Clear Sky Science · en
TGF-β-induced fibrotic scar formation limits recovery of spinal cord injury
Why scars in the spinal cord matter
When the spinal cord is badly injured, many people are left with permanent paralysis or loss of sensation because damaged nerve fibers fail to grow back. This study asks a simple but crucial question: what, exactly, is blocking that regrowth—and can we safely remove the barrier? By uncovering how a particular type of scar forms inside the injured cord, the authors point to a new way to help the nervous system repair itself.
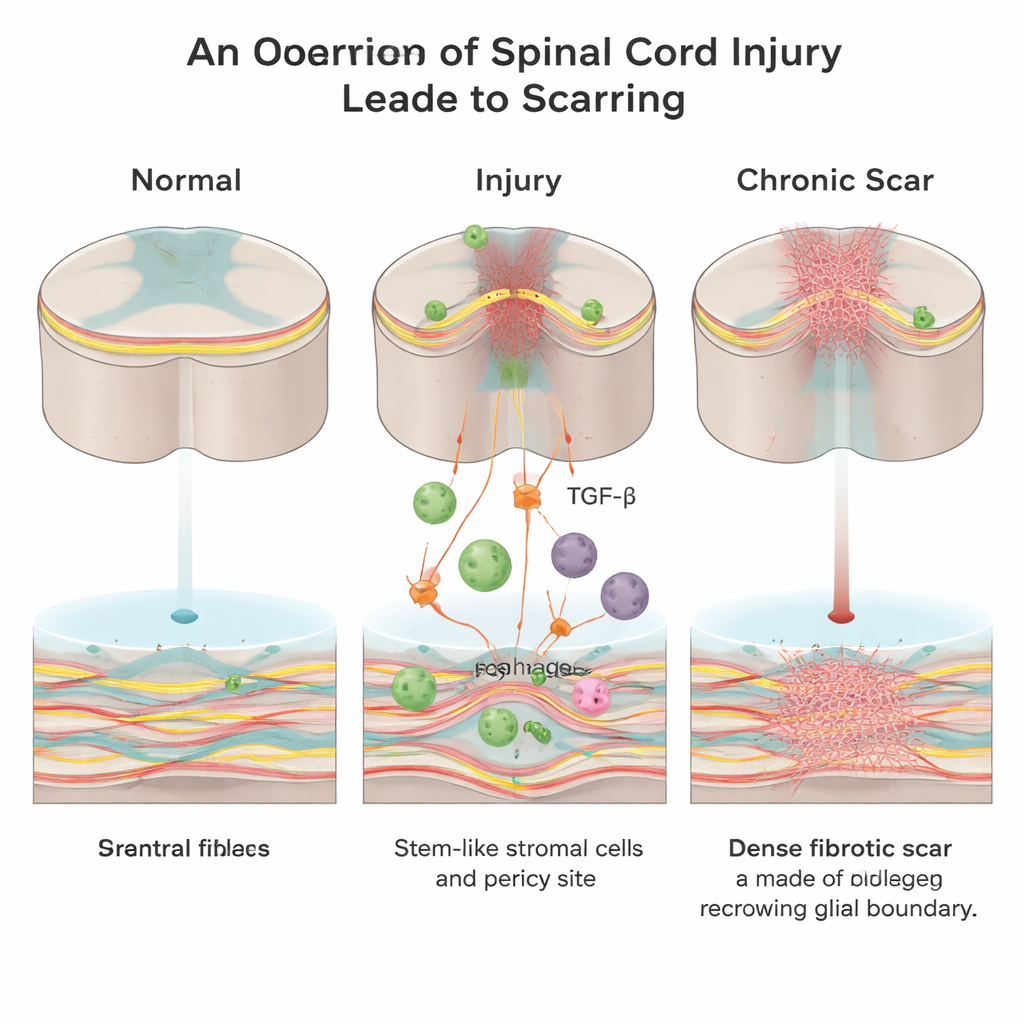
A hidden wall inside the injury
After a spinal cord injury, the body rushes to seal the wound. Two main kinds of scar tissue form: a “glial” scar made by support cells called astrocytes, and a “fibrotic” scar rich in collagen, fibronectin, and fibroblast cells. While the glial scar may have protective roles, the fibrotic scar creates a dense physical and chemical wall that growing nerve fibers cannot easily cross. Until now, scientists knew far less about how this fibrotic core appears and which signals drive its formation.
The role of immune cells and a powerful signal
Working in adult mice, the researchers focused on a signaling molecule called transforming growth factor‑beta (TGF‑β), which helps coordinate wound repair throughout the body and is often overactive in fibrotic diseases. They found that, after spinal cord injury, invading immune cells known as macrophages become a major source of active TGF‑β1 at the lesion site. This surge in TGF‑β1 recruits local mesenchymal stromal/stem cells and vessel‑associated cells called pericytes and pushes them to turn into fibroblasts—the very cells that lay down the collagen‑rich fibrotic scar.
Turning down the signal to open a path
To test whether this pathway truly causes harmful scarring, the team used several genetic tricks in mice. When they removed macrophages, or deleted the TGF‑β1 gene only in macrophage‑lineage cells, the amount of fibrotic tissue in the injured cord dropped sharply, and more nerve fibers and serotonin‑containing pathways were able to cross the injury zone. Likewise, deleting the TGF‑β receptor specifically in pericytes reduced their response to TGF‑β, led to less collagen build‑up, and improved movement and sensation on behavioral tests. Importantly, pericytes and related stem‑like cells still existed but were less likely to become scar‑forming fibroblasts.
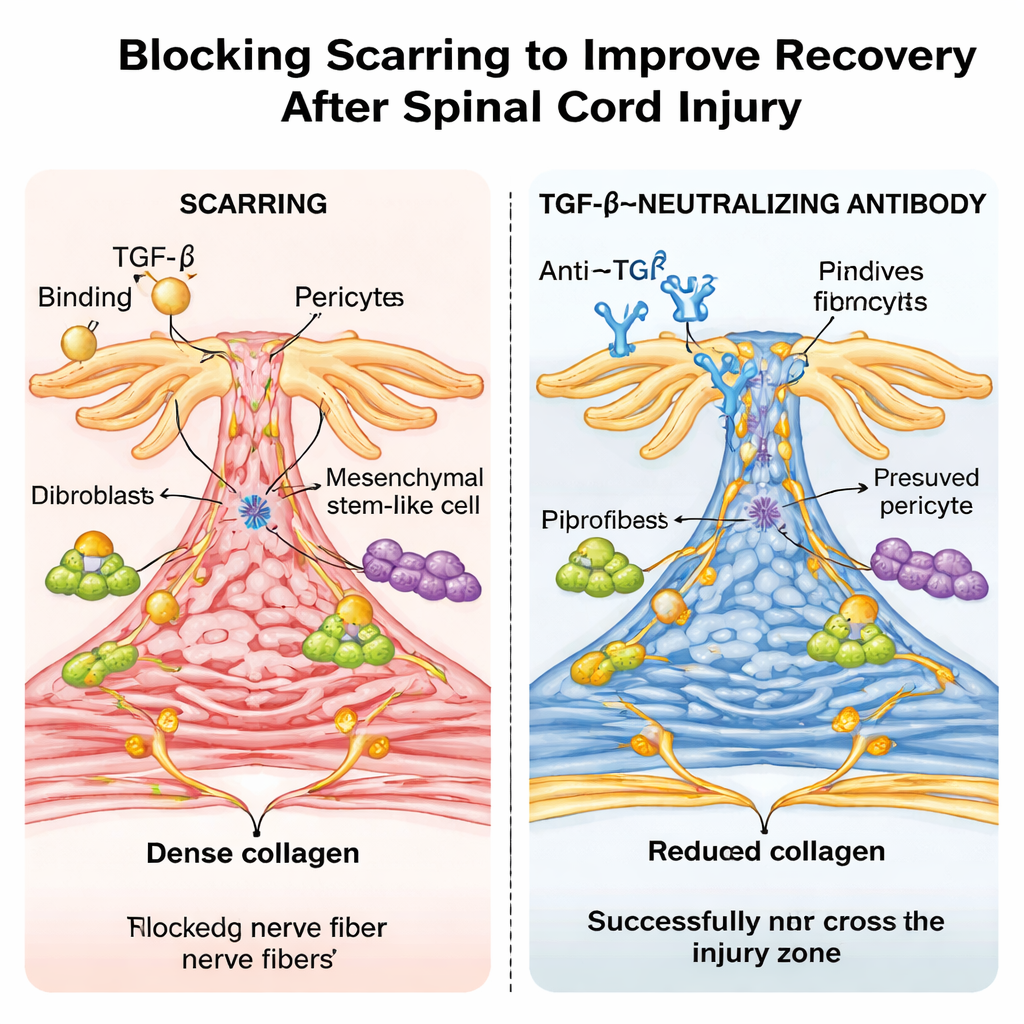
A drug-like antibody and a surprising age effect
The researchers then tried a more clinically relevant approach: treating ordinary mice with a laboratory antibody that neutralizes TGF‑β. Repeated doses after injury lowered active TGF‑β levels in blood and spinal cord, shrank the fibrotic scar, and allowed nerve fibers to bridge the lesion more effectively, with marked gains in walking and sensory function. Single‑cell RNA sequencing confirmed that TGF‑β‑responsive genes were highly active in scar‑forming cell types in untreated mice and were tamped down by the antibody. Strikingly, when the same type of spinal cord crush was performed in newborn mice, the animals healed almost without fibrotic scarring, showed no detectable TGF‑β activation at the injury site, and recovered near‑normal movement—resembling the scar‑free repair seen in some non‑mammalian animals.
What this could mean for future treatment
Taken together, the findings suggest that excessive activation of TGF‑β after spinal cord injury diverts helpful repair cells into building a rigid fibrotic wall that blocks nerve regrowth and functional recovery. By preventing this overactivation—either by targeting TGF‑β production in macrophages, blocking its receptor on pericytes and stromal cells, or using a neutralizing antibody—it may be possible to reduce the bad scar while preserving or even enhancing more beneficial types of tissue response. Although translating these strategies to humans will require careful study to avoid side effects, this work highlights TGF‑β‑driven fibrotic scarring as a central, and potentially druggable, barrier to spinal cord repair.
Citation: Pan, D., Wu, P., Noller, K. et al. TGF-β-induced fibrotic scar formation limits recovery of spinal cord injury. Bone Res 14, 27 (2026). https://doi.org/10.1038/s41413-026-00507-7
Keywords: spinal cord injury, fibrotic scar, TGF-beta, macrophages, nerve regeneration