Clear Sky Science · en
Efficacy of stem cell boost (SCB) for chimeric antigen receptor-T cell therapy (CAR-T)-related hematologic toxicity in patients with relapsed/refractory multiple myeloma (RRMM)—real world experience from the US multiple myeloma immunotherapy consortium
Why this matters for patients and families
For people with difficult-to-treat multiple myeloma, CAR-T cell therapy has been a major breakthrough, but it can leave the bone marrow struggling to make new blood cells. This study looks at whether giving patients a “boost” of their own previously collected stem cells can safely help their blood counts recover faster, potentially reducing infections, bleeding, hospital visits, and the stress that comes with prolonged weakness and transfusions.
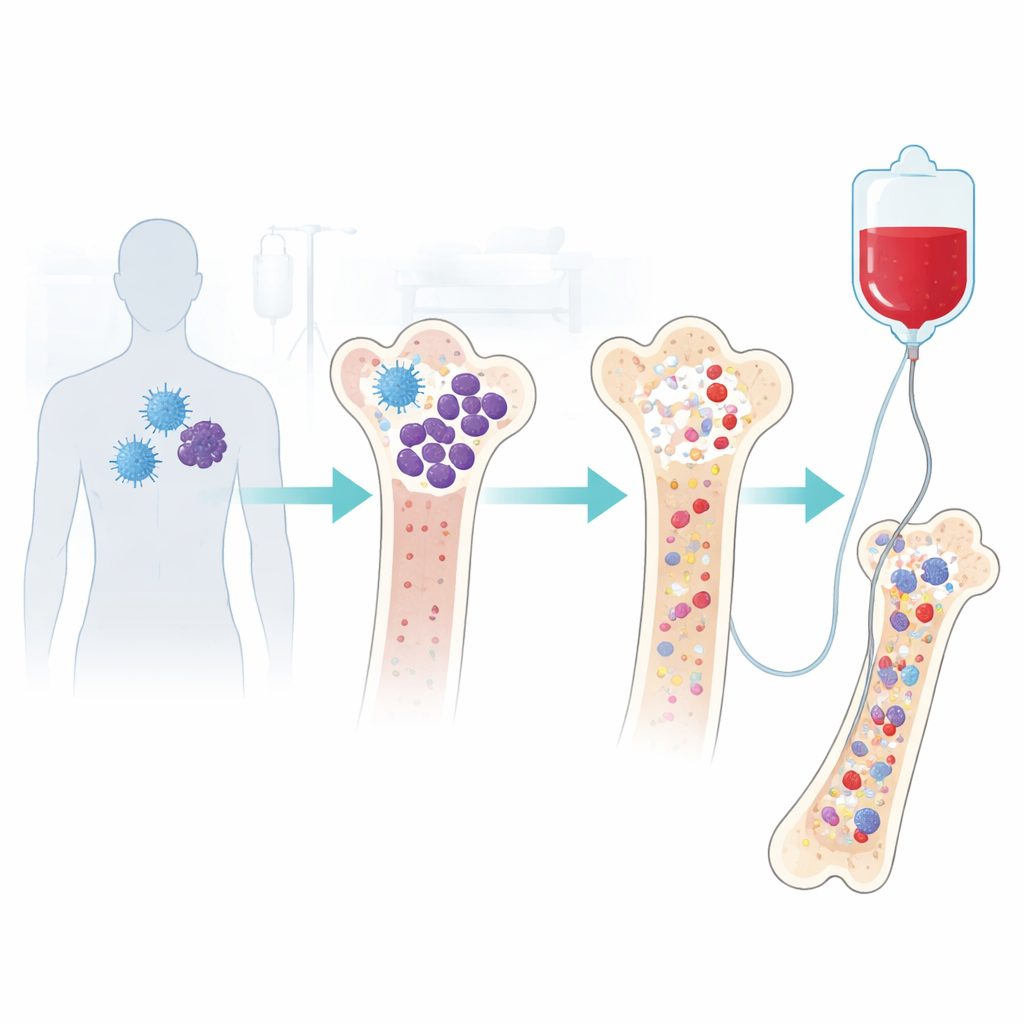
A powerful treatment with a lingering downside
CAR-T cell therapy reengineers a patient’s own immune cells to hunt down and destroy myeloma cells. While often effective, this intense treatment can leave the bone marrow—our internal blood factory—temporarily damaged. Many patients see their blood counts recover within one to three months. But a sizeable minority develop prolonged “cytopenias,” meaning dangerously low levels of white cells, red cells, and platelets. This raises the risk of serious infections, fatigue, and bleeding, and can tie patients to frequent transfusions and growth-factor shots. Doctors have needed better ways to help these patients recover without blunting the benefits of CAR-T.
A real-world test of a simple idea
The researchers examined 590 adults with relapsed or refractory multiple myeloma treated with commercial CAR-T products across 19 U.S. cancer centers. Ninety-one developed prolonged low blood counts; of these, 39 received an autologous stem cell boost and 52 received standard supportive care alone. The boost used stem cells that had been collected and frozen earlier, often when patients underwent a prior stem cell transplant. No extra chemotherapy was given before the boost. To make the comparison fair, control patients were chosen because they had similarly severe low white cells and platelets at 30 days after CAR-T.
Faster, more complete blood recovery
In the stem cell boost group, nearly every evaluable patient (97.4%) achieved blood count recovery based on standard transplant criteria. After the boost, neutrophils (infection-fighting white cells) typically recovered in about 13 days, platelets in 18 days, and hemoglobin in 24 days. By 90 days after CAR-T, patients who received the boost had clearly better blood counts than matched patients who did not: higher hemoglobin and far higher platelets, with a trend toward better white cell counts as well. These benefits appeared even when focusing only on the sickest subset within the boost group, suggesting the effect was not just due to milder cases being selected.
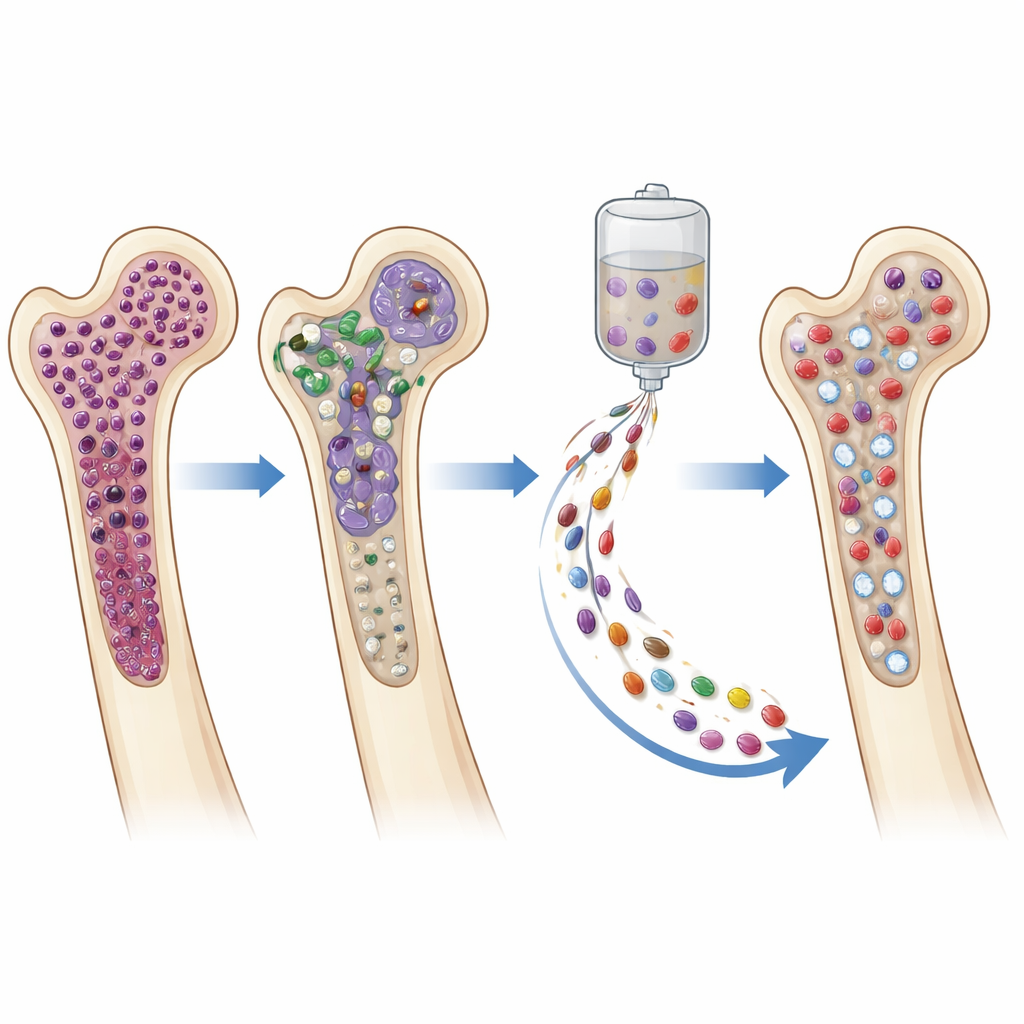
Safety, infections, and cancer control
Importantly, the stem cell boost did not introduce new safety problems. No complications were directly attributed to the boost itself, and serious infections in the boost group happened before, not after, the stem cells were given. A few patients had complex immune-related issues tied to CAR-T therapy, but there was no signal that the boost worsened these problems in this cohort. Just as crucial, the extra stem cells did not appear to weaken the anti-cancer effect of CAR-T. Rates of deep remission and “minimal residual disease” negativity were similar between groups, and survival outcomes were at least as good in the boost group, recognizing that patients had to live long enough to receive the boost.
What this could mean for future care
For patients, the take-home message is that having their own stem cells saved can offer a powerful rescue option if blood counts stall after CAR-T therapy. In this large real-world series, a stem cell boost—given without additional chemotherapy—was safe and led to rapid, reliable recovery of blood production in almost all treated patients, while preserving the cancer-fighting benefits of CAR-T. The authors suggest practical criteria for when to consider a boost, such as persistent transfusion needs beyond 30 days and very low counts that show no sign of improving. Prospective studies are still needed, but these findings support making stem cell collection and storage a routine part of planning for CAR-T in high-risk myeloma.
Citation: Varga, C., Robinson, M., Davis, J.A. et al. Efficacy of stem cell boost (SCB) for chimeric antigen receptor-T cell therapy (CAR-T)-related hematologic toxicity in patients with relapsed/refractory multiple myeloma (RRMM)—real world experience from the US multiple myeloma immunotherapy consortium. Blood Cancer J. 16, 39 (2026). https://doi.org/10.1038/s41408-026-01469-z
Keywords: CAR-T therapy, multiple myeloma, stem cell boost, bone marrow recovery, hematologic toxicity