Clear Sky Science · en
Expansion of bone marrow adipocytes in obese mice leads to PD-L1-driven bone marrow immunosuppression and osteoclastogenesis
Why fat inside bones matters
Most people think of fat as something that sits under the skin or around the belly, but our bones also contain their own specialized fat. This study in mice reveals that when this bone marrow fat expands during obesity, it quietly reshapes the immune environment inside bones and speeds up the breakdown of bone itself. Understanding this hidden connection between body weight, immunity, and bone strength could help explain why people with obesity are more prone to fractures and may point toward new treatments that protect bones by targeting fat and immune pathways in the marrow.
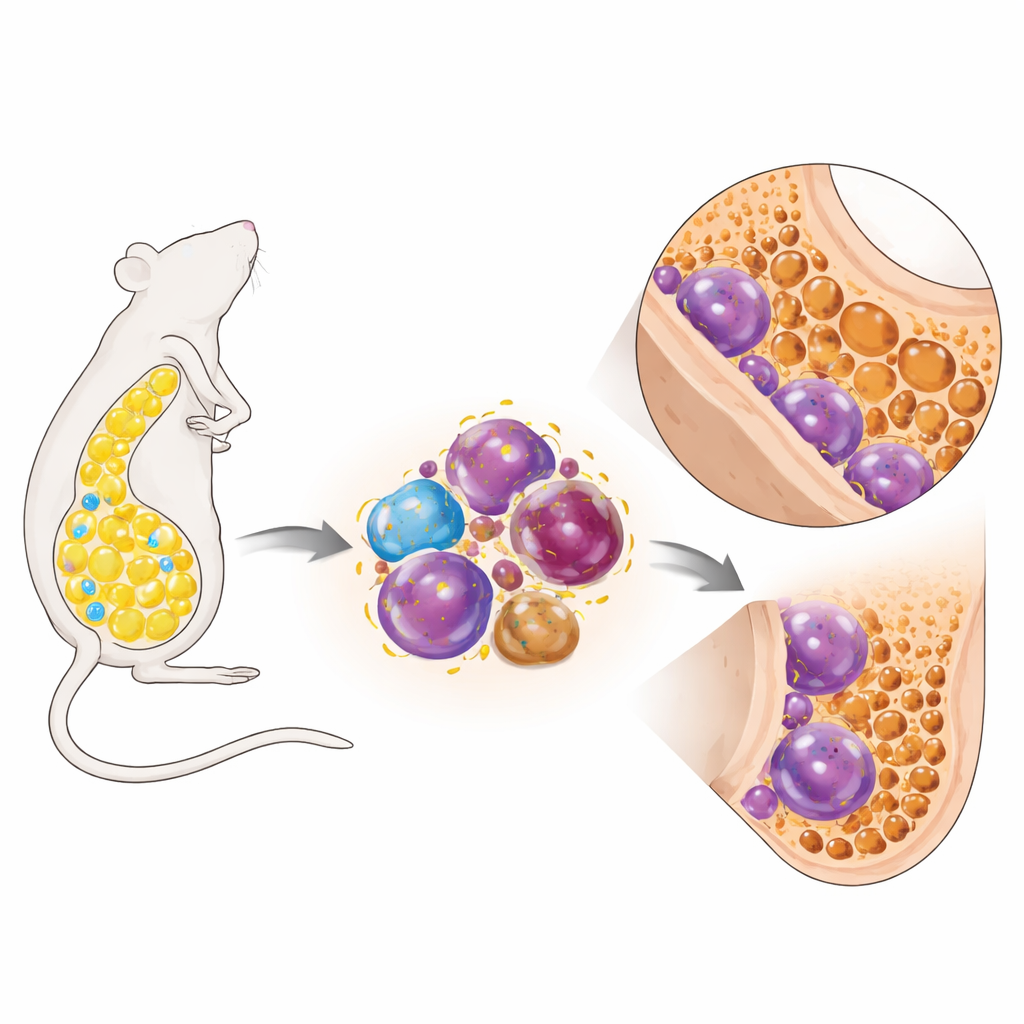
Obesity and weakening bones
Obesity was once thought to protect bones, because heavier bodies place more load on the skeleton. However, recent data show that people with obesity often have poorer bone quality and more fractures. In this study, researchers used mice fed a high‑fat diet and carefully defined which animals truly became obese based on large gains in body weight and fat mass. These obese mice lost a substantial amount of the spongy, inner part of their bones (trabecular bone) and, in males, also thinned the dense outer shell (cortical bone). The bone loss was tied to more and larger bone‑eating cells, called osteoclasts, and reduced performance of bone‑building osteoblasts—evidence that the normal balance of bone breakdown and formation had shifted in a harmful direction.
Bone marrow fat as more than a filler
Looking inside the bones, the team found that marrow fat cells expanded dramatically in obese mice—there were more of them and they were larger. Mice on the same diet that did not become obese showed far less marrow fat and had better bone volume, hinting that marrow fat itself might be driving damage. When the researchers grew bone marrow fat cells in dishes together with immature myeloid cells, which can turn into osteoclasts, the presence of fat cells from obese mice greatly boosted the number and size of mature osteoclasts. Surprisingly, the marrow fat did not look overtly inflamed. Instead, these adipocytes produced high amounts of a signal called MCP‑1, known to attract and expand certain myeloid cells and to promote their development into osteoclasts.
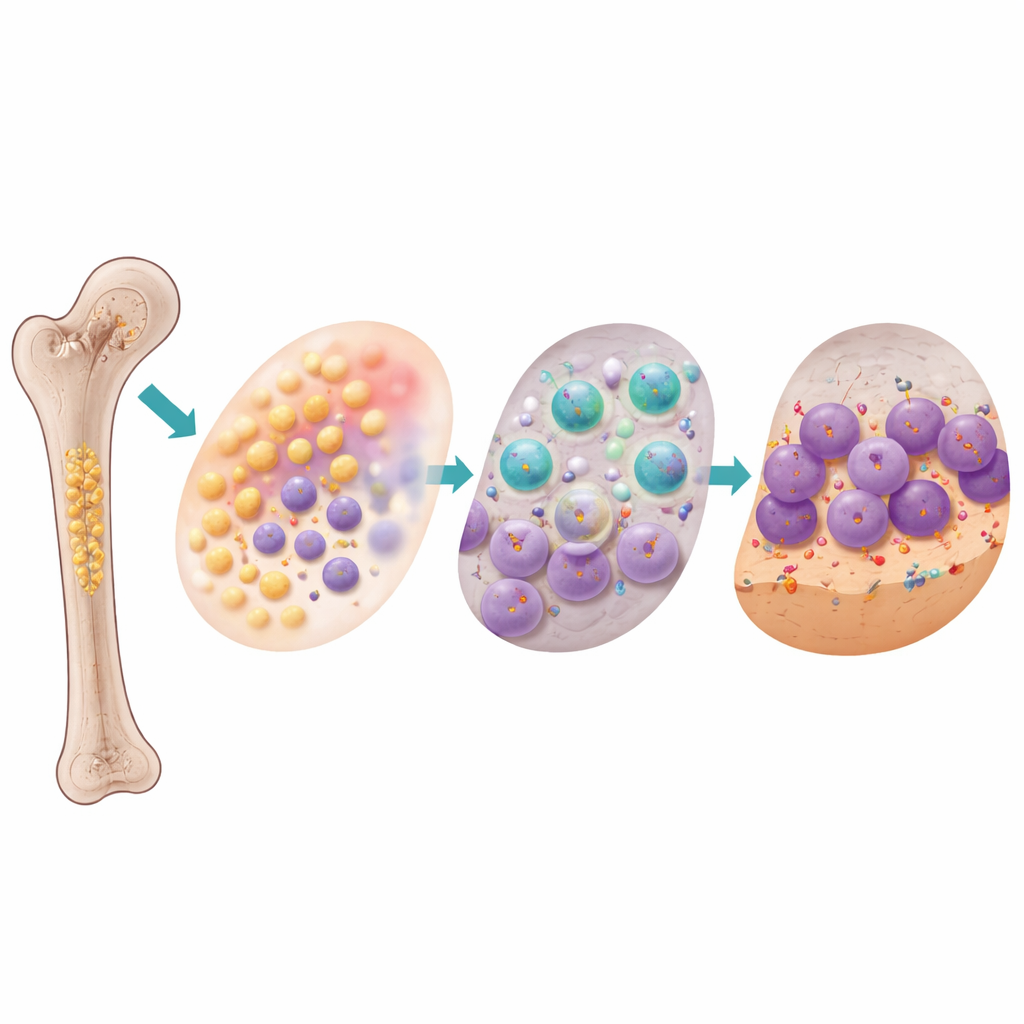
A quiet immune brake that fuels bone loss
The bone marrow is also an immune organ, and obesity changed its immune makeup in striking ways. Obese mice produced more myeloid progenitors and more mature myeloid cells bearing an immune “brake” protein on their surface called PD‑L1. At the same time, fewer helper T cells were present, and T cells that were exposed to these PD‑L1–rich myeloid cells stopped dividing, showing that the marrow environment had become immunosuppressed. Crucially, the researchers discovered a companion population of osteoclast precursor cells that carried PD‑1, the binding partner for PD‑L1. When PD‑1 and PD‑L1 interacted early during osteoclast development, they acted not as a brake but as a gas pedal: blocking this interaction in cell culture during the first days of differentiation sharply reduced osteoclast number, size, fusion, and expression of genes needed for bone resorption.
Turning off marrow fat to protect bone
To test whether marrow fat is truly upstream of these immune changes, the team used genetically engineered mice that cannot form normal, lipid‑filled marrow adipocytes but still become obese on a high‑fat diet. These marrow‑fat‑deficient obese mice had far fewer PD‑L1‑positive myeloid cells, fewer PD‑1‑bearing osteoclast precursors, and lower blood markers of bone resorption. Their trabecular bone volume was noticeably higher than in obese controls with intact marrow fat, and the inner bone structure was more plate‑like and supportive rather than thin and rod‑like. Bone‑forming osteoblasts were not boosted, but bone‑eating osteoclasts were clearly reduced, showing that simply preventing marrow fat expansion can blunt obesity‑related bone loss by easing the pro‑osteoclast pressure in the marrow immune environment.
What this means for bone health
Overall, the study shows that in obesity, expanding bone marrow fat cells help convert the marrow into an immunosuppressed niche rich in PD‑L1–expressing myeloid cells. These cells not only dampen T‑cell activity but also engage PD‑1 on osteoclast precursors, nudging them more strongly toward becoming bone‑resorbing osteoclasts. The resulting overactivity of osteoclasts erodes trabecular bone and can weaken cortical bone, increasing fracture risk. By blocking marrow fat formation or interfering with PD‑1/PD‑L1 signaling during key stages of osteoclast development, it may be possible to protect bone in obesity and in conditions like osteoporosis and certain cancers, where marrow fat and bone loss often go hand in hand.
Citation: Costa, S.N., Chlebek, C., Gray, L. et al. Expansion of bone marrow adipocytes in obese mice leads to PD-L1-driven bone marrow immunosuppression and osteoclastogenesis. Bone Res 14, 32 (2026). https://doi.org/10.1038/s41413-026-00509-5
Keywords: bone marrow fat, obesity and bone loss, immune checkpoints, osteoclasts, bone health