Clear Sky Science · en
Penpulimab combined with rituximab, high-dose methotrexate, and cytarabine (Pen-RMA) in newly diagnosed primary central nervous system lymphoma (PCNSL): a phase 2 trial
Why this brain cancer study matters
Primary central nervous system lymphoma is a rare but aggressive cancer that starts in the brain and nearby structures. Many patients are older, treatment can be harsh, and the disease often comes back within a few years. This study tests whether adding a newer type of immune therapy, called penpulimab, to standard chemotherapy can control the cancer for longer while keeping side effects manageable.
A tough-to-treat brain lymphoma
Primary central nervous system lymphoma grows in the brain, spinal cord, fluid around the brain, or eyes, without obvious disease elsewhere in the body. Even with strong chemotherapy based on a drug called high-dose methotrexate, most patients relapse in one to two years. Some combinations, like the MATRix regimen, improve survival but at the cost of serious side effects, especially in older or frail patients. Doctors urgently need regimens that keep the cancer at bay but are less punishing on the body.
Bringing the immune system into the fight
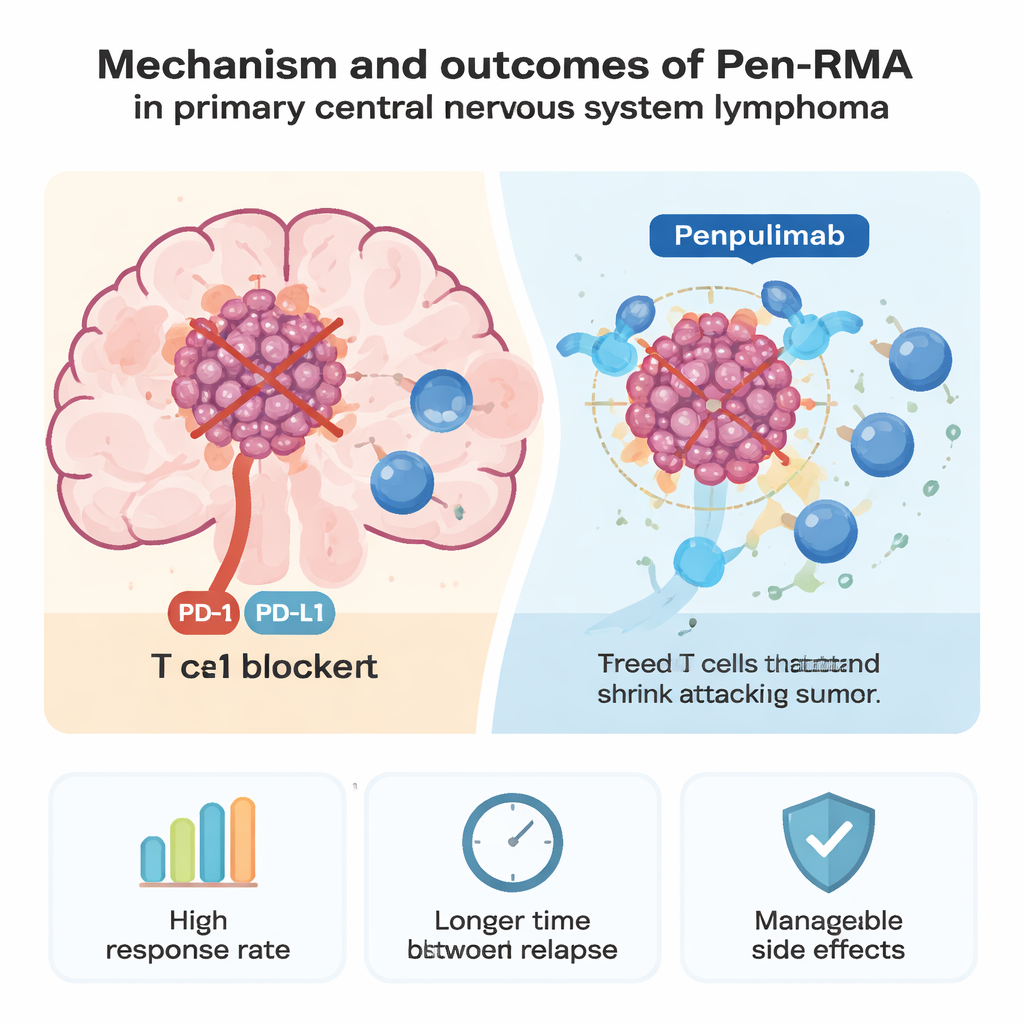
Recent research has shown that many of these lymphomas switch on a pathway called PD-1/PD-L1, which helps tumors hide from immune attack. Drugs that block PD-1, such as pembrolizumab and nivolumab, have already helped some patients whose brain lymphoma has come back after other treatments. Penpulimab is another PD-1–blocking antibody, designed to reduce certain immune-related side effects by altering one end of the molecule. The researchers designed a new combination—penpulimab plus rituximab and high-dose chemotherapy drugs methotrexate and cytarabine—called Pen-RMA, and tested it as the very first treatment in newly diagnosed patients.
How the trial was carried out
This phase 2 trial enrolled 26 adults with newly diagnosed primary central nervous system lymphoma at a single hospital in China; 23 patients were included in the main analysis. The median age was 65, and most had poor performance status and high-risk features. All patients received six cycles of Pen-RMA every three weeks. Younger, fitter patients who responded went on to receive a stem cell transplant, then penpulimab as maintenance. Older or transplant-ineligible patients who reached complete remission stayed on penpulimab alone, while those with partial remission received brain radiation plus penpulimab. Patients whose disease did not shrink or who worsened left the study. The main yardstick was how many patients were alive without their cancer growing again two years after starting treatment.
Stronger control with acceptable side effects
The results were encouraging. After the initial six cycles, almost all patients (95.7%) had their tumors shrink, and more than nine in ten (91.3%) had no visible disease on brain scans. With a median follow-up of about 29 months, the researchers could not yet determine the average time before the cancer returned or the average overall survival, because many patients were still doing well. An estimated 70.7% were alive without progression at two years, and 75.0% were alive overall—better than what the team had seen previously with a similar regimen lacking penpulimab. Side effects were common but mostly mild to moderate. Blood count drops, liver test changes, and infections were frequent, but only around 30% of patients had severe treatment-related problems, and immune-related complications were relatively rare.
Tracking tumor DNA in spinal fluid
The team also explored a more high-tech way to monitor disease: measuring traces of tumor DNA in the cerebrospinal fluid, the clear liquid that bathes the brain and spinal cord. In most patients, this cancer DNA was easier to detect in spinal fluid than in blood and closely matched the genetic changes seen in tumor tissue. Patients whose spinal fluid tumor DNA disappeared after induction treatment tended to fare better, while those who still had detectable DNA had shorter times before their disease returned or they died. This suggests that a simple spinal tap could one day help doctors identify patients who need closer follow-up or extra treatment even when scans look clean.
What this means for patients
For people facing primary central nervous system lymphoma, this study offers cautious optimism. Combining penpulimab with established chemotherapy produced high response rates and promising two-year outcomes, without an overwhelming increase in serious side effects, even in an older, high-risk group. Maintenance penpulimab after transplant or radiation also appeared practical and may help keep the disease in check. Because this was a relatively small, single-center trial without a direct comparison group, the findings need confirmation in larger, randomized studies. Still, the work points toward a future where brain lymphoma is treated not only with strong chemotherapy and radiation, but also with smartly timed immune therapy and precise monitoring using tumor DNA in spinal fluid.
Citation: Shen, HR., Wu, JZ., Yin, H. et al. Penpulimab combined with rituximab, high-dose methotrexate, and cytarabine (Pen-RMA) in newly diagnosed primary central nervous system lymphoma (PCNSL): a phase 2 trial. Blood Cancer J. 16, 23 (2026). https://doi.org/10.1038/s41408-026-01450-w
Keywords: primary central nervous system lymphoma, immunotherapy, PD-1 inhibitor, penpulimab, circulating tumor DNA