Clear Sky Science · en
Immunosuppressive immune microenvironment landscapes in VISTA-high gastric cancer
Why the Body’s Defenses Sometimes Fail Against Stomach Cancer
Modern cancer treatments increasingly rely on waking up the immune system so it can attack tumors. Yet in many people with advanced stomach cancer, these powerful drugs work poorly or stop working. This study explores one important reason why: a brake molecule called VISTA that reshapes the local environment around the tumor, turning immune cells from fighters into bystanders or even helpers of the cancer. Understanding this hidden control switch could open the door to more precise and effective immunotherapy.
A Hidden Brake in the Tumor Neighborhood
The authors focused on VISTA, a protein found mainly on certain white blood cells that sit in and around tumors. VISTA acts as a stop signal for immune responses. While other brakes such as PD-1 and PD-L1 are already targeted by approved drugs, VISTA’s role in stomach cancer has been unclear. To investigate this, the team analyzed tissue from 172 patients using advanced multicolor staining to map many cell types at once. They also used single-cell RNA sequencing and spatial transcriptomics on smaller sets of samples to examine which cells carry VISTA, what those cells are doing, and exactly where they sit in the tumor landscape. 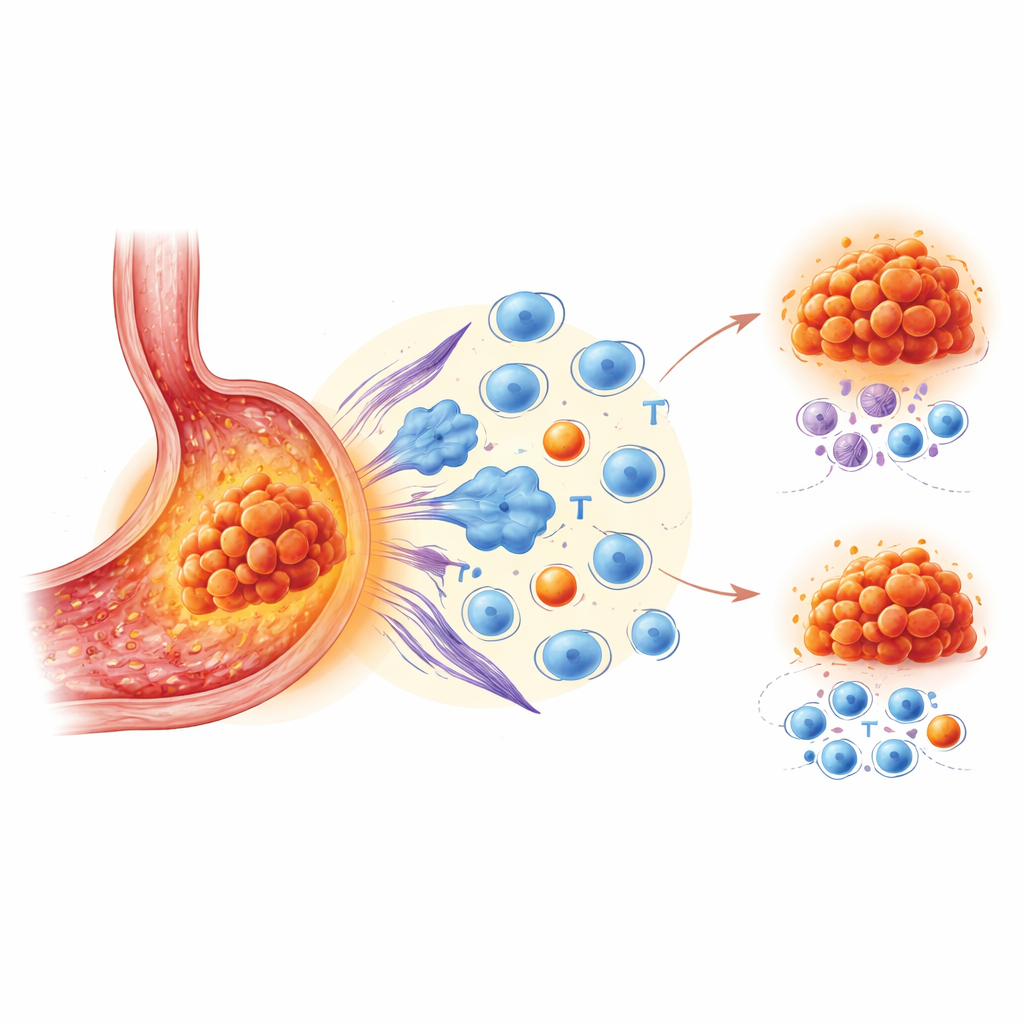
When VISTA Is High, the Immune System Is Muffled
By comparing tumors with high versus low levels of VISTA, the researchers found a consistent pattern. Cancers rich in VISTA were surrounded by immune cells, but not the kind that mount a strong attack. Instead, there was a buildup of exhausted killer T cells that had lost their punch, regulatory T cells that dampen immune responses, scar-forming support cells called fibroblasts, and macrophages skewed toward a tumor-nurturing state. Patients whose tumor regions contained more VISTA had shorter periods before their disease worsened after immunotherapy, even when accounting for other clinical factors. In other words, a VISTA-heavy neighborhood looked busy under the microscope but functioned as an “immune-silent” or suppressive zone.
Macrophages as Key Middlemen
Diving deeper, the scientists zoomed in on monocytes and macrophages—immune cells that can either devour cancer or shield it. At the single-cell level, the gene encoding VISTA (called VSIR) was especially active in several macrophage subgroups, particularly those that efficiently display bits of tumor proteins on their surface and those with features of so‑called M2, or wound-healing, macrophages. Using a computational timeline of cell development, the team saw VISTA turn on as cells moved from early monocytes into more mature macrophages that clustered around tumor cells. Spatial mapping confirmed that VISTA-positive macrophages tended to sit close to cancer cells, and patients whose tumors had more of these cells precisely in these locations fared worse after immune checkpoint treatment. 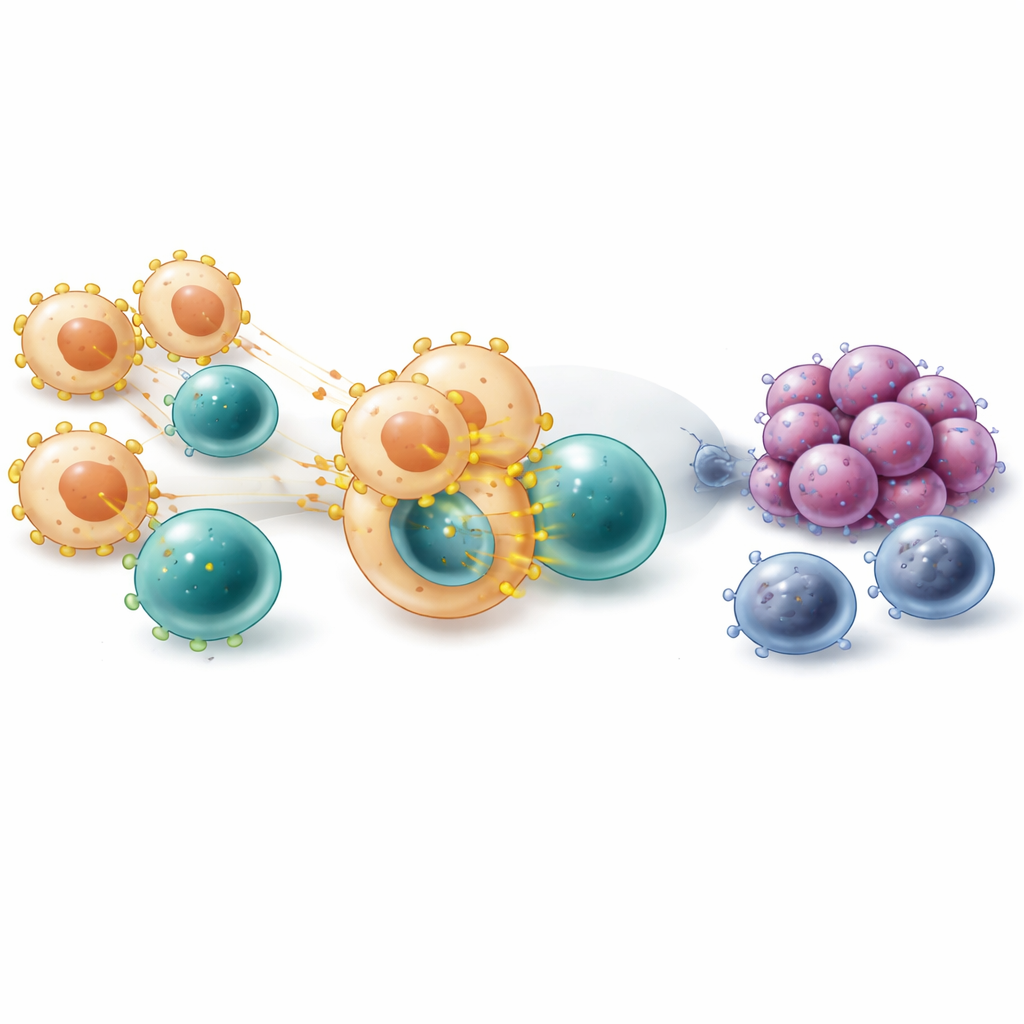
How VISTA-Positive Cells Wear Down T Cells
The study also explored how VISTA-bearing macrophages talk to T cells. Communication maps built from gene-expression data suggested that these macrophages frequently contact T cells through molecular pairs such as LGALS9 and PTPRC, and through molecules involved in presenting tumor fragments to T cells. This constant presentation can, over time, push T cells from an early, active state into a chronically stimulated, exhausted state marked by multiple inhibitory signals on their surfaces. In samples where macrophages showed higher VSIR expression, there were more regulatory T cells and more exhausted killer T cells, reinforcing the idea that VISTA-positive macrophages help drive immune burnout rather than a sharp, effective attack.
What This Could Mean for Future Treatments
Overall, the work paints VISTA as a central switch that helps stomach tumors build an immunosuppressive niche, especially through specialized macrophages that both present tumor material and send inhibitory signals to T cells. For patients, this suggests that high VISTA expression marks a form of resistance to current immune drugs targeting PD‑1 or PD‑L1 alone. Blocking VISTA—possibly in combination with existing checkpoint inhibitors—might reinvigorate T cells and tip the balance back toward tumor control. While more laboratory and clinical studies are needed, this research offers a roadmap for designing new treatment combinations and for identifying which patients are most likely to benefit.
Citation: Luo, Y., Peng, H., Yao, Q. et al. Immunosuppressive immune microenvironment landscapes in VISTA-high gastric cancer. Br J Cancer 134, 1066–1079 (2026). https://doi.org/10.1038/s41416-025-03290-0
Keywords: gastric cancer, tumor microenvironment, immune checkpoint, macrophages, T cell exhaustion