Clear Sky Science · en
Spectrum and functions of ion channels and transporters in osteoclasts
Why Our Bones Need Tiny Gatekeepers
Our skeleton looks solid and unchanging, but it is constantly being torn down and rebuilt. Specialized cells called osteoclasts act as the demolition crew, dissolving old bone so that fresh bone can form. This review article explores a hidden cast of microscopic “gatekeeper” proteins—ion channels and transporters—that let charged atoms and nutrients move in and out of osteoclasts. By understanding how these tiny gates control bone breakdown, scientists hope to design better treatments for osteoporosis and other bone diseases.
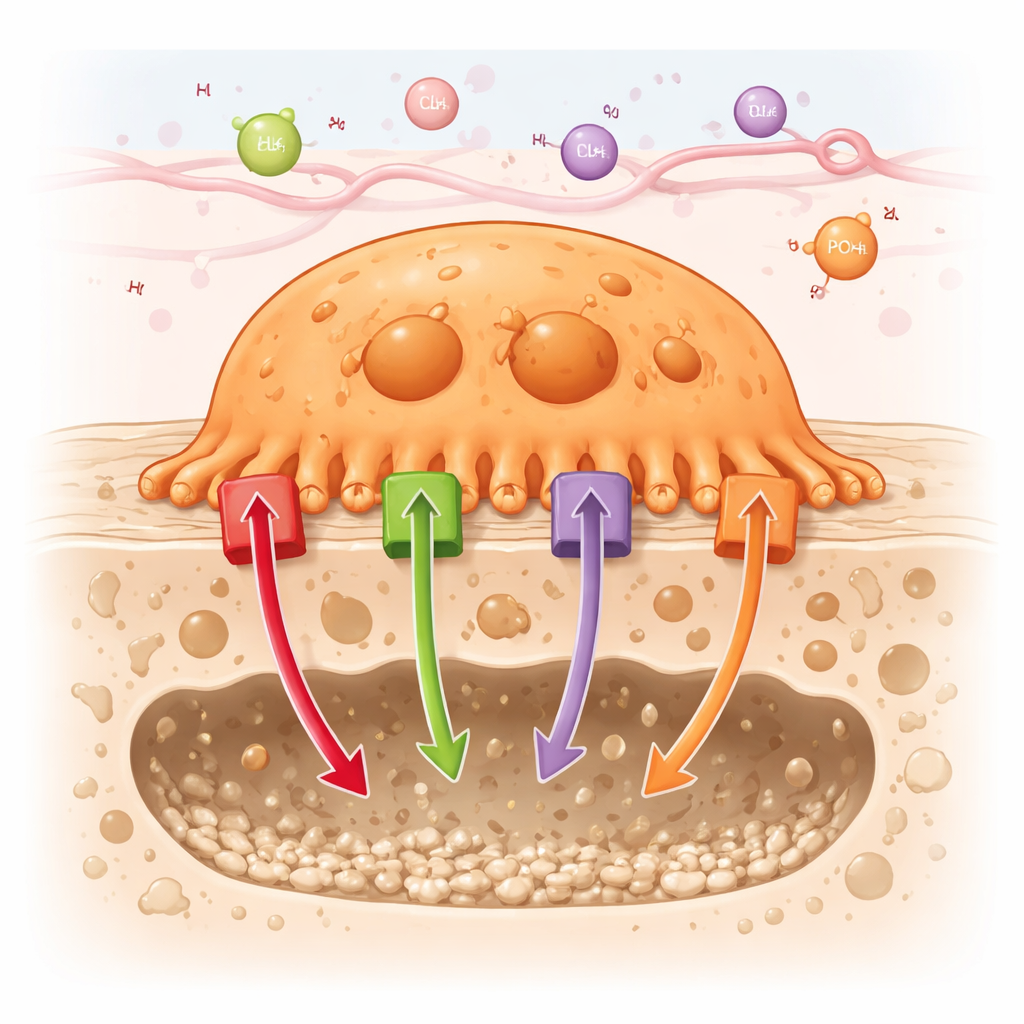
How Bone-Eating Cells Do Their Job
Osteoclasts work by attaching tightly to the bone surface and creating a sealed-off pocket where they can safely dissolve mineral and digest proteins. To make this miniature “digestion chamber” extremely acidic, osteoclasts pump large numbers of protons (hydrogen ions) into the pocket. At the same time, they shuttle other ions—such as chloride, calcium, and phosphate—through their membranes and internal compartments. The article surveys about 90 different channels and transporters found in osteoclasts and groups them into six families, including energy‑driven pumps (ATPases), cation and anion channels, coupled transporters, nutrient carriers, and ATP‑binding cassette (ABC) transporters.
Acid, Salt, and Energy: The Core Machinery
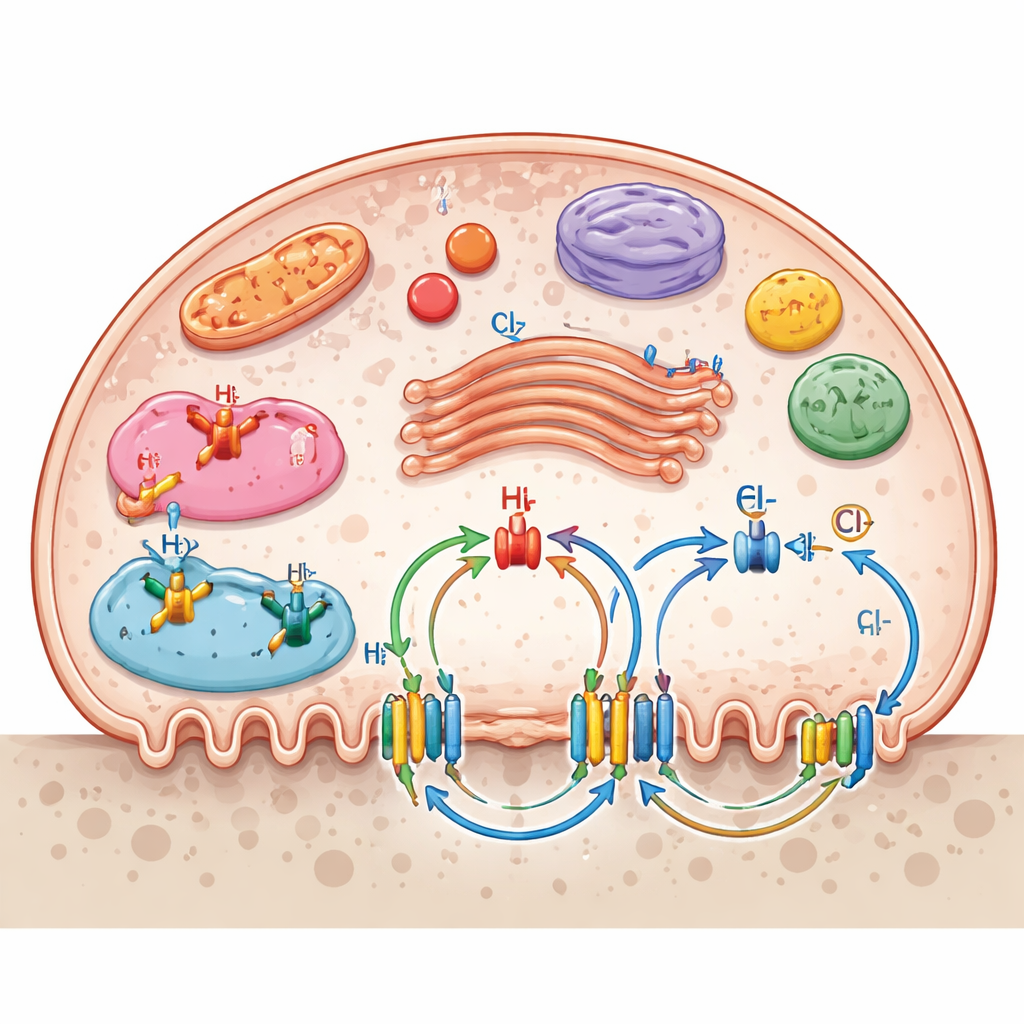
Two protein systems stand out as central to bone resorption. First, vacuolar proton pumps known as V-ATPases actively push protons into the resorption pocket, making it acidic enough to dissolve bone mineral and activate protein‑cutting enzymes. Specific V‑ATPase subunits, such as a3, d2, and several V1 components, are enriched at the osteoclast’s ruffled border and in lysosomes, and genetic defects in these subunits can either weaken bone or cause abnormal bone thickening. Second, a chloride–proton exchanger called ClC‑7, which partners with a protein named OSTM1, brings chloride ions into the same area. This balances electrical charge and allows continuous proton pumping. When ClC‑7 is mutated, humans and mice develop osteopetrosis, a condition marked by overly dense but fragile bones, illustrating how essential proper ion flow is for normal skeletal health.
Calcium, Phosphate, and Other Supporting Players
Beyond acid production, osteoclasts rely on finely tuned calcium and phosphate handling. A web of calcium channels and pumps in the cell membrane, endoplasmic reticulum, lysosomes, and mitochondria generate rhythmic calcium “oscillations” that switch on key genes for osteoclast formation and fusion. Transporters at the ruffled border and the opposite side of the cell move calcium and phosphate out of dissolved bone, either recycling them inside the cell or passing them back to the bloodstream. Other metals and minerals—such as manganese, magnesium, zinc, copper, and iron—are also controlled by specialized transporters and influence how aggressively osteoclasts resorb bone. For example, iron and certain forms of cell death linked to iron can ramp up or damp down osteoclast activity, while zinc transporters tend to restrain excessive bone loss.
Traffic Control Inside the Cell
Ion channels are not confined to the outer membrane. Many sit on internal compartments such as lysosomes, endosomes, the Golgi apparatus, and mitochondria, creating a layered transport network. These internal gates help set pH in digestive vesicles, fuel production in mitochondria, and the movement of enzymes and debris. The review highlights how multiple channel systems work together: sodium–hydrogen exchangers regulate internal acidity, potassium–chloride cotransporters help maintain membrane voltage and chloride balance, and purinergic and mechanosensitive channels translate chemical or mechanical signals into changes in bone resorption. Nutrient transporters for glucose, amino acids, nucleosides, and vitamin C further support the high energy and biosynthetic demands of actively resorbing osteoclasts.
From Basic Biology to New Treatments
Because so many bone disorders arise when osteoclasts are either overactive or underactive, the channels and transporters that guide their behavior are attractive drug targets. The authors review existing and experimental compounds that block V‑ATPases, TRP calcium channels, chloride exchangers, purinergic receptors, and other transporters, as well as newer ideas such as disrupting specific subunit interactions or using targeted nanoparticles to deliver drugs directly to bone. They emphasize that many of these proteins are also found in other tissues, so achieving true osteoclast specificity remains a challenge. Still, as genomic and imaging tools reveal more about where these channels sit and how they interact, the expanding “ion map” of osteoclasts is likely to yield more precise therapies for osteoporosis, inflammatory bone loss, and rare genetic bone diseases.
Citation: Chen, H., Zhang, Y., Zhu, Y. et al. Spectrum and functions of ion channels and transporters in osteoclasts. Bone Res 14, 35 (2026). https://doi.org/10.1038/s41413-026-00513-9
Keywords: osteoclasts, ion channels, bone resorption, V-ATPase, osteoporosis