Clear Sky Science · en
HIF-1α and BMAL1 in bone regeneration: crosstalk between hypoxia response and circadian rhythm
Why Broken Bones Heal Better Than You Think
When we break a bone or have a tooth pulled, our bodies quietly launch a remarkably well‑orchestrated repair project. Deep inside the injured area, oxygen levels plunge and our internal day–night clock keeps ticking. This article explores how two master switches—HIF‑1α, which senses low oxygen, and BMAL1, a core clock protein—work together to guide bone cells through inflammation, new tissue growth, and final strength restoration. Understanding this partnership could improve treatments for fractures, osteoporosis, arthritis, and even dental implants.
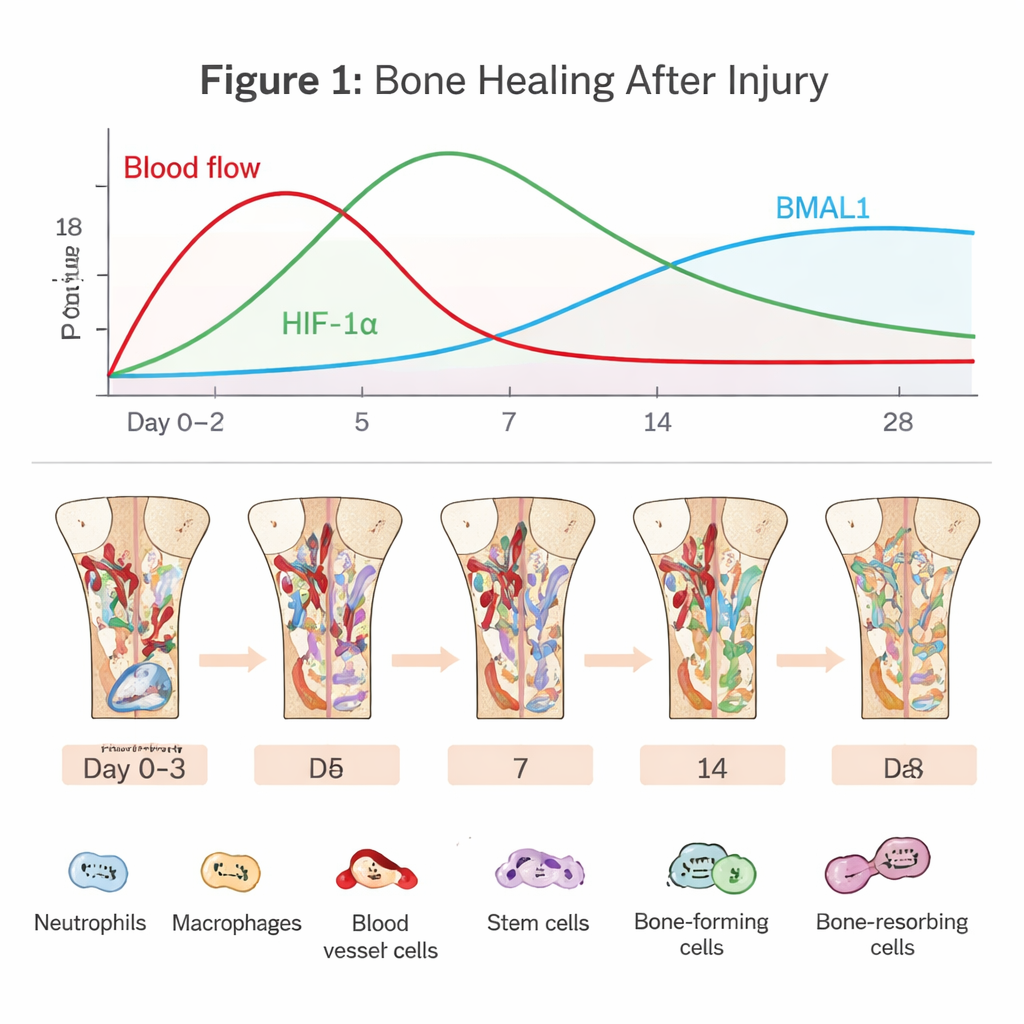
The Hidden Drama Inside a Healing Bone
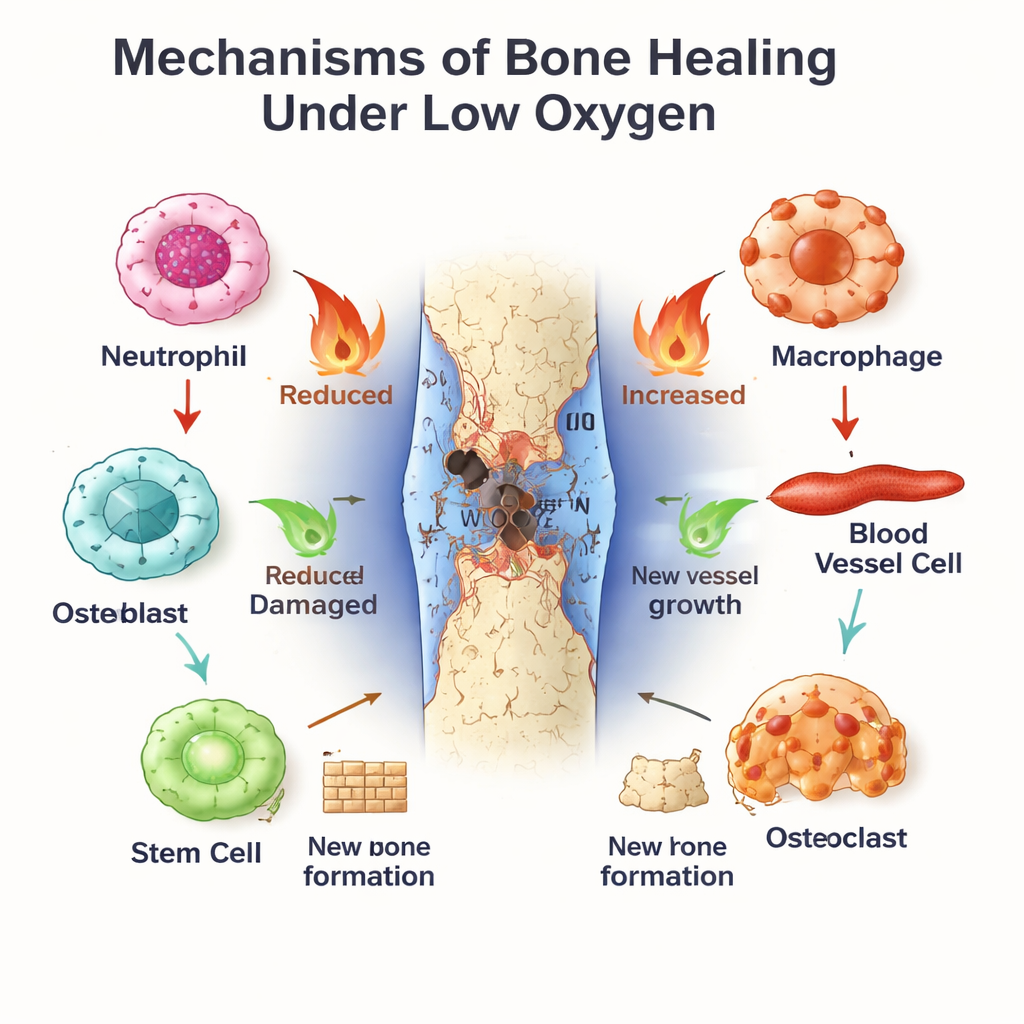
Bone regeneration begins the moment blood vessels tear. A clot forms, cutting off much of the oxygen supply and creating a harsh, low‑oxygen pocket. Immune cells such as neutrophils and macrophages rush in to clean up debris and fend off microbes. At the same time, bone marrow stem cells and blood vessel cells are recruited to rebuild the area. This early “inflammatory phase” is a double‑edged sword: a short, well‑timed burst of inflammation kick‑starts repair, but if it lasts too long or burns too hot, healing can stall or fail. The article emphasizes that navigating this narrow path depends heavily on how cells sense oxygen and time.
How Cells Use Low Oxygen to Their Advantage
In the oxygen‑poor fracture zone, the protein HIF‑1α stabilizes and switches on genes that help cells adapt. It pushes endothelial cells to sprout new blood vessels, ensuring fresh oxygen and nutrients reach the defect. It also reprograms cell metabolism toward glycolysis, a way of generating energy that works even when oxygen is scarce. For stem cells and bone‑forming osteoblasts, moderate activation of HIF‑1α can boost proliferation, migration, and bone growth, partly by increasing factors like VEGF that link blood vessel formation to new bone. But if hypoxia is too severe or prolonged, stress molecules and reactive oxygen species build up, tipping osteoblasts toward death and stimulating bone‑resorbing osteoclasts. The same hypoxic signal can therefore either support or sabotage healing, depending on its intensity and duration.
The Body’s Internal Clock as a Bone Repair Manager
Running in parallel is the circadian clock, a molecular timekeeper present in nearly every cell. BMAL1, one of its central components, helps set daily rhythms in immune activity, blood vessel behavior, and stem cell differentiation. Neutrophils and macrophages show time‑of‑day differences in how strongly they respond to injury and how quickly they age or switch from inflammatory (M1‑like) to healing (M2‑like) states. In blood vessel cells and bone marrow stem cells, BMAL1 promotes healthy proliferation and orderly bone formation, in part by tuning growth pathways such as Wnt and TGF‑β/SMAD. When BMAL1 is disrupted—by gene defects, shift work, or metabolic disease—bones tend to lose mass, stem cells tire out, and the balance between bone formation and breakdown skews toward loss.
A Conversation Between Oxygen Sensing and the Body Clock
The heart of the review is the emerging “crosstalk” between HIF‑1α and BMAL1. These two proteins share similar structures and can physically interact, even forming mixed pairs that bind DNA and control overlapping sets of genes. Each can influence the other’s production and stability, creating feedback loops that tie the timing of the day–night cycle to the way cells respond to hypoxia. Together they also manage cellular redox balance—how much damaging reactive oxygen species are produced versus how much antioxidant capacity is available—and they co‑regulate key enzymes that decide whether cells rely more on oxygen‑dependent respiration or oxygen‑free glycolysis. This joint control helps determine whether the fracture environment favors constructive rebuilding or excessive inflammation and bone resorption.
What This Means for Disease and Treatment
The same HIF‑1α–BMAL1 partnership shows up in many bone‑related conditions. In shift workers, disturbed sleep and light exposure likely alter BMAL1, increase oxidative stress, and interfere with HIF‑1α‑guided repair, contributing to osteoporosis. In diabetes, high blood sugar weakens both clock function and hypoxia signaling, undermining blood vessel growth and stem‑cell‑driven bone formation. In osteoarthritis, loss of BMAL1 in cartilage cells disrupts their rhythm and their protective relationship with HIF‑1α, speeding cartilage breakdown. Around titanium implants, controlled hypoxia and a healthy circadian rhythm appear to support better osseointegration, suggesting that future implant coatings might be designed to gently activate both HIF‑1α and BMAL1. Overall, the article concludes that successful bone regeneration depends not just on which cells are present, but on how they read oxygen and time—and that targeting this dialogue could open new avenues to faster, stronger healing.
Citation: Weng, Y., Xiong, J., Zhao, Q. et al. HIF-1α and BMAL1 in bone regeneration: crosstalk between hypoxia response and circadian rhythm. Bone Res 14, 25 (2026). https://doi.org/10.1038/s41413-026-00506-8
Keywords: bone regeneration, circadian rhythm, hypoxia, osteoporosis, fracture healing