Clear Sky Science · en
PRMT6 is required for initiating and amplifying macrophage-induced inflammation in heterotopic ossification by increasing CCL2 expression
When Healing Goes Too Far
Sometimes, after a serious injury or surgery, the body tries so hard to heal that it actually creates new bone where none should exist. This painful condition, called unwanted bone growth in soft tissues, can freeze joints, complicate amputations, and make everyday movement difficult. Current treatments are limited and often fail to prevent the problem from coming back. This study uncovers a key early switch in the immune system that links trauma to this abnormal bone formation—and points to a short, critical window when targeted treatment might stop the process before it starts. 
The Problem of Bone in the Wrong Place
After major trauma, such as burns, fractures, or orthopedic surgery, some patients develop islands of bone inside muscles, tendons, and other soft tissues. The new bone forms through a process similar to normal bone development, beginning with inflammation and ending with mature, mineralized tissue. While doctors know that immune cells called macrophages are central to this response, the step-by-step control of how they ignite and sustain inflammation has been unclear. Without that knowledge, therapies like anti-inflammatory drugs, radiation, or late surgical removal mostly treat the consequences, not the cause, and recurrence is common.
Inflammation’s Key Middleman
Using a mouse model that combines tendon injury with a burn—closely mimicking severe trauma in people—the researchers followed what happens at the injury site over time. They saw a rapid buildup of macrophages during the first days after injury, which then persisted as abnormal bone formed. When they depleted these cells, the extra bone almost vanished, and the surrounding tissue showed far less disruption of blood vessels and support cells. A deeper gene readout from injured tissue highlighted one molecule in these macrophages that stood out: PRMT6, an enzyme that modifies proteins and helps control which genes are turned on.
A Molecular Volume Knob for Inflammation
The team found that PRMT6 levels in macrophages rose quickly after injury and in response to danger signals and bacterial components in the lab. Mice lacking PRMT6, or mice in which PRMT6 was selectively reduced only in macrophages, had far fewer macrophages at the injury site and developed dramatically less abnormal bone. Importantly, the injured tendons in these animals actually healed better, with more orderly tissue and less scar formation. This indicates that PRMT6 is not required for healthy repair, but rather acts as a volume knob that amplifies damaging inflammation. When the researchers blocked PRMT6 with a drug, it only worked if given early—during the first weeks after injury. Starting treatment later had little effect, revealing a narrow but powerful therapeutic window. 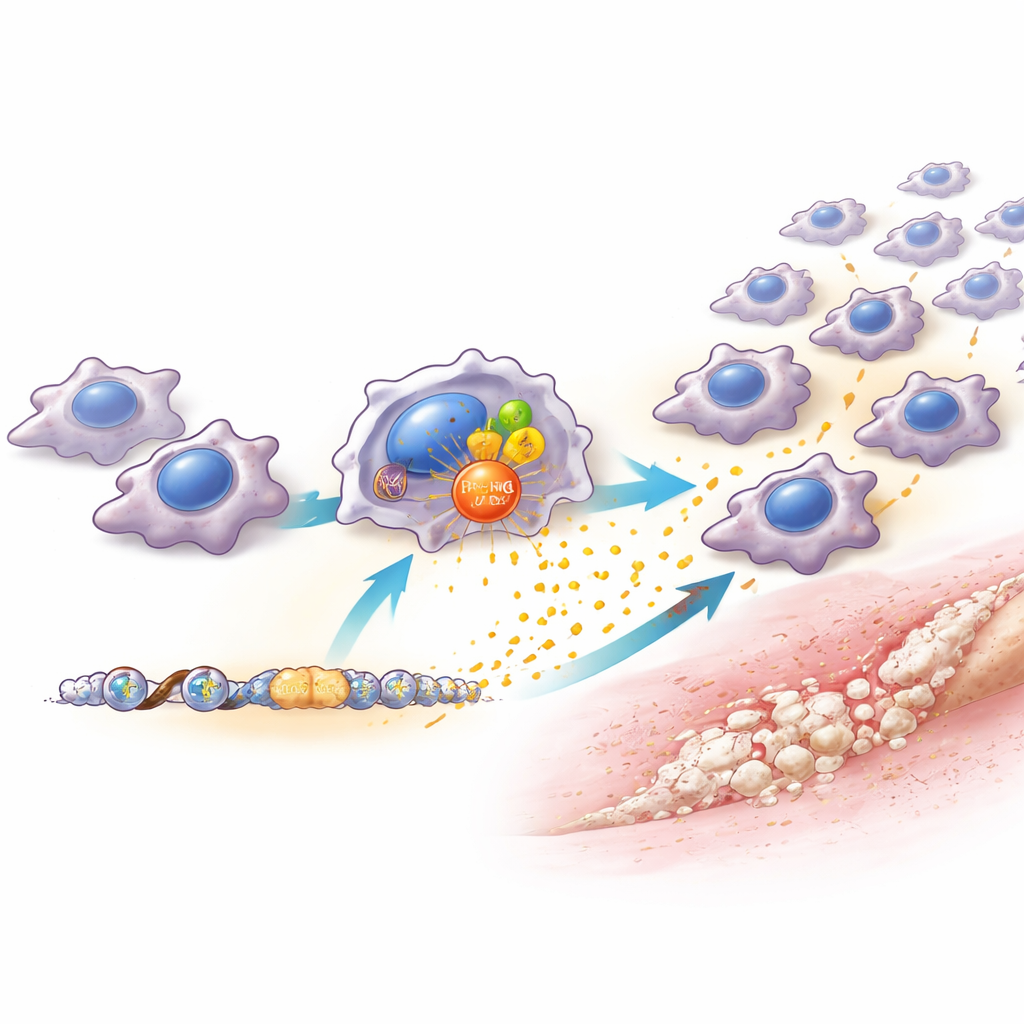
How One Signal Attracts Many Immune Cells
To understand how PRMT6 exerts this effect, the scientists studied macrophages in detail. Without PRMT6, these cells produced much lower amounts of several chemical attractants, especially one called CCL2, which normally draws more monocytes and macrophages out of the bloodstream and into injured tissue. PRMT6 helps switch on the CCL2 gene in two ways: it joins forces with the well-known inflammation controller NF-κB, and it chemically marks nearby DNA-packaging proteins to make that gene easier to read. As a result, macrophages with high PRMT6 pump out more CCL2, pulling in additional macrophages and building an “inflammation center” that nurtures blood vessels and bone-forming progenitor cells. When CCL2 was specifically reduced only in macrophages, the outcome closely mimicked PRMT6 loss—fewer macrophages, weaker inflammatory niches, and less extra bone. Adding back CCL2 partially restored both macrophage influx and abnormal bone growth.
A Targeted Chance to Prevent Unwanted Bone
Taken together, the study shows that PRMT6 in macrophages acts as an early epigenetic amplifier: it boosts a key chemical signal that recruits more immune cells, which in turn drives bone formation where it does not belong. Because blocking PRMT6 only during the early inflammatory phase sharply reduced unwanted bone while leaving normal tendon healing intact—and even improved tissue organization—this pathway offers a promising new strategy. In principle, a short, well-timed course of PRMT6-targeted therapy after major trauma or surgery could prevent long-term disability from ectopic bone, without compromising the body’s natural ability to repair itself.
Citation: Chu, W., Peng, W., Wu, Z. et al. PRMT6 is required for initiating and amplifying macrophage-induced inflammation in heterotopic ossification by increasing CCL2 expression. Bone Res 14, 29 (2026). https://doi.org/10.1038/s41413-026-00512-w
Keywords: heterotopic ossification, macrophages, inflammation, epigenetic regulation, CCL2 signaling