Clear Sky Science · en
Synovial inflammatory macrophage-derived extracellular vesicles exacerbate cartilage lesions with a FMRP-selectively sorted manner in osteoarthritis
Why joint inflammation matters for aching knees
Many people think of osteoarthritis as simple “wear and tear” of aging joints, but mounting evidence shows that low‑grade inflammation inside the joint can quietly push the disease forward. This study asks a pointed question: how do immune cells in the joint lining talk to cartilage cells, and can that conversation be interrupted to protect joints? The answers reveal a tiny molecular courier system that worsens damage—but also offers a new way to diagnose and treat osteoarthritis.
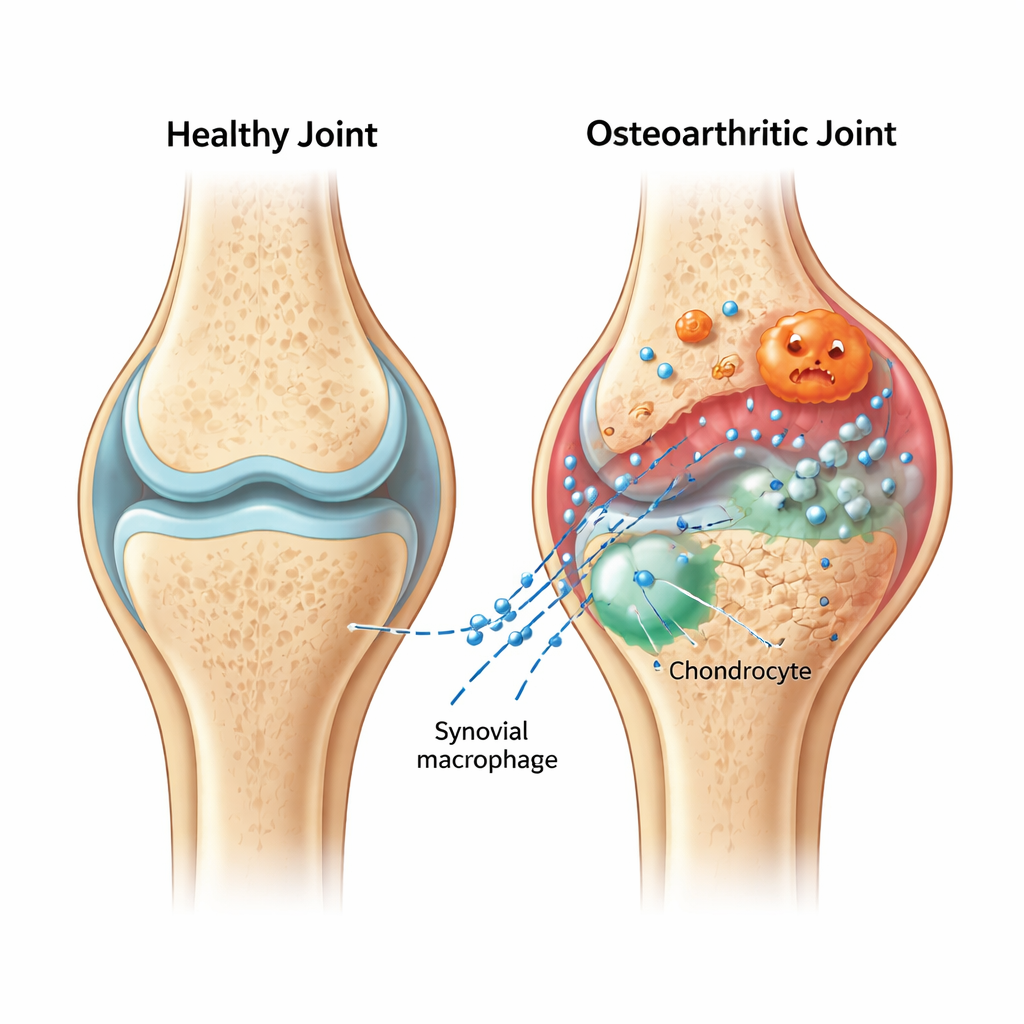
Tiny packages that carry bad news
Inside an arthritic knee, the soft synovial lining becomes inflamed and fills with immune cells called macrophages. The authors focused on the “hot‑headed” version of these cells—pro‑inflammatory macrophages—and the microscopic bubbles they release, known as extracellular vesicles. These vesicles act like sealed packages, shuttling molecules between cells. In rats with surgically induced osteoarthritis, the team showed that vesicles from inflamed macrophages home to the cartilage and make damage worse: the normally smooth, cushiony surface became rougher and thinner, and key cartilage proteins declined while breakdown enzymes increased.
How inflammation shuts down cartilage self‑cleaning
Cartilage cells, or chondrocytes, rely on a housekeeping process called autophagy to recycle worn‑out components and stay healthy over a lifetime. When the researchers compared genes turned on in cartilage exposed to inflammatory vesicles versus a harmless salt solution, they saw strong signs that autophagy pathways were being suppressed. Under the electron microscope, cartilage from vesicle‑treated joints contained far fewer of the characteristic recycling vesicles. In cell cultures, chondrocytes exposed to macrophage vesicles also formed fewer autophagosomes and autolysosomes, confirming that their self‑cleaning machinery was being switched off just as damage accumulated.
A single microRNA flips a critical switch
Diving deeper, the team examined the tiny RNA regulators packed inside macrophage vesicles. One molecule, miR‑155‑5p, stood out: it was enriched in vesicles from osteoarthritic animals and in human osteoarthritis joint tissues, especially in the synovium. Experiments showed that vesicle‑delivered miR‑155‑5p enters chondrocytes and directly targets a protein called GSK‑3β, which normally helps restrain a growth‑control pathway known as mTORC1. When GSK‑3β is suppressed, mTORC1 becomes overactive and autophagy falls, leaving cartilage cells less able to cope with stress. Blocking miR‑155‑5p with a designer “antagomiR” in rats restored autophagy, preserved cartilage structure, and reduced signs of osteoarthritis, while mice engineered to lack miR‑155 in their macrophages developed milder disease and less synovitis.
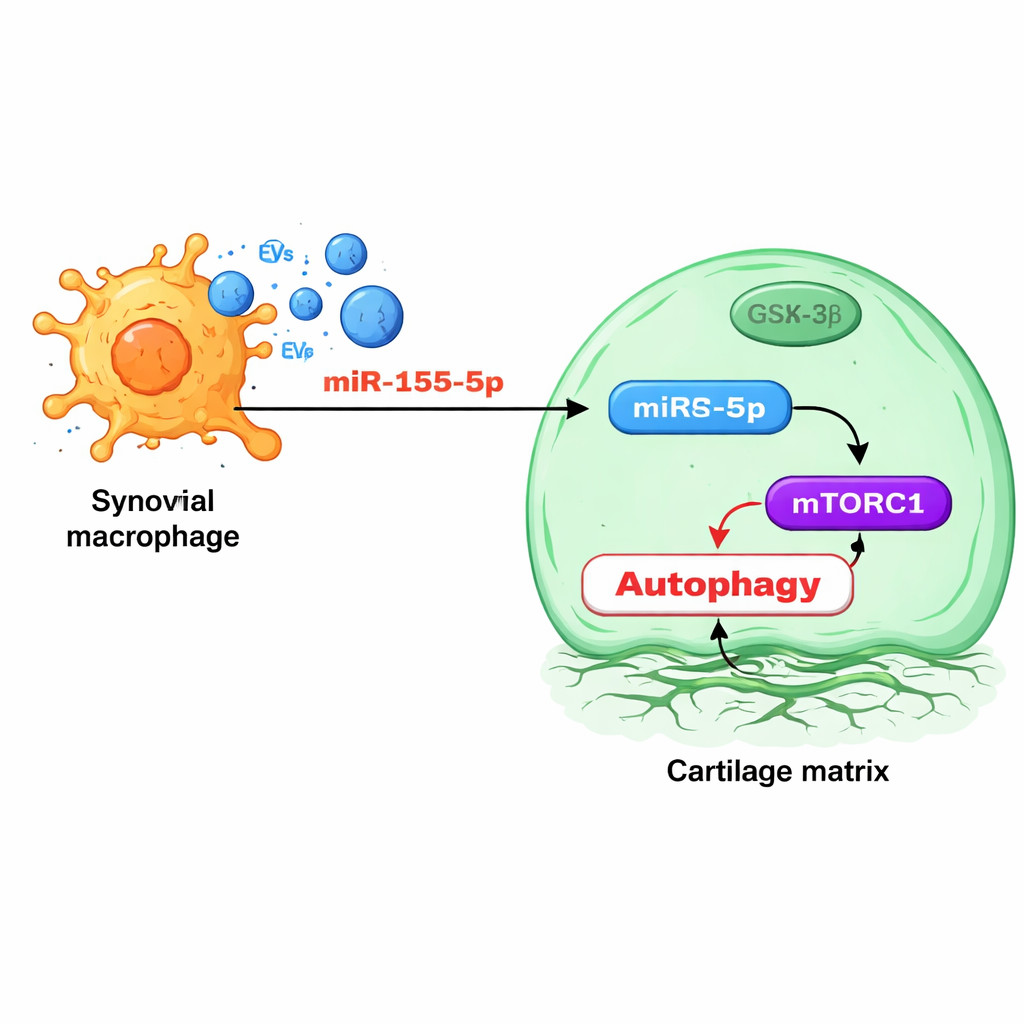
Sorting and targeting the damaging signal
Intriguingly, the study found that macrophages use a specific RNA‑binding protein, FMRP, to selectively load miR‑155‑5p into vesicles. When FMRP levels were increased, vesicles carried more miR‑155‑5p and harmed cartilage cells more; when FMRP was reduced, vesicles became less toxic and chondrocyte autophagy rebounded. Levels of FMRP in vesicles circulating in the blood rose alongside osteoarthritis progression in both rats and human patients, suggesting that this protein, or the vesicles carrying it, could serve as a blood‑based marker of disease activity that does not require imaging or invasive procedures.
A cell‑free therapy that homes to the right cells
Building on these mechanistic insights, the researchers engineered a potential therapy using vesicles from adipose‑derived stromal cells, a type of stem‑like cell found in fat tissue. They decorated these vesicles with a small peptide that acts like a homing device for pro‑inflammatory macrophages, and loaded them with antagomiR‑155‑5p. In osteoarthritic rats, repeated injections of these targeted vesicles reduced synovial inflammation, shifted macrophages toward a more “healing” state, lowered miR‑155‑5p levels in the joint, restored GSK‑3β, and improved cartilage structure and composition more strongly than untargeted vesicles. When applied to human synovial tissue samples in the lab, the engineered vesicles also reduced inflammatory markers and boosted anti‑inflammatory signals.
What this means for people with osteoarthritis
This work recasts osteoarthritis as a disease driven not just by mechanical wear, but by a molecular dialogue between inflamed joint‑lining cells and cartilage. Pro‑inflammatory macrophages send out vesicles loaded with miR‑155‑5p, which turn down cartilage cells’ self‑repair systems through the GSK‑3β/mTORC1 pathway and hasten tissue breakdown. By blocking this signal—either genetically or with targeted therapeutic vesicles—the researchers were able to ease both inflammation and cartilage damage in animal models and human tissues. While much work remains before such strategies reach the clinic, the study highlights new possibilities: blood tests based on vesicle cargo to track disease, and precise, cell‑free treatments that calm harmful inflammation while helping cartilage protect itself.
Citation: Zhao, S., Wang, J., Xue, M. et al. Synovial inflammatory macrophage-derived extracellular vesicles exacerbate cartilage lesions with a FMRP-selectively sorted manner in osteoarthritis. Bone Res 14, 26 (2026). https://doi.org/10.1038/s41413-025-00502-4
Keywords: osteoarthritis, extracellular vesicles, macrophages, cartilage, microRNA-155