Clear Sky Science · en
Immune escape mechanisms and therapeutic advances in virus-associated hematological malignancies
Viruses and Hidden Cancers in the Blood
Most of us think of viruses as causes of short‑term illnesses like colds or flu. But a handful of viruses can quietly take up long‑term residence in our bodies and, years later, help trigger serious blood cancers such as lymphomas and leukemias. This review article pulls together what scientists now know about how these viruses dodge our immune defenses and how that knowledge is opening the door to more precise, less toxic treatments.
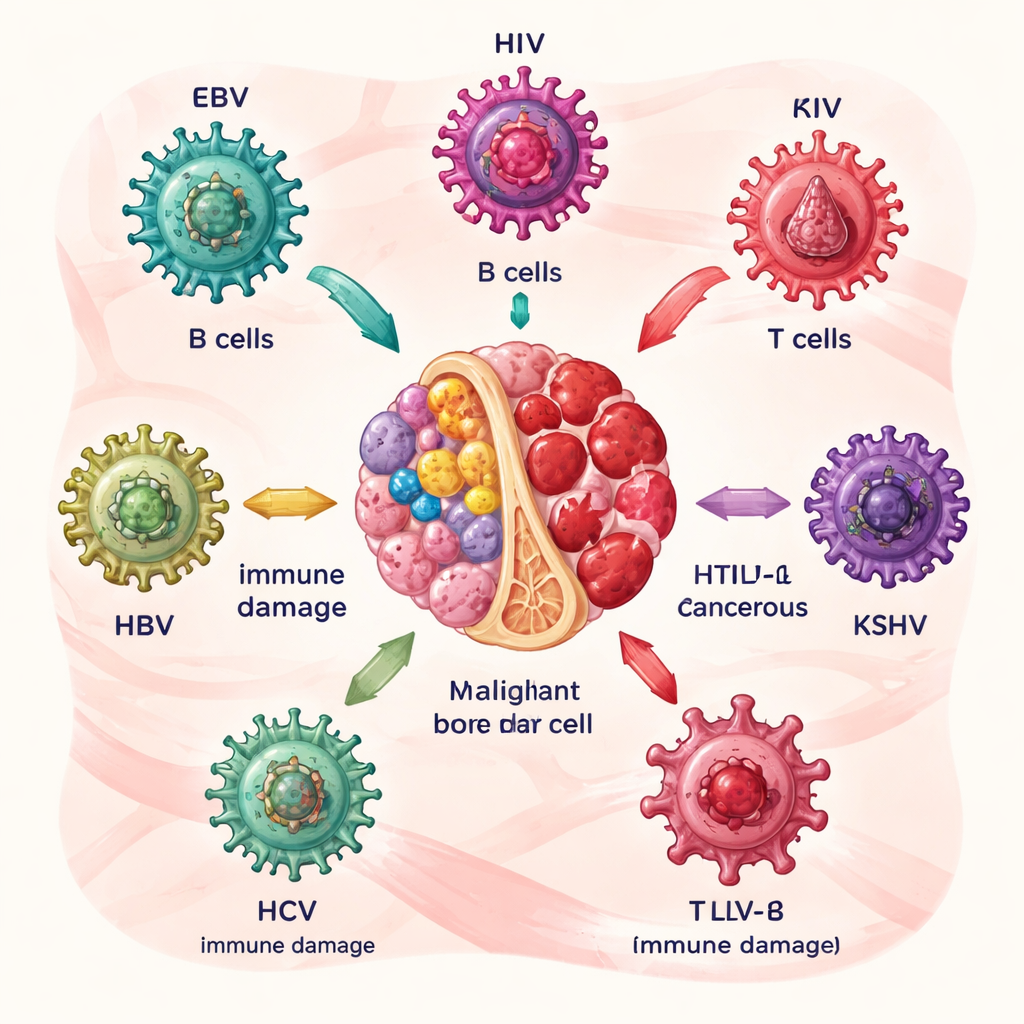
How Certain Viruses Set the Stage for Blood Cancer
The authors first describe six major culprits: Epstein–Barr virus (EBV), human immunodeficiency virus (HIV), human T‑cell leukemia virus type 1 (HTLV‑1), Kaposi’s sarcoma–associated herpesvirus (KSHV), and the hepatitis B and C viruses (HBV and HCV). Each targets blood or immune cells in its own way. EBV and HCV mainly disturb B cells, the antibody‑producing cells; HTLV‑1 attacks T cells; HIV weakens the overall immune system; KSHV promotes abnormal growth of certain white blood cells; and HBV and HCV can lurk in lymphocytes and stem cells. Over years, viral proteins push infected cells to multiply, avoid normal cell death, and accumulate DNA damage, gradually raising the risk of lymphomas and related diseases.
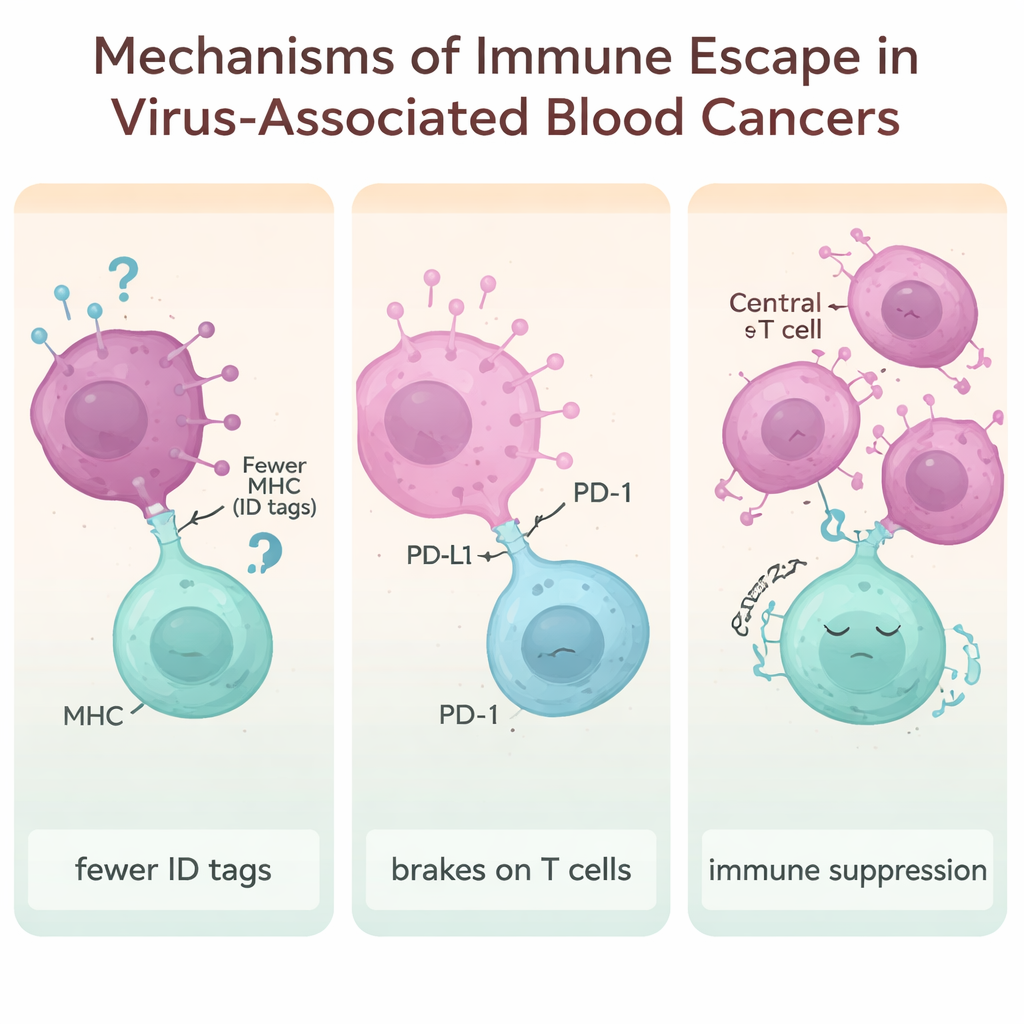
How Cancer Cells Learn to Hide from the Immune System
A central theme of the paper is “immune escape” – the tricks virus‑driven cancers use to stay invisible or untouchable to our defenses. One strategy is to reduce the molecular “ID badges” (called MHC molecules) on the cell surface that T cells normally scan to detect danger. Viral proteins from EBV, HTLV‑1, HIV and HBV can dial these ID badges down, so rogue cells are less likely to be recognized and attacked. Another tactic is to exploit immune “brakes” such as PD‑1, PD‑L1, CTLA‑4, LAG‑3 and TIM‑3. When these checkpoint molecules are overactive, T cells become exhausted and sluggish. Many virus‑associated lymphomas show very high levels of these brakes, especially in tumors linked to EBV and HBV.
Immune Cells Turned from Fighters into Peacekeepers
The review also highlights a subtler ploy: reshaping the immune environment so it favors the tumor. Regulatory T cells – a subset of T cells that normally prevent excessive inflammation – can be expanded and activated by long‑lasting viral infections. Signals such as the immune messengers IL‑10 and TGF‑β encourage these regulatory cells to multiply and to suppress nearby “killer” T cells. In chronic infections like HTLV‑1, HBV and HIV, this skewed balance creates a protective bubble around emerging cancer cells, making it even harder for the body to mount an effective response.
New Treatment Tactics Built on Viral Weak Spots
Armed with a clearer picture of these mechanisms, researchers are developing therapies that specifically target the viral machinery or the distorted immune landscape. Small‑molecule drugs are being designed to block key viral proteins from EBV and HTLV‑1, or even mark them for destruction inside the cell. Immune checkpoint inhibitors – antibodies that release the PD‑1/PD‑L1 or CTLA‑4 brakes – are being tested in a range of virus‑related lymphomas, often in combination with standard chemotherapy. Oncolytic viruses, engineered to selectively infect and burst cancer cells while stirring up a fresh immune response, are entering early trials. At the same time, more complex approaches such as virus‑specific T‑cell infusions, CAR‑T cell therapy, and therapeutic vaccines aim to rebuild or retrain the immune system to recognize viral targets and tumor cells more efficiently and for longer periods.
What This Means for Patients and Future Care
Taken together, the article argues that virus‑associated blood cancers are not just ordinary tumors with a virus attached. They are diseases whose very existence depends on a long‑running tug‑of‑war between viruses, blood cells and the immune system. By understanding exactly how viruses help cancer cells hide and thrive, doctors can design smarter combinations of treatments that block escape routes, awaken exhausted immune cells, and directly clear virus‑infected cells. Although many of these strategies are still in clinical trials, they point toward a future in which patients with these complex cancers may receive more personalized, effective therapies – and, in some cases, long‑lasting control or even cures.
Citation: Li, T., Wang, C., Xiao, Q. et al. Immune escape mechanisms and therapeutic advances in virus-associated hematological malignancies. Blood Cancer J. 16, 24 (2026). https://doi.org/10.1038/s41408-026-01453-7
Keywords: virus-associated lymphoma, immune escape, immune checkpoint therapy, CAR-T and T-cell therapy, therapeutic cancer vaccines