Clear Sky Science · en
Smart biomaterials for skeletal aging repair and regeneration
Stronger Bones in an Aging World
Many people expect weaker bones and joint pain as unavoidable parts of growing older. But scientists are now designing “smart” materials that can sense when and where bones are failing and then help them heal themselves. These materials aim to repair fractures more reliably, slow or reverse osteoporosis and osteoarthritis, and reduce the need for repeated surgeries—offering older adults a better chance at staying active and independent.
How Bones Age and Why They Break More Easily
Bone is a living tissue that constantly rebuilds itself, but with age this balance falters. Stem cells inside bone marrow shift from making bone-forming cells to making more fat cells; bone-building cells tire and bone-eating cells become overactive. The supporting matrix of collagen and mineral becomes drier and more brittle, and the microscopic “scaffolding” thins out. Hormone changes, chronic low-grade inflammation, excess reactive oxygen molecules, and an acidic local environment all add stress. On the outside, this shows up as thinner, more porous bone, stiffer joints, curved spines, and a higher risk of fractures and conditions like osteoporosis and osteoarthritis.
Why Current Treatments Are Not Enough
Today’s main treatments include drugs that slow bone loss or stimulate bone formation, along with surgeries and metal or ceramic implants to stabilize fractures or replace damaged joints. These approaches have saved many lives but have important drawbacks. Systemic drugs often reach less than a tiny fraction of bone tissue and can cause side effects such as jaw problems, blood clots, or even increased cancer risk with long-term use. Stem cell therapies show promise in animals but face issues of cell survival, unpredictable behavior, and potential tumor formation. Conventional implants provide strength but are “dumb” objects: they do not sense their surroundings, cannot adjust drug release, and may fail in fragile, elderly bone.
Smart Drug Carriers That Patrol the Body
Smart biomaterials bring intelligence into this picture. One major strategy is systemic “smart delivery” of medicines using nanoparticles or soft gels that circulate in the bloodstream and only activate in problem spots. These carriers can be built to respond to internal signals such as acidity, high levels of damaging oxygen species, or bone-dissolving enzymes; or to external cues like light, ultrasound, heat, or magnetic fields. For example, some particles stay intact in normal tissue but open and release their drug cargo in the acidic, enzyme-rich pockets where overactive bone-resorbing cells are chewing away bone. Others scavenge excess reactive oxygen while slowly releasing an anti-inflammatory medicine, or carry imaging dyes so doctors can see where they go in real time. By stacking multiple triggers—say, both pH and oxidants—researchers can sharpen targeting and reduce side effects.
Local Scaffolds That Act Like Temporary Smart Bones
When aging bones suffer large defects or complex fractures, local “smart scaffolds” can be implanted directly into the gap. These 3D structures, made from metals, ceramics, polymers, or combinations, are engineered to match bone strength, porosity, and flexibility. Their sponge-like architecture lets blood vessels and new bone grow in, while the material gradually dissolves and is replaced by the patient’s own tissue. 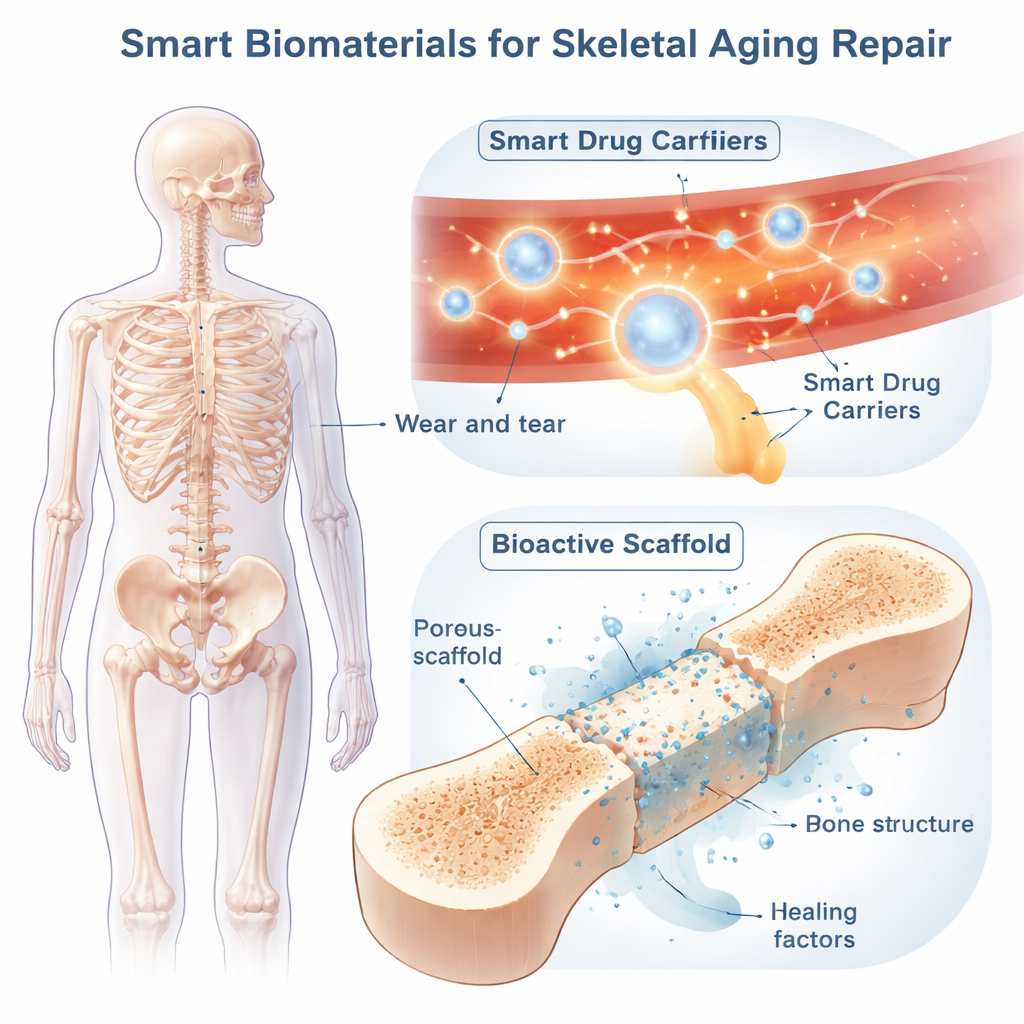
From Lab to Clinic: Tools, Hurdles, and Future Directions
To make these intricate systems, scientists use advanced manufacturing such as 3D printing and additive manufacturing to tune pore size, stiffness, and drug-loading with high precision. Yet, turning smart biomaterials into routine treatments is challenging. Most injected particles still accumulate in organs like the liver and spleen rather than bone, and many animal models fail to capture the complexity of aging humans. Large-scale production, quality control, regulatory approval, and cost all remain major hurdles. Researchers see hope in combining smart materials with artificial intelligence to personalize scaffold design and drug dosing, as well as in adding built-in sensors that can track healing and automatically adjust therapy in “closed-loop” fashion. 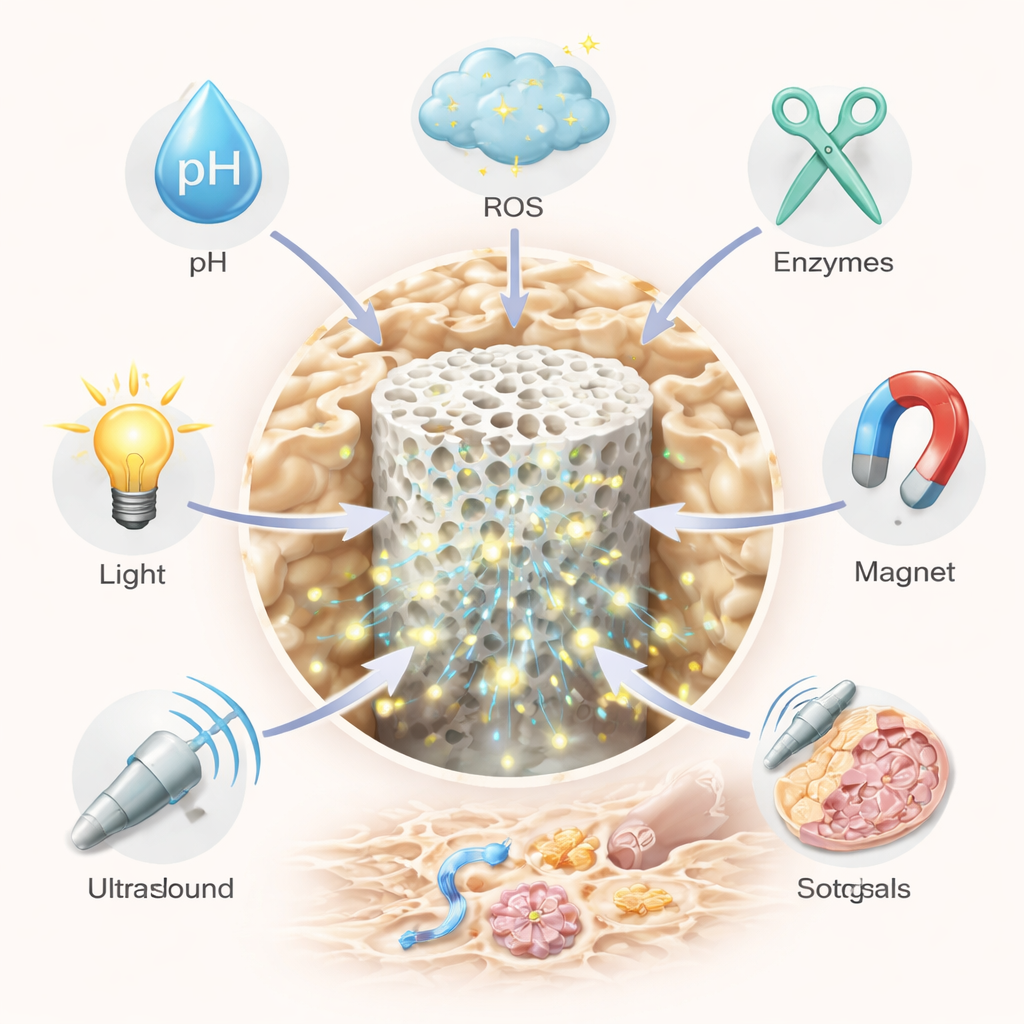
What This Could Mean for Patients
In essence, this work shows that bone fragility in old age is not simply a one-way decline but a complex engineering problem that may be solvable. Smart biomaterials act like tiny, programmable repair crews: patrolling the bloodstream, settling into damaged sites, and releasing exactly what is needed, when it is needed, to restore bone structure and function. Although these technologies are still largely in the research phase, they point toward a future in which fractures heal faster, implants last longer, and therapies are tailored to each person’s unique bone biology—helping more people stay mobile and independent well into advanced age.
Citation: Liang, D., Wang, H., Jiang, Y. et al. Smart biomaterials for skeletal aging repair and regeneration. Bone Res 14, 24 (2026). https://doi.org/10.1038/s41413-026-00505-9
Keywords: smart biomaterials, skeletal aging, bone regeneration, osteoporosis, stimuli-responsive scaffolds