Clear Sky Science · en
Tooth-whitening treatment with potassium sodium tartrate: a non-invasive method that preserves enamel integrity
Why a Gentler Tooth-Whitening Option Matters
Many people want a brighter smile but worry about painful sensitivity and damage to their teeth from whitening products. This study explores a new kind of whitening ingredient, potassium sodium tartrate, that aims to remove stains as well as common peroxide products while keeping the hard outer shell of the tooth, the enamel, intact. It suggests that we may not have to choose between whiter teeth and long-term tooth health.
The Problem with Today’s Whitening Methods
Most whitening toothpastes and treatments rely on either strong chemicals or rough scrubbing. Peroxide-based products can be very effective because they break apart dark molecules that make teeth look yellow. However, they can also weaken enamel and dentin, the tissues that protect the sensitive inner parts of the tooth, leading to pain and higher risk of wear. Abrasive and acid-based methods can scrape or dissolve surface stains, but repeated use may roughen the tooth surface, cause sensitivity, or irritate the mouth. People who are younger, pregnant, or have weakened enamel are often advised to avoid strong peroxide treatments altogether.
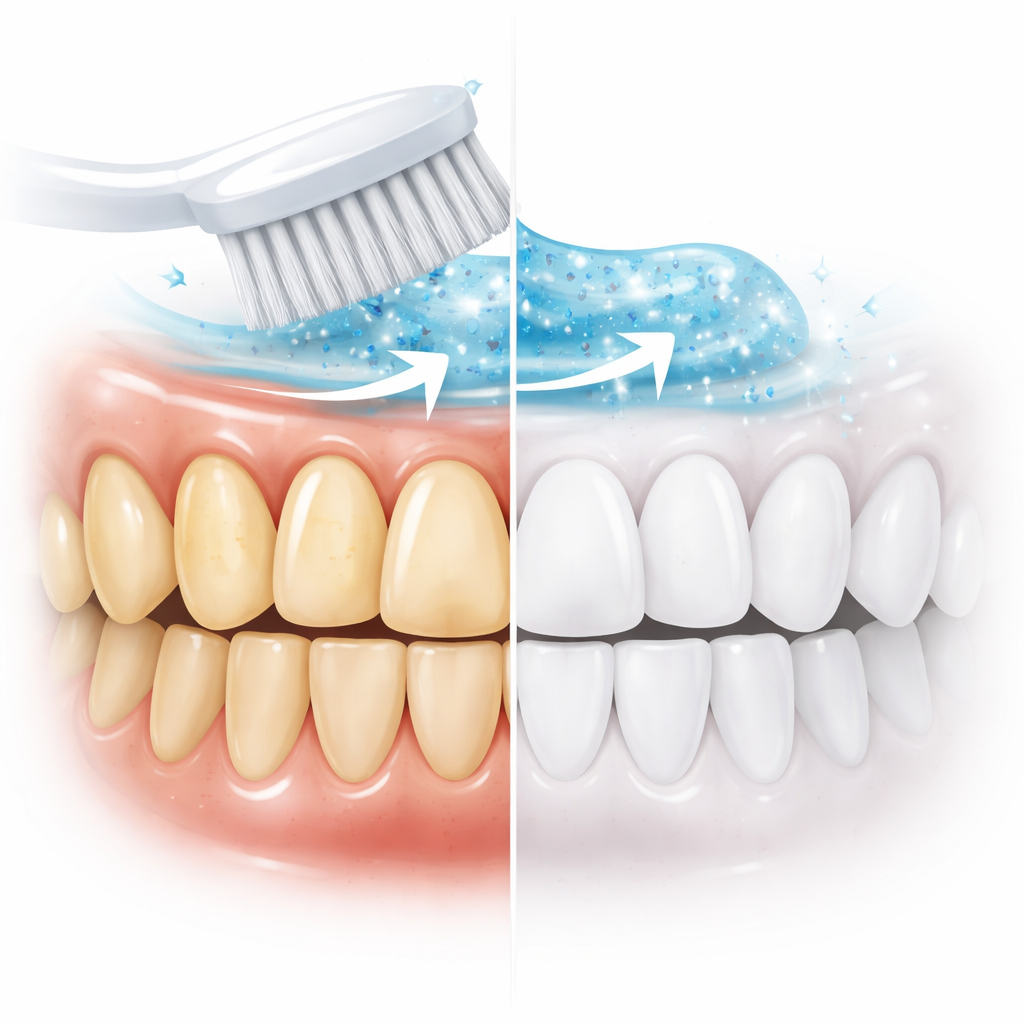
A New Approach: Using Brushing Energy Itself
The researchers tested potassium sodium tartrate, a crystal long used in electronics for its “piezoelectric” behavior, meaning it generates tiny electrical charges when pressed or flexed. When these particles are mixed into toothpaste and moved back and forth by brushing, they create short-lived reactive species—highly energized forms of oxygen—that can attack and break down stain molecules right where they sit on the tooth surface. Unlike pouring in large doses of peroxide, this process produces very small, localized bursts of cleaning activity that are triggered only where the toothbrush actually moves, promising a more targeted and potentially less destructive way to whiten.
How the Study Tested Safety and Whitening Power
To see how well this new ingredient works, the team ran two laboratory (in vitro) experiments using enamel blocks from animal and human teeth. First, they carefully stained bovine enamel with a mixture of coffee, tea and other components to mimic heavy everyday staining. These samples were treated with four options: a toothpaste containing 2% potassium sodium tartrate, a toothpaste with 2% carbamide peroxide (a standard whitening ingredient), a base toothpaste without any whitening agent, and plain water. The researchers measured how much stain was removed and how the hardness of the enamel changed, and they also used electron microscopes to look for surface damage.
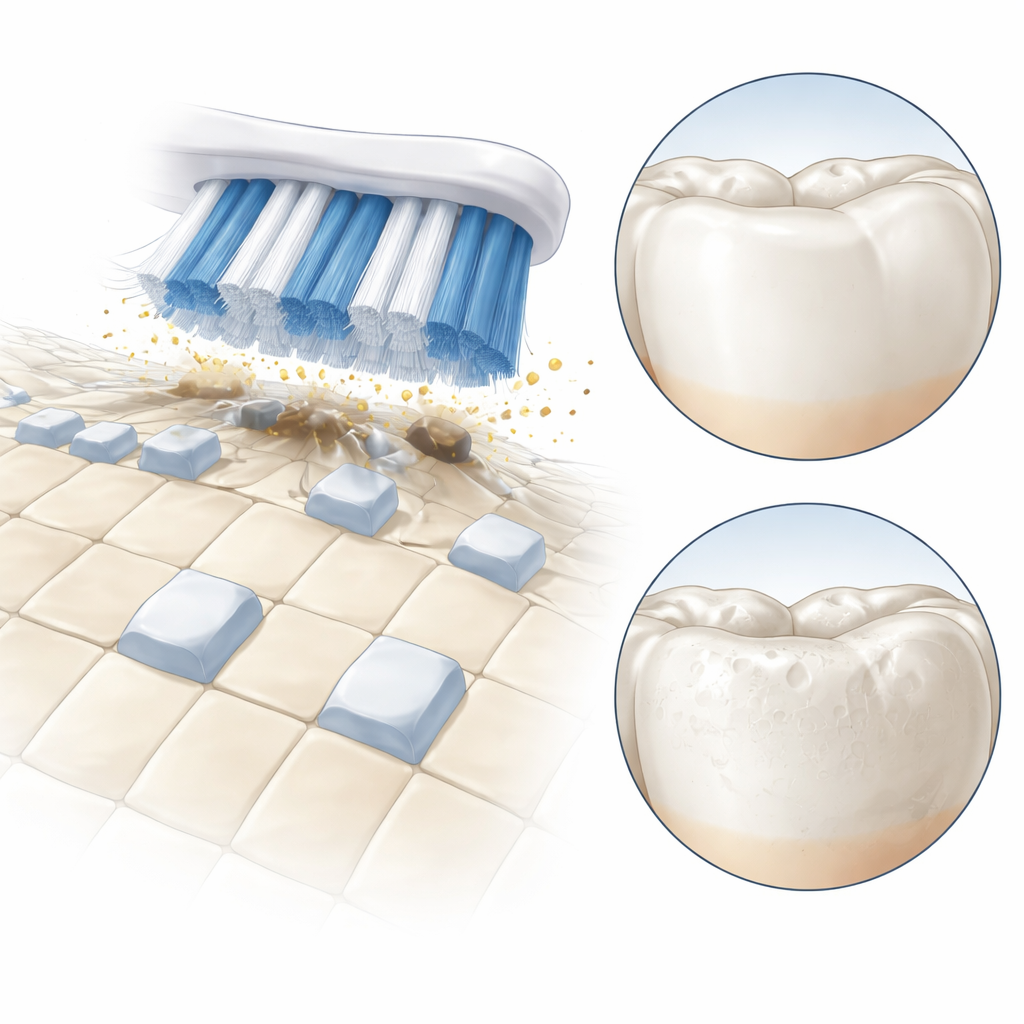
Whitening Results Without Enamel Damage
In the first experiment, the potassium sodium tartrate toothpaste removed stains just as effectively as the peroxide toothpaste, with both clearing about 30% of the staining under a tough test model. The base toothpaste removed roughly half as much, and water did almost nothing. But when the team examined enamel strength, a key difference emerged: the peroxide toothpaste caused a marked drop in surface hardness, a sign of demineralization that is linked to sensitivity and wear. By contrast, the potassium sodium tartrate toothpaste left enamel hardness unchanged and looked as smooth and intact as untreated samples under the microscope, similar to the non-whitening control. In a second experiment using stained human enamel and more commercial-style toothpastes, all whitening pastes brightened teeth by several shade steps over simulated weeks of brushing. After a month, the potassium sodium tartrate toothpaste with fluoride matched or slightly exceeded the whitening of the peroxide-fluoride paste, while still protecting enamel.
What This Could Mean for Everyday Toothpaste
The findings suggest that potassium sodium tartrate can deliver whitening on par with a benchmark peroxide formula while avoiding the softening and surface damage peroxide can cause. Because it must be “switched on” by the physical motion of brushing, it is best suited to toothpastes and similar products, not gels or strips that simply sit on the teeth. Still, this could mark a shift from the usual “whiten first, repair later” model—where we damage enamel and then try to fix it with remineralizing additives—to a “preserve while whitening” approach. If future clinical trials in people confirm these lab results, consumers may gain access to whitening toothpastes that provide noticeable brightening over weeks of regular use with far less risk to enamel health.
Citation: Ivanova, A., Buzova, V. Tooth-whitening treatment with potassium sodium tartrate: a non-invasive method that preserves enamel integrity. BDJ Open 12, 14 (2026). https://doi.org/10.1038/s41405-026-00405-4
Keywords: tooth whitening, enamel safety, toothpaste, potassium sodium tartrate, piezoelectric whitening