on tumor cells and then imported into macrophages via another transporter, MCT1. Once inside, lactate nudges macrophages away from a defensive mode and toward the so‑called M2 state, which is associated with tissue repair, suppression of inflammation, and, unfortunately, support of tumor growth. In laboratory experiments, macrophages exposed to lactate‑rich tumor fluid displayed more markers of the M2 type, and the fluid they secreted in turn boosted cancer cell growth, movement, and invasion. 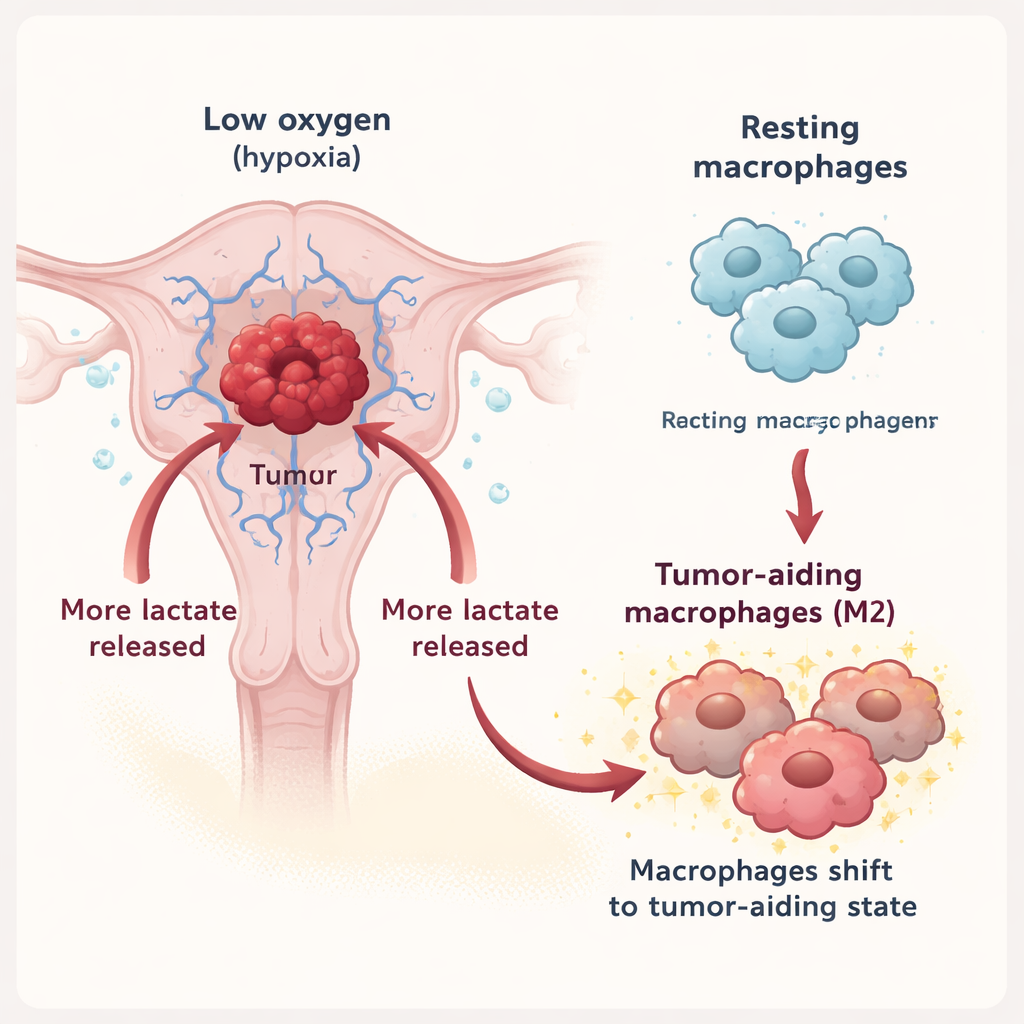
An epigenetic switch that silences a cellular “pH valve”
The study goes a step further by uncovering the molecular switch inside macrophages that connects tumor‑derived lactate to this harmful behavior. Lactate does not just float around; it can decorate histone proteins in the nucleus—a chemical tag known as lactylation—that influences which genes are turned on or off. The researchers found that lactate increases a specific histone mark on the control region of the DNMT1 gene, raising its activity. DNMT1 is an enzyme that adds methyl groups to DNA and can silence genes. One of its targets here is NHE7, a protein that helps control acidity inside certain cell compartments. When DNMT1 is boosted, it increases methylation of the NHE7 gene, turning it down. Reduced NHE7 disrupts internal pH balance and activates a signaling route known as the MAPK pathway. This pathway, in turn, pushes macrophages toward the tumor‑aiding M2 state and a form of aging called senescence, which locks them into a chronic, pro‑tumor mode.
From molecular chain reaction to faster‑growing tumors
To see whether this chain of events matters in living organisms, the scientists created mouse models in which human endometrial cancer cells were grown together with human‑like macrophages. When extra lactate was supplied, tumors grew larger, showed more dividing cells, and contained more M2‑type, senescent macrophages with high DNMT1 and low NHE7. Strikingly, when the macrophages were engineered to overproduce NHE7, this blunted the tumor‑boosting effect of lactate: tumors were smaller, had more dying cells, and contained fewer M2 and senescent macrophages. Blocking the DNMT1 enzyme or the MAPK pathway also weakened lactate’s ability to reprogram macrophages, reinforcing the idea that these molecules form a connected axis. 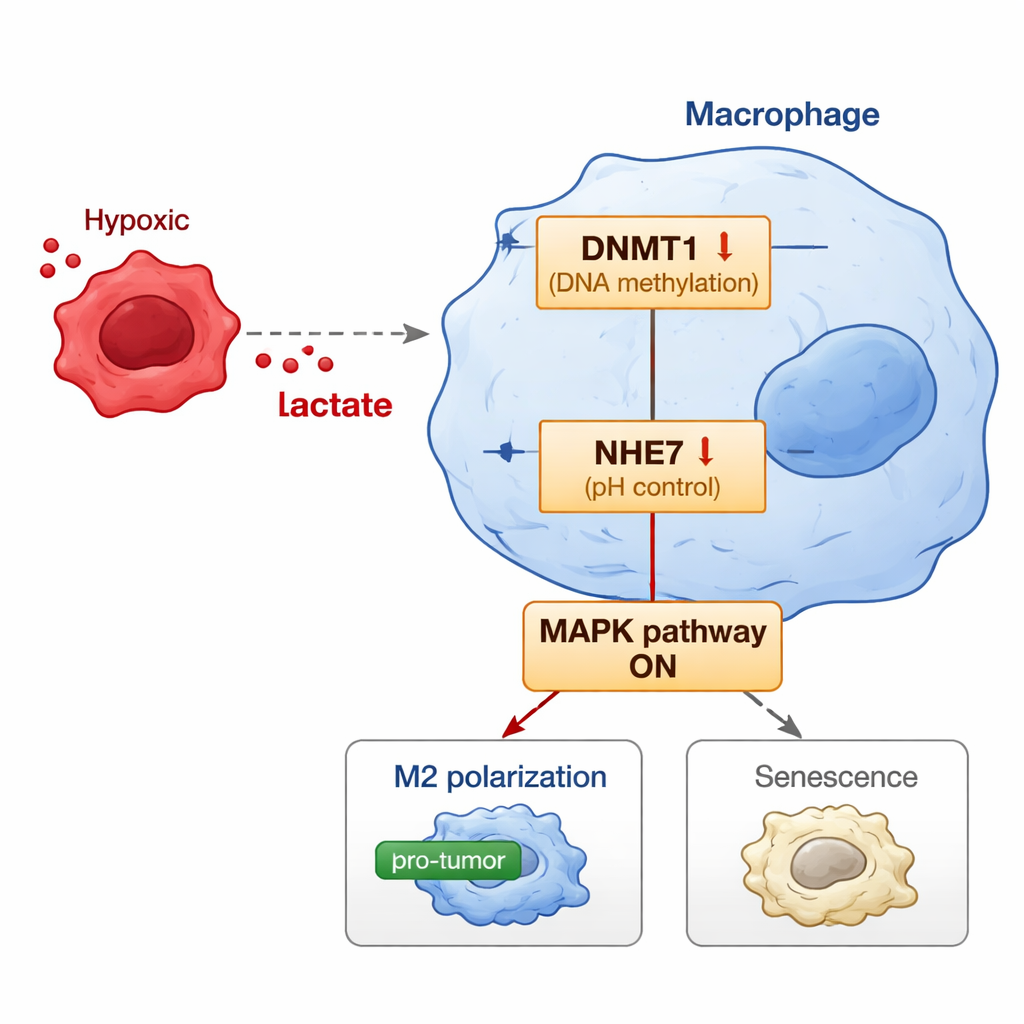
Turning a tumor’s own tricks into treatment targets
For non‑specialists, the key message is that endometrial tumors can use low oxygen and lactate—once thought of as mere cellular leftovers—to “brainwash” nearby immune cells into helping the cancer. They do this through an epigenetic relay: lactate boosts DNMT1, which silences NHE7, which then activates signaling that fixes macrophages in a tumor‑friendly, aging state. By interfering with any step in this lactate–DNMT1–NHE7–MAPK chain, it may be possible to restore more anti‑tumor immune activity and slow cancer growth. This work points toward future therapies that do not just target the cancer cells directly but also re‑educate the surrounding immune cells the tumor has turned to its advantage.
Citation: Yang, S., Ma, Y., Wu, T. et al. Lactate transmission from hypoxic tumor cells promotes macrophage senescence and M2 polarization via the DNMT1-NHE7 axis to accelerate endometrial cancer progression. Cell Death Dis 17, 185 (2026). https://doi.org/10.1038/s41419-026-08411-y
Keywords: endometrial cancer, tumor microenvironment, lactate metabolism, macrophage polarization, epigenetic regulation