Clear Sky Science · en
Identification of Smmhc-expressing mesenchymal cells in orofacial bone at single-cell resolution
Why Jawbone Repair Matters
Broken or missing jawbone is surprisingly hard to fix. Surgeons often have to borrow bone from a patient’s hip or leg to rebuild defects in the face, a process that can be painful and limited by how much bone is available. This study looks inside the tiny spaces of the jawbone to find the key local stem cells that build and maintain facial bones and teeth-supporting tissues. Understanding these cells could lead to gentler, more precise ways to repair the jaw using the body’s own regenerative power.
Mapping the Hidden World Inside Jawbone
Instead of treating jawbone as a uniform block of tissue, the researchers set out to catalog every type of non-blood cell living inside the bone marrow of the mouse lower jaw. They used single-cell RNA sequencing, a technique that reads which genes are active in thousands of individual cells, to create a detailed “cell atlas.” By first enriching rare stromal and stem-like cells, they identified thirteen main cell groups. Among these were early and late mesenchymal progenitors, several flavors of bone-forming cells, cells that help organize the bone marrow environment, and supportive cells linked to blood vessels and nerves. This level of resolution revealed that orofacial mesenchymal stem/stromal cells are not a single population but a collection of specialized subgroups.
A Newly Spotted Stem Cell at the Top of the Ladder
Within this crowded cellular ecosystem, one group stood out: a previously unrecognized population of mesenchymal cells that produce a protein called smooth muscle myosin heavy chain (Smmhc). Computational “trajectory” analysis placed these Smmhc-expressing cells at the very beginning of the developmental path that leads toward bone-forming lineages. Communication mapping showed that they both send and receive many molecular signals with neighboring bone-related cells, suggesting they act as an organizing hub. Gene activity patterns indicated that these cells sit next to blood vessels and share features with perivascular progenitors, a type of cell thought to be a natural reservoir for stem cells in many organs.
From One Source to Many Jaw Tissues
To test what these Smmhc-positive cells actually do in a living animal, the team used genetic lineage tracing in mice. They marked Smmhc-expressing cells and followed their descendants over time. During early life, the labeled cells appeared throughout the jaw: in the bone that holds the teeth, within the hard tissue of teeth themselves, in the ligament that anchors teeth to bone, and around blood vessels. Many of these descendants carried markers of active bone builders and other specialized tissue cells, showing that Smmhc-positive cells are multipotent—they can give rise to several different orofacial tissues. In adult animals, the descendants were more tightly clustered around blood vessels, hinting that the stem cell pool becomes more quiescent and regionally restricted with age while still retaining regenerative potential.
What Happens When These Cells Are Removed
The researchers then asked what would happen if this stem cell subset were selectively wiped out. Using a two-step genetic strategy, they rendered Smmhc-positive cells sensitive to diphtheria toxin and then eliminated them at either early or later stages of life. In both cases, detailed 3D scans and microscopic analyses showed that the mandible lost bone volume, its internal spongy network became thinner and more widely spaced, and overall jawbone architecture was compromised. Markers of bone-forming activity dropped sharply, indicating fewer or less active osteoblasts. Surprisingly, cells responsible for bone breakdown, the osteoclasts, were also reduced, along with key genes that drive their function. This means the special Smmhc-positive cells help coordinate not only bone building but also bone removal, a balance that is vital for healthy remodeling.
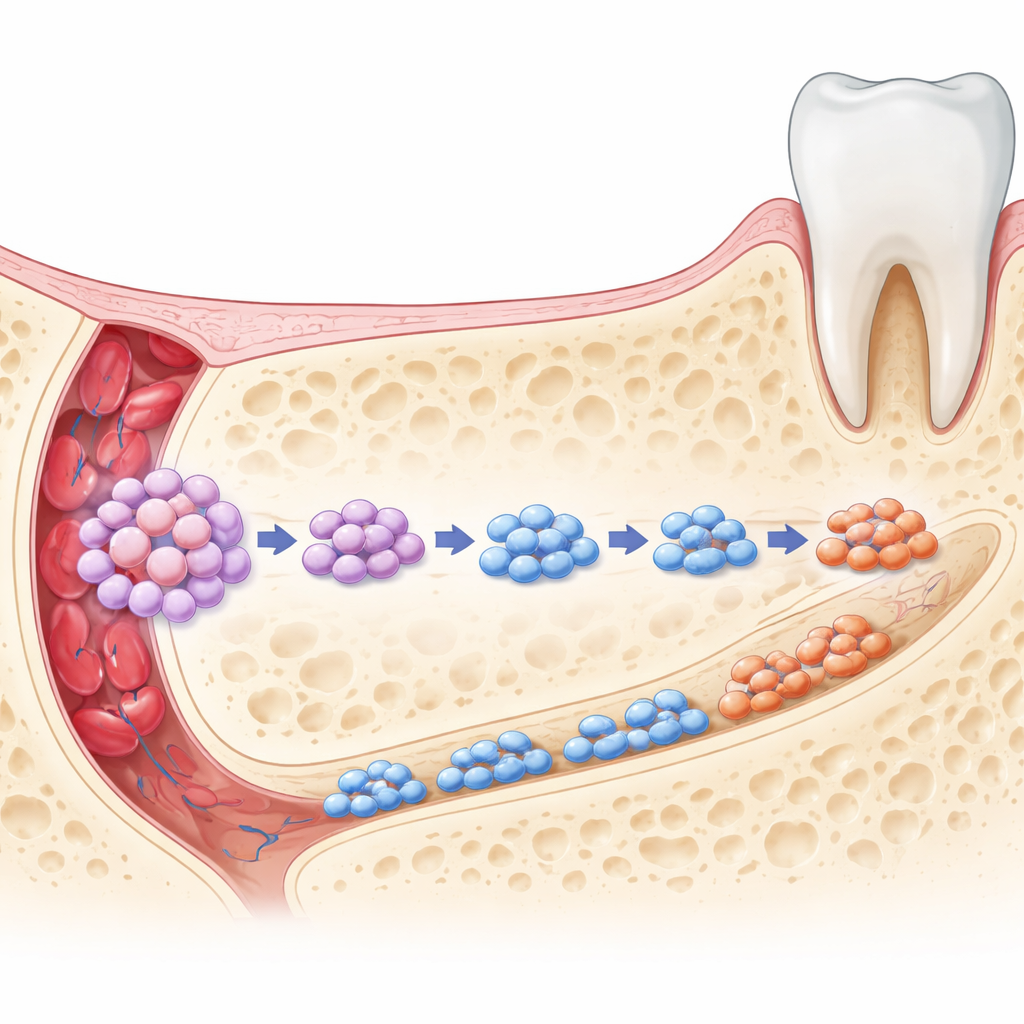
Implications for Future Face and Jaw Repair
To a non-specialist, the takeaway is that the jawbone contains a distinct, vessel-associated stem cell population that quietly supervises the construction and upkeep of facial bone and tooth-supporting tissues. When these Smmhc-expressing cells are present, they help generate new bone, maintain ligaments and dentin, and signal to bone-resorbing cells so that old bone is removed in step with new bone formation. When they are lost, the system falters: bone mass falls, structure weakens, and normal renewal is disturbed. By defining this critical stem cell subset and its interactions, the study points toward future therapies that might harness or protect these cells for targeted, site-specific regeneration of craniofacial defects—potentially reducing the need for large grafts and improving outcomes in dental and maxillofacial surgery.
Citation: Fan, Y., Wei, Y., Wu, Z. et al. Identification of Smmhc-expressing mesenchymal cells in orofacial bone at single-cell resolution. Bone Res 14, 33 (2026). https://doi.org/10.1038/s41413-026-00518-4
Keywords: jawbone stem cells, craniofacial regeneration, orofacial bone marrow, mesenchymal stromal cells, bone remodeling