Clear Sky Science · en
Functional footprints of homologous recombination deficiency in prostate cancer revealed by ctDNA fragmentation and transcription factor accessibility
Why this matters for cancer patients
Many men with advanced prostate cancer could benefit from drugs that exploit weaknesses in how their tumors repair damaged DNA. Yet today, doctors often miss who will respond because testing usually requires difficult tissue biopsies and focuses on only a handful of genes. This study shows how a simple blood draw can be turned into a rich, multi-layered readout of these DNA repair weaknesses, potentially guiding more precise and gentler treatment choices.
A new way to read cancer signals from blood
The researchers focused on a particular repair problem called homologous recombination deficiency, or HRD, which makes tumors especially vulnerable to drugs like PARP inhibitors and to certain chemotherapies. Instead of relying on tumor samples taken from bone or prostate tissue, they analyzed bits of tumor DNA floating in the bloodstream, known as circulating tumor DNA. From 375 men with metastatic prostate cancer, they selected 106 whose blood carried enough tumor DNA to study in depth and then applied several complementary tests to the same plasma samples.
Looking beyond single gene mutations
First, they sequenced a panel of key DNA repair genes, including well-known players such as BRCA2, BRCA1, and PALB2, along with other genes that shape how aggressive prostate tumors become. BRCA2 emerged as the most commonly altered repair gene and often appeared together with loss of other important safeguards like PTEN and RB1. But the team also examined large-scale changes in chromosome structure across the entire genome, using low-depth whole-genome sequencing to calculate a genomic instability score. Tumors with damaged BRCA genes, or with high scores, had heavily rearranged genomes and were linked to worse overall survival, highlighting that big structural changes can be just as informative as specific mutations.
Footprints of repair failure in mutation patterns
In a subset of patients, the scientists zoomed in further by sequencing all protein-coding regions to read the detailed pattern of mutations that had accumulated over time. Certain combinations of base changes and small insertions or deletions act like fingerprints of the processes that created them. They found that classic HRD-linked signatures, such as one called SBS3 and another indel pattern known as ID6, were enriched in tumors with repair gene defects and high genomic instability. Other signatures pointed to separate problems, like mismatch repair failure or a distinct subtype driven by CDK12, underlining that different DNA repair breakdowns leave recognizably different scars in the genome.
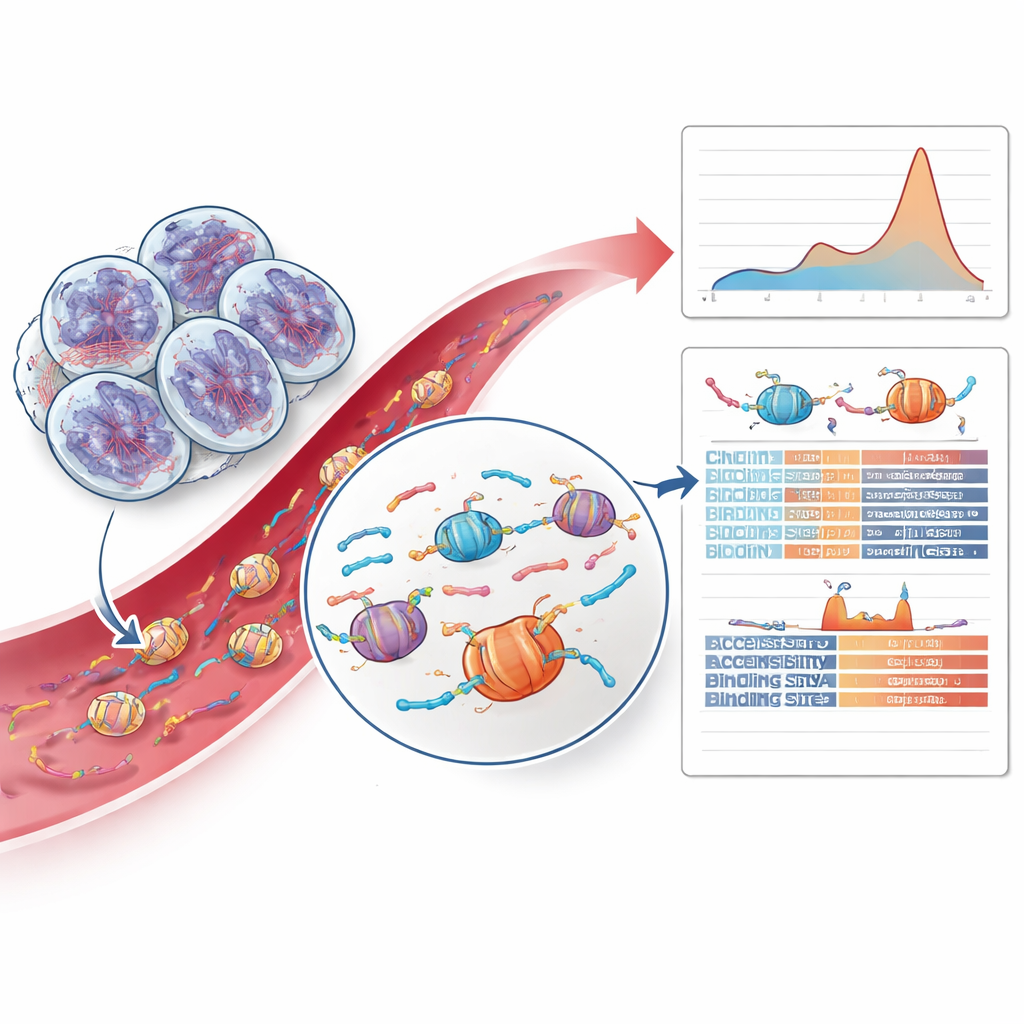
Decoding DNA fragment patterns and chromatin clues
The most innovative part of the work went beyond mutations entirely and examined how the tumor DNA fragments were chopped up. When cells die, their DNA is cut around protein packages called nucleosomes, creating fragments with characteristic lengths and end patterns. The team discovered that tumors with HRD showed a relative excess of slightly longer fragments corresponding to two nucleosomes, a shift not seen in other prostate cancers or in healthy controls. By training a cautious machine-learning model on fragment lengths and fragment-end features, they could pick out HRD-positive cases from blood alone with encouraging accuracy. They also probed how accessible different regions of the genome were around binding sites for transcription factors—proteins that control gene activity—and found that certain zinc finger binding sites were less accessible in HRD tumors, suggesting deeper, repair-related changes in chromatin organization.
What this could mean for patients
Together, these layers of information—from specific gene mutations and large chromosome reshuffling to subtle shifts in DNA fragment size and chromatin accessibility—form a more complete picture of DNA repair weakness in prostate cancer. For a layperson, the key message is that a carefully analyzed blood sample can reveal not just whether a familiar gene like BRCA2 is mutated, but whether a tumor behaves like one with a serious repair defect, even when the usual tests look normal. If validated in larger and more diverse groups of patients, this multimodal blood-based approach could help doctors more reliably identify who is likely to benefit from PARP inhibitors or platinum drugs, monitor changes over time, and ultimately personalize therapy using a simple, repeatable test.
Citation: Vlachos, G., Moser, T., Lazzeri, I. et al. Functional footprints of homologous recombination deficiency in prostate cancer revealed by ctDNA fragmentation and transcription factor accessibility. Br J Cancer 134, 949–960 (2026). https://doi.org/10.1038/s41416-025-03301-0
Keywords: prostate cancer, liquid biopsy, DNA repair, circulating tumor DNA, PARP inhibitors