Clear Sky Science · en
Beyond metabolism: exploring the regulatory and therapeutic implications of lactate and lactylation in cancer-regulated cell death
Why a “waste product” from sugar matters for cancer
For decades, lactate—the substance that makes overworked muscles burn—was written off as cellular garbage. This review article shows that in cancer, lactate is anything but waste. It acts as fuel, as a chemical signal, and even as a switch that can decide whether tumor cells live or die. Understanding this hidden role may open new ways to make cancer cells self‑destruct while sparing healthy tissues.
The sugar shortcut that rewires cancer cells
Cancer cells are notorious for their “sweet tooth.” Even when oxygen is plentiful, they prefer to break down glucose quickly into lactate instead of fully burning it in their mitochondria, a shift known as the Warburg effect. This strategy lets tumors generate energy and building blocks at high speed. The resulting flood of lactate is pumped out through special transporters, shaping an acidic, nutrient‑rich tumor microenvironment that supports growth and helps tumors adapt to stress, such as low oxygen or chemotherapy.
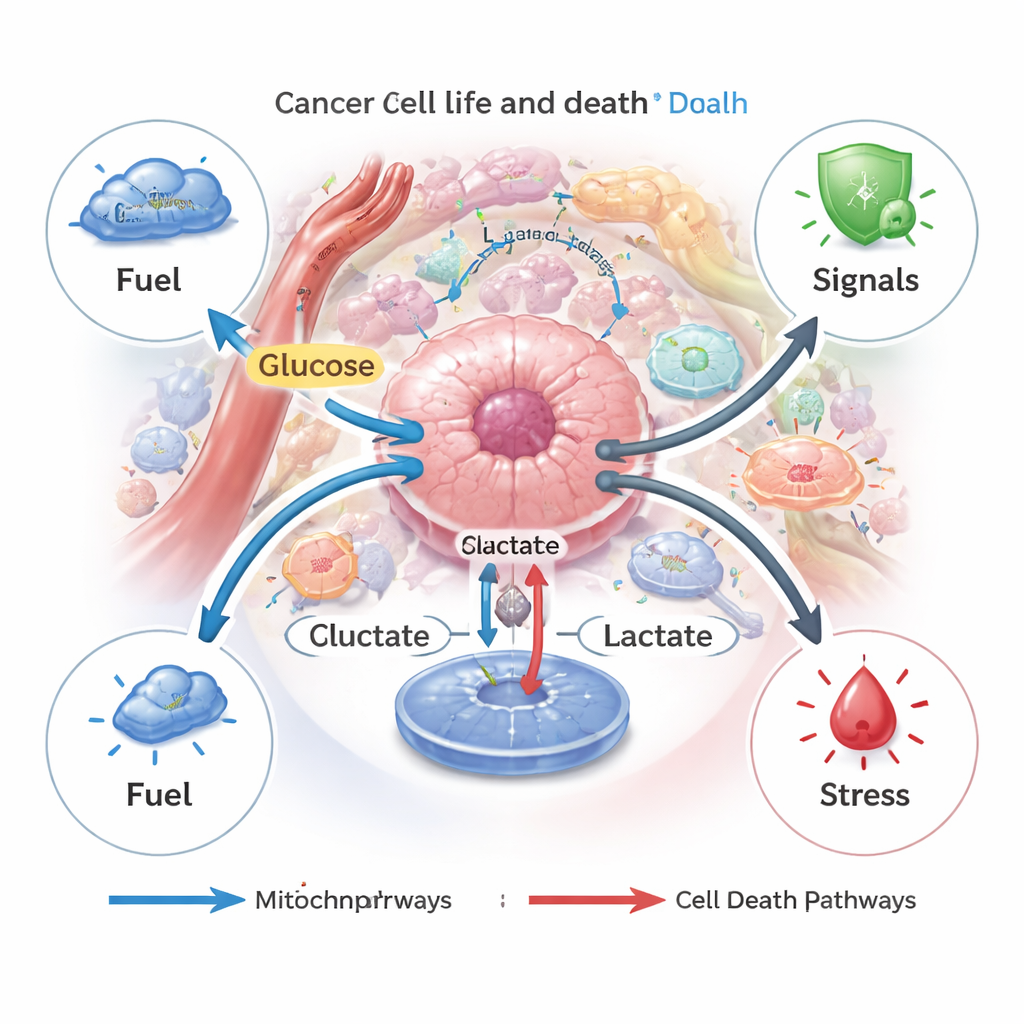
Lactate as a dial for different kinds of cell death
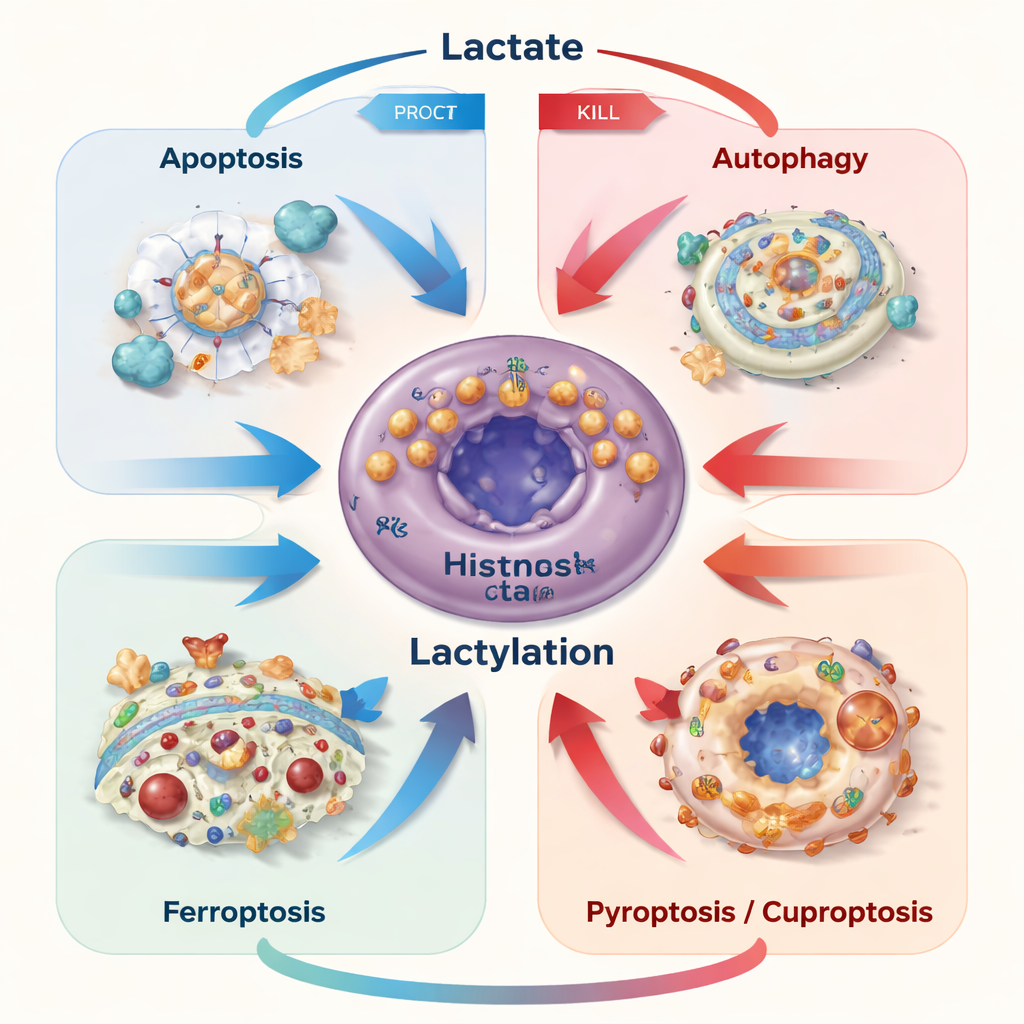
Cell death in our bodies is usually tightly controlled, helping remove damaged or dangerous cells. Cancer cells often escape these programs. The authors describe how lactate can both block and trigger several regulated death routes—apoptosis (cell “suicide”), autophagy (self‑digestion), ferroptosis (iron‑driven membrane damage), pyroptosis (inflammatory bursting), and cuproptosis (copper‑driven collapse). When cancer cells generate or import moderate amounts of lactate, they tend to use it as fuel and as a survival signal, turning on pathways that protect them from chemotherapy, nutrient shortage, or targeted drugs. But when lactate builds up inside cells, for example if its export is blocked, the resulting acid stress and mitochondrial injury can flip the switch and push cells toward apoptosis or ferroptosis instead.
Lactylation: when lactate writes on proteins
A striking recent discovery is that lactate can be converted into a small chemical tag and attached to lysine residues on proteins, a modification known as lactylation. Enzymes act as “writers” and “erasers” of these tags, decorating both DNA‑associated histones and many other proteins. These marks change which genes are turned on, how enzymes behave, and how stable key regulators are. In cancer, lactylation fine‑tunes the balance between survival and self‑destruction. It can, for example, increase proteins that block apoptosis, ramp up recycling through autophagy, shield cells from ferroptosis by tightening iron control and boosting antioxidants, or alter new forms of death like cuproptosis by changing how cells handle copper‑sensitive proteins.
A two‑way conversation between metabolism and death programs
The relationship is not one‑sided: cell death pathways also reshape how tumors use sugar and produce lactate. When mitochondria are damaged during early apoptosis or mitophagy (the selective removal of mitochondria), cells often fall back on fast glycolysis, raising lactate output. Surrounding support cells, such as cancer‑associated fibroblasts, can undergo similar rewiring and become lactate factories that feed nearby tumor cells. Other types of death, like ferroptosis, tend to suppress glycolysis and therefore lower lactate levels. The result is a dynamic feedback loop in which metabolism and death mechanisms continually adjust to one another as the tumor evolves and responds to treatment.
Turning a weakness into a treatment strategy
Because lactate and lactylation can either protect or kill cancer cells depending on context, the authors argue that therapies must do more than simply block lactate production. Instead, future treatments may selectively reroute lactate flows, tweak its transporters, or target specific lactylation marks to tip cancer cells over the edge while re‑energizing anti‑tumor immune cells that are currently paralyzed by the acidic environment. Smart drug‑delivery systems and nanomedicines that respond to local lactate or pH levels are emerging as ways to apply this precision. In simple terms, the article’s message is that what once looked like metabolic trash is actually a powerful control knob on cancer cell fate—and learning to turn that knob the right way could make existing therapies far more effective.
Citation: Chen, C., Lin, A., Zhao, J. et al. Beyond metabolism: exploring the regulatory and therapeutic implications of lactate and lactylation in cancer-regulated cell death. Cell Death Dis 17, 184 (2026). https://doi.org/10.1038/s41419-026-08410-z
Keywords: lactate, lactylation, cancer cell death, tumor metabolism, ferroptosis