Clear Sky Science · en
C/EBPβ dictates postmenopausal FSHβ transcription and blockade of AEP/C/EBPβ pathway alleviates osteoporosis
Why this research matters for life after menopause
Many women are told that falling estrogen is the main culprit behind fragile bones after menopause. This study adds a new piece to the puzzle: another hormone, follicle-stimulating hormone (FSH), and a protein pair in the brain and bone—C/EBPβ and AEP—work together to accelerate bone loss. Even more intriguing, the researchers show in mice that blocking this pathway with an experimental pill can protect bones as effectively as an approved osteoporosis drug.
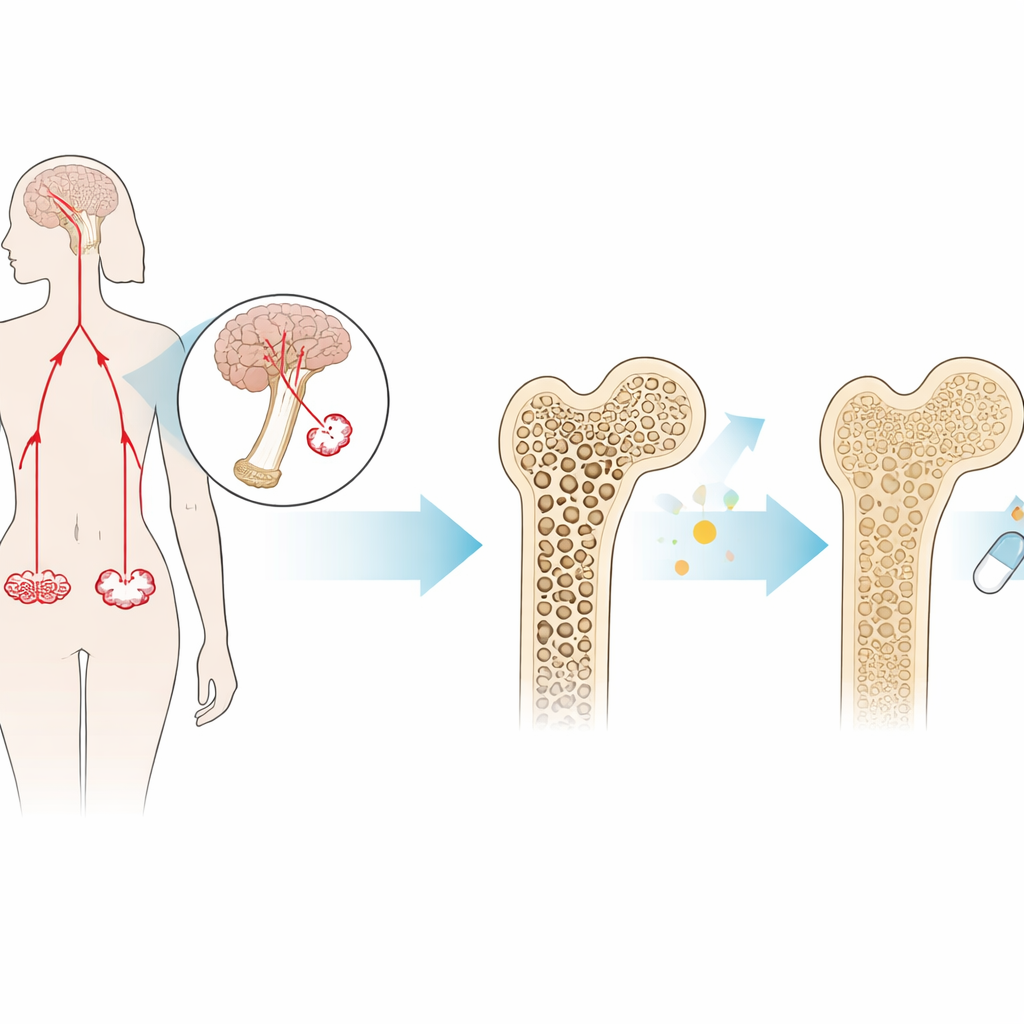
A lesser-known hormone with a big impact on bone
FSH is produced in the pituitary gland and is best known for its role in fertility. In women approaching menopause, FSH levels rise sharply several years before the final menstrual period. Beyond its reproductive role, FSH can act directly on bone: it binds to receptors on bone-resorbing cells (osteoclasts), pushing them to chew away bone faster than it is rebuilt. Earlier work showed that mice lacking FSH or its receptor are protected from bone loss, even when their estrogen is low. This suggested that FSH itself, not just estrogen deficiency, can drive postmenopausal osteoporosis.
The control switch in the pituitary
The new study focuses on C/EBPβ, a protein that sits on DNA and turns genes on or off. The authors discovered that C/EBPβ binds directly to the control region of the FSH beta-subunit gene—the part of the hormone that sets its identity—and boosts its production in the pituitary. In cultured pituitary cells, dialling C/EBPβ up increased FSH, while silencing C/EBPβ reduced FSH, especially when the cells were stimulated by the brain’s reproductive signal, GnRH. In ovariectomized mice, which mimic menopause by removing estrogen-producing ovaries, animals with reduced C/EBPβ made much less FSH in their pituitary glands and blood. These experiments reveal C/EBPβ as a key switch that determines how much FSH surges after estrogen drops.
A self-reinforcing loop and a new drug target
C/EBPβ also controls another protein called AEP, a cutting enzyme that can activate or disable other molecules. In the brain, a C/EBPβ–AEP chain has been linked to Alzheimer’s-like damage. Here, the team asked whether this same chain feeds back on FSH and bone. In mice lacking AEP, levels of both C/EBPβ and FSH fell in the pituitary, and bone loss after ovary removal was reduced. Blocking AEP with a small-molecule drug, dubbed #11a, produced a similar effect: after months of treatment, ovariectomized mice had lower FSH, stronger bones on high-resolution scans, and fewer overactive bone-resorbing cells. Another class of compounds—TrkB activators that indirectly dampen AEP—helped bone cells in dishes but could not lower FSH in animals, largely because their target receptor is scarce in the pituitary. This contrast sharpened the conclusion that directly inhibiting AEP is a more powerful way to shut down the C/EBPβ–FSH axis at its source.
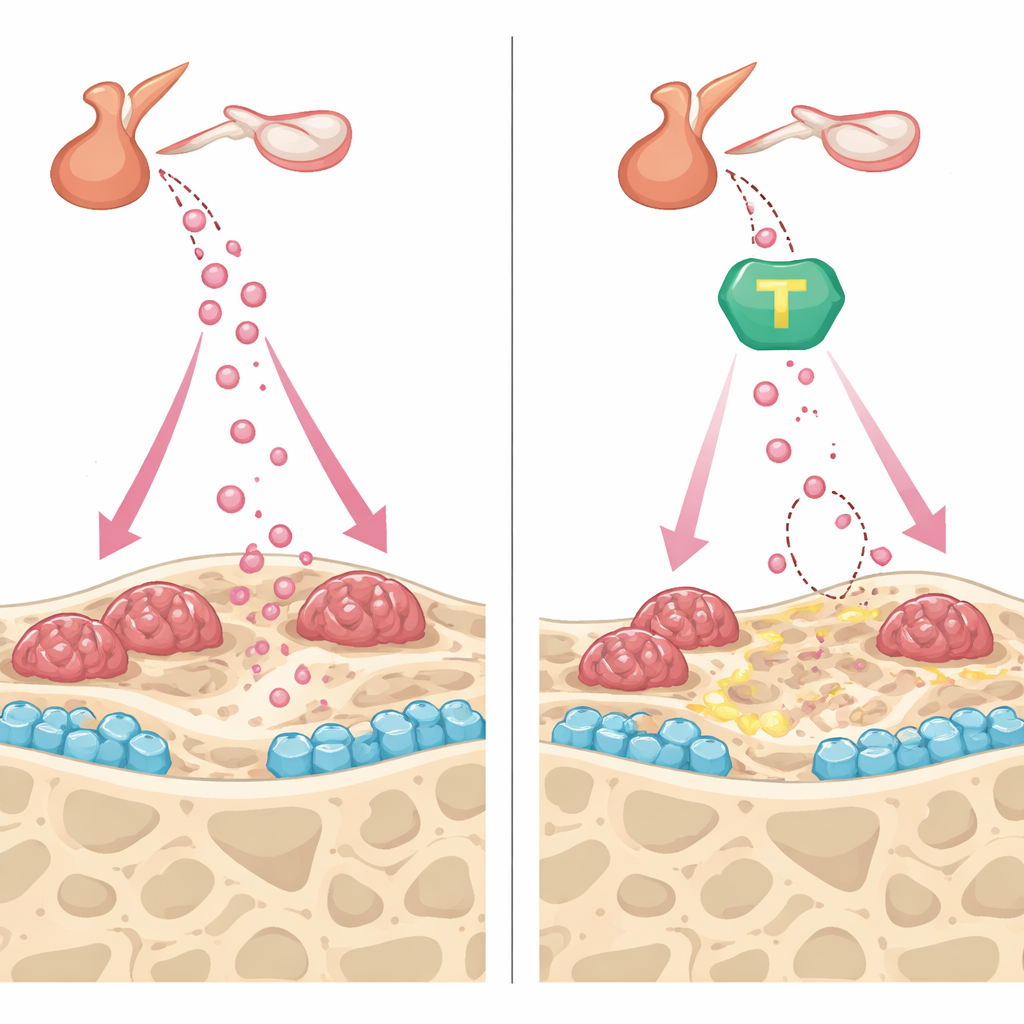
Protecting bone from both angles
Beyond lowering FSH, #11a and the TrkB activator CF3CN showed complementary benefits inside the bone itself. In cultures of bone-forming cells, both compounds sped up maturation and mineral deposition, partly by preserving fibronectin, a structural protein that AEP normally clips, and by boosting pro-bone signals such as osteoprotegerin. In bone-resorbing cells, the drugs blunted the effects of a key resorption signal, reducing the number of large, multinucleated cells and their ability to carve pits into bone slices. In living mice, both #11a and CF3CN curbed the rapid bone turnover triggered by ovary removal, stabilizing bone density and microarchitecture. Strikingly, when #11a was compared head-to-head with teriparatide, an FDA-approved injectable that builds bone, the experimental pill matched teriparatide’s ability to restore bone volume and strength in this model.
What this could mean for future osteoporosis care
To a non-specialist, the takeaway is that this work identifies a double-duty pathway that links menopause-related inflammation to rising FSH and to bone damage, and shows that targeting AEP can interrupt this chain. In mice, an oral AEP inhibitor not only lowers the excess FSH that fuels bone loss but also directly shifts the balance inside bone toward building rather than breaking down. While these findings still need to be tested in people, they suggest that future osteoporosis treatments might move beyond simply replacing estrogen or stimulating bone formation, and instead quiet this newly mapped hormonal and enzymatic circuit that drives postmenopausal bone fragility.
Citation: Xie, Z., Liao, J., Xiong, J. et al. C/EBPβ dictates postmenopausal FSHβ transcription and blockade of AEP/C/EBPβ pathway alleviates osteoporosis. Bone Res 14, 31 (2026). https://doi.org/10.1038/s41413-026-00510-y
Keywords: postmenopausal osteoporosis, follicle-stimulating hormone, C/EBPβ AEP pathway, bone remodeling, hormone-targeted therapy