Clear Sky Science · en
Tumour immune contexture and immune evasion in sporadic and Lynch syndrome-associated microsatellite unstable colorectal cancers
Why This Matters for Cancer Patients
Immunotherapy has transformed the outlook for many people with colorectal cancer, but not everyone benefits. This study asks a key question: why do some tumors that should, in theory, be highly visible to the immune system still manage to slip through its net? By comparing two types of genetically unstable colon cancers—those linked to the inherited Lynch syndrome and those that arise sporadically—the researchers uncover important differences in how immune cells surround, attack, or sometimes protect these tumors.
Two Paths to the Same Kind of Tumor
Microsatellite unstable (MSI) colorectal cancers carry large numbers of DNA errors, which create abnormal proteins that can act like flags for the immune system. These tumors can arise either because of inherited defects in DNA repair (Lynch syndrome) or because of changes that occur only in the tumor itself (sporadic MSI cancers). Although both tumor types look similar under the microscope, their immune surroundings—and possibly their responses to treatment—are not the same. The team assembled 43 MSI colorectal cancers from Finnish patients, including both Lynch-associated and sporadic cases, and analyzed them using tissue staining, whole-genome sequencing, and RNA sequencing to map out in detail which immune cells were present and how the tumors avoided being destroyed.
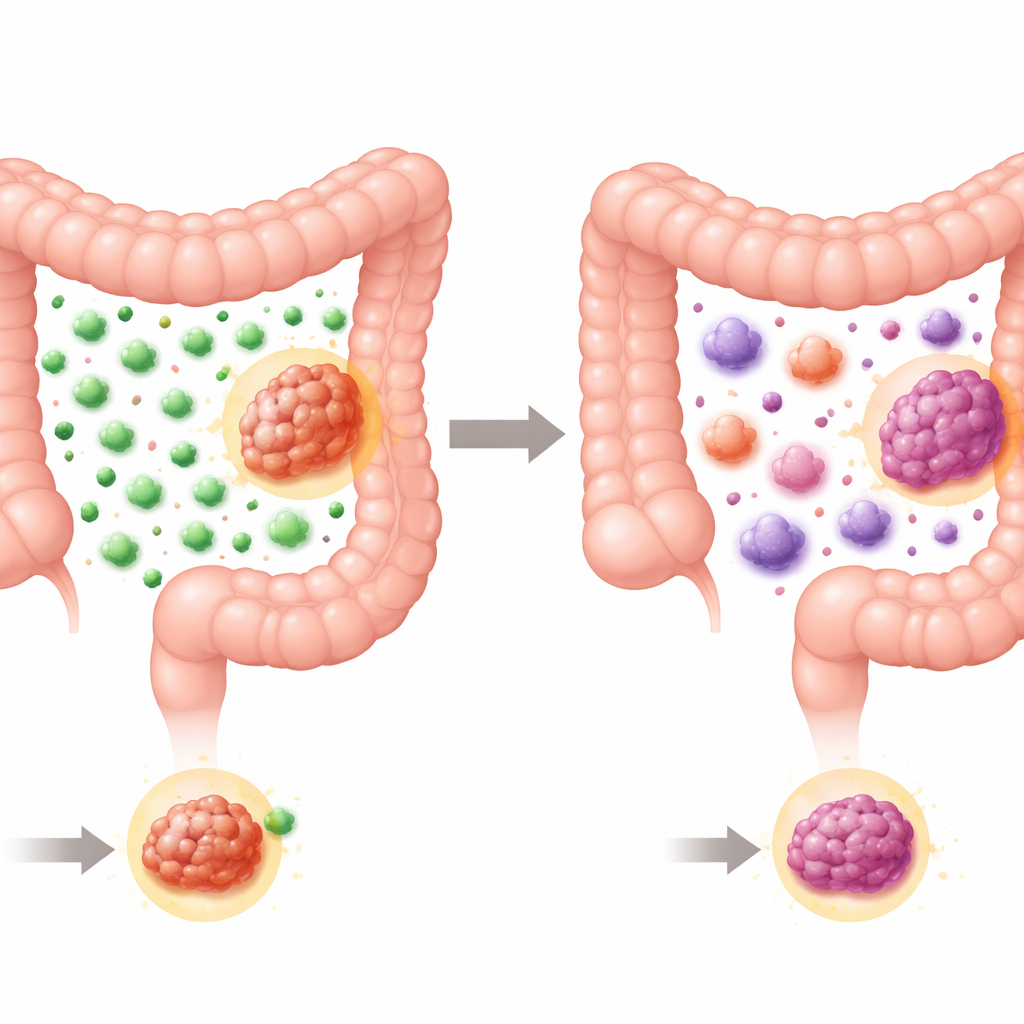
How Many Immune Cells Show Up
The researchers first focused on T cells, the immune system’s targeted “hit men.” They measured an established immune cell score that combines the density of total and killer T cells in the center and the invasive edge of each tumor. As expected, MSI tumors overall had higher scores than more typical colorectal cancers, reflecting their heightened ability to attract T cells. But when they looked within the MSI group, a clear pattern emerged: every Lynch syndrome tumor had a high immune cell score, while sporadic MSI tumors split roughly in half between high and low scores. In other words, inherited MSI cancers almost always drew in strong T cell armies, whereas sporadic MSI cancers were more variable, with some looking comparatively “cold” to the immune system.
Friendly and Unfriendly Helper Cells
Next, the study turned to myeloid cells—innate immune cells such as macrophages and neutrophils that can either help T cells fight cancer or, paradoxically, protect tumors. Using multiplex staining that marks several proteins at once, the authors classified macrophages into two broad flavors: M1-like cells, which support inflammation and tumor killing, and M2-like cells, which tend to be soothing, wound-healing, and often tumor-promoting. Both Lynch and sporadic MSI tumors contained many myeloid cells, but sporadic tumors stood out for having more M2-like macrophages, especially in the supportive tissue at the tumor’s invasive edge. In that region, M2-like cells even outnumbered M1-like cells in sporadic tumors. Within the sporadic group, those tumors that did have high T cell scores also had more M1-like macrophages, while M2-like levels stayed high regardless—hinting that the balance between these two types of macrophages may help decide whether T cells can mount an effective attack.
Genetic Tricks and Tumor Diversity
To understand why some MSI tumors evade the immune system, the researchers examined genetic changes that affect how tumor proteins are presented to immune cells and how “brakes” on the immune response are applied. Both Lynch and sporadic MSI cancers frequently carried mutations in key components of the antigen presentation machinery, but only subtle differences separated the two groups. More striking were the differences in immune “checkpoint” signals. Sporadic tumors expressed higher levels of PD-L2 and CD40L, molecules that can shape how immune cells behave and that are increasingly being explored as drug targets in combination with existing checkpoint inhibitors. The team also measured how many potential neoantigens each tumor harbored and how genetically uniform or mixed (clonal vs. heterogeneous) the tumors were. Surprisingly, a higher predicted neoantigen load did not translate into more T cells. Instead, tumors that were more genetically diverse—especially sporadic MSI cancers—tended to carry more neoantigens but also showed patterns, such as reduced neutrophil presence, that suggested the immune system was not fully exploiting these targets.
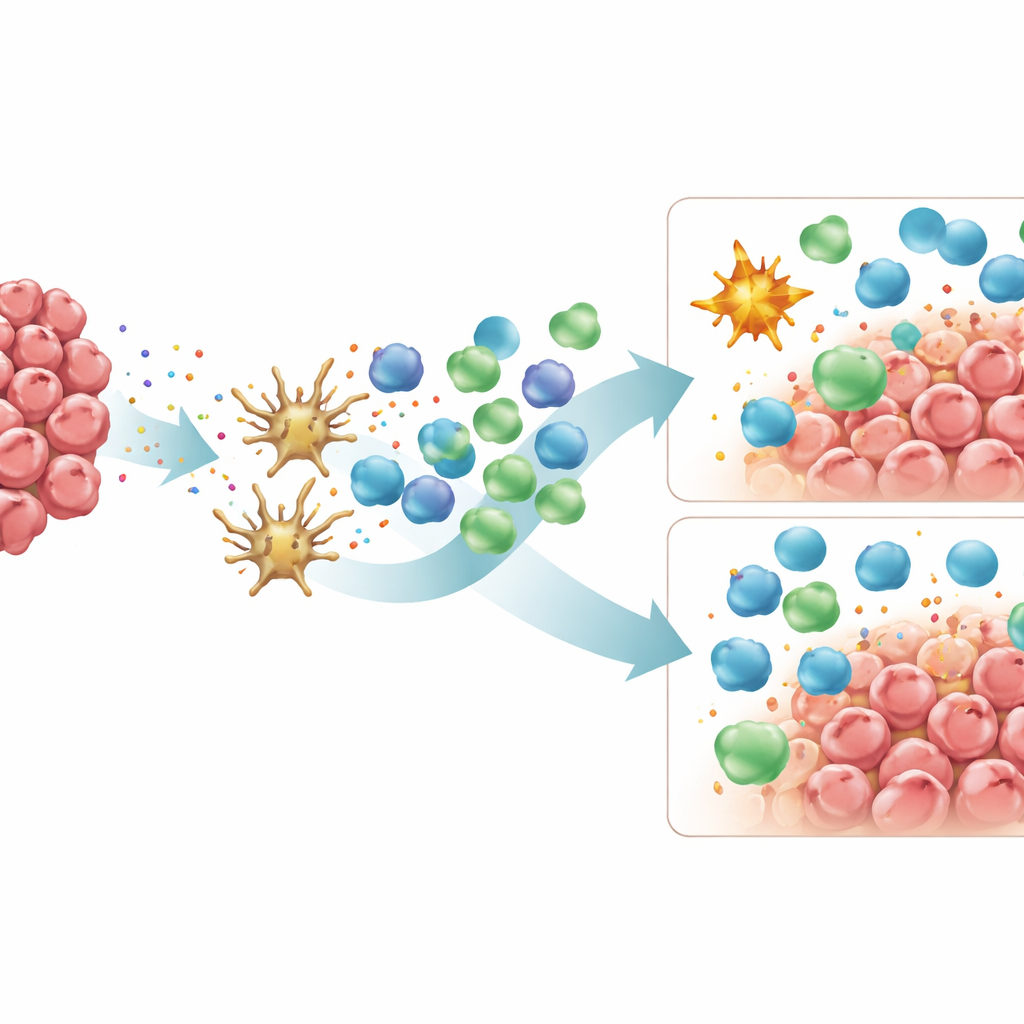
What This Means for Future Treatments
Overall, the study paints Lynch-associated MSI colorectal cancers as consistently “hot” tumors rich in T cells, whereas sporadic MSI cancers form a more mixed group, with some strongly infiltrated and others buffered by immunosuppressive macrophages and higher expression of certain checkpoint molecules. At the same time, sporadic tumors more often display high genetic diversity and a heavy burden of neoantigens that are spread across different tumor cell subclones, a pattern that previous work suggests can blunt the effectiveness of T cell responses. For patients, these findings help explain why not all MSI colorectal cancers respond equally well to immunotherapy, despite sharing a high mutation rate. They also point toward new strategies, such as drugs that reprogram or deplete M2-like macrophages or therapies aimed at additional checkpoints like PD-L2 and CD40L, which might be especially relevant for sporadic MSI tumors. By better matching treatment approaches to each tumor’s unique immune landscape, clinicians may be able to improve and personalize immunotherapy outcomes in colorectal cancer.
Citation: Martin, S., Elomaa, H., Väyrynen, J.P. et al. Tumour immune contexture and immune evasion in sporadic and Lynch syndrome-associated microsatellite unstable colorectal cancers. Br J Cancer 134, 1019–1030 (2026). https://doi.org/10.1038/s41416-025-03302-z
Keywords: microsatellite unstable colorectal cancer, Lynch syndrome, tumor immune microenvironment, tumor-associated macrophages, cancer immunotherapy